Abstract
Podocalyxin/podocalyxin-like protein 1 [PCLP1]/thrombomucin/MEP21 is a CD34-related sialomucin. We have performed a detailed analysis of its expression during murine development and assessed its utility as a marker of hematopoietic stem cells (HSCs) and their more differentiated progeny. We find that podocalyxin is highly expressed by the first primitive hematopoietic progenitors and nucleated red blood cells to form in the embryonic yolk sac. Likewise, podocalyxin is expressed by definitive multilineage hematopoietic progenitors and erythroid precursors in fetal liver. The level of podocalyxin expression gradually declines with further embryo maturation and reaches near-background levels at birth. This is followed by a postnatal burst of expression that correlates with the seeding of new hematopoietic progenitors to the spleen and bone marrow. Shortly thereafter, podocalyxin expression gradually declines, and by 4 weeks postpartum it is restricted to a rare population of Sca-1+, c-kit+, lineage marker- (Lin-) cells in the bone marrow. These rare podocalyxin-expressing cells are capable of serially reconstituting myeloid and lymphoid lineages in lethally irradiated recipients, suggesting they have HSC activity. In summary, we find that podocalyxin is a marker of embryonic HSCs and erythroid cells and of adult HSCs and that it may be a valuable marker for the purification of these cells for transplantation.
Introduction
The first circulating blood cells to form during vertebrate ontogeny are large, nucleated, primitive red cells produced in the blood islands of the yolk sac (YS). These cells are essential for the survival and rapid growth of the embryo, and in mice they emerge at 7.5 to 8.5 days postcoitum (dpc) (reviewed by Palis and Yoder1 and Keller et al2 ). Shortly thereafter, primitive YS hematopoietic precursors enter the newly formed vasculature and continue to divide and differentiate within the bloodstream until 13 dpc (E13),3 when they migrate through endothelial layers and enter new intraembryonic hematopoietic tissues.4 Subsequently, primitive YS precursors gradually disappear and are supplanted by definitive hematopoietic cells derived from the intraembryonic aorta-gonad-mesonephros (AGM).5 Soon after they appear, definitive hematopoietic precursors migrate across endothelial barriers and seed the fetal liver (FTL), which then becomes the major site of fetal hematopoiesis. Hematopoietic activity then shifts transiently to the fetal spleen (SPL) before becoming firmly established within the bone marrow (BM) just prior to and after birth.6 The cellular and molecular mechanisms that govern the shift of hematopoiesis to distinct anatomic sites during development are poorly understood.
Podocalyxin (also called podocalyxin-like protein 1 [PCLP1], Myb-Ets progenitor antigen 21 [MEP21], and thrombomucin) was first defined immunologically as a heavily sialylated and sulfated membrane protein expressed on the apical surface of glomerular epithelial cells, or podocytes, in kidney.7,8 Subsequently, podocalyxin was also detected on vascular endothelia, mesothelial cells lining the coelomic cavity, hemangioblasts, early hematopoietic progenitors, and platelets.9-15 Podocalyxin belongs to a large family of cell surface sialomucins and is most closely related to 2 other molecules: CD34 and endoglycan.13,16-19 All 3 molecules exhibit a strikingly similar genomic organization and pattern of alternative splicing, and they each encode proteins with similar biochemical motifs, suggesting they are derived from a common ancestral gene.13,17
Although their function remains enigmatic, recent experiments suggest that the CD34 family of sialomucins can play a role in blocking adhesion. For example, overexpression of podocalyxin in Chinese hamster ovary (CHO) or Madin-Darby canine kidney (MDCK) cells has been shown to block homotypic cell aggregation and the formation of cell-cell junctions.20 Similarly, we have recently shown that podocalyxin is up-regulated on the most highly invasive human breast cancers and that it is capable of disrupting cell polarity and junction formation in these cells.19 Conversely, we have shown that disruption of the podocalyxin-encoding gene in mice leads to perinatal lethality due to profound defects in kidney and gut formation that are consistent with excessive cell-cell adhesion.13 Interestingly, these mice do not present any obvious defects in tissues where podocalyxin and CD34 are coexpressed (hematopoietic and vascular cells), suggesting functional compensation between the CD34-related molecules. Consistent with this hypothesis, we have observed up-regulated expression of CD34 in podxl-/- tissues.13
To better understand podocalyxin's role in embryogenesis, we have now performed a detailed analysis of its expression in murine development with particular attention to hematopoietic tissues. Our data suggest that podocalyxin is expressed by the earliest hematopoietic progenitors (hematopoietic stem cells [HSCs]) and by erythroid precursors during embryonic development but that its expression on erythroid lineage cells ceases shortly after birth. Interestingly, erythroid expression of podocalyxin is rapidly reactivated in the adult in response to hemolytic anemia, suggesting its expression by these cells may reflect an erythropoietic stress response. On normal adult BM, we find that podocalyxin expression is restricted to a rare population of cells bearing a stem cell phenotype (lineage marker- [Lin-] Sca-1+ c-kit+ [LSK]). This was confirmed in transplantation experiments: Podocalyxin-positive LSK cells were capable of serially transplanting myeloid and lymphoid lineages in lethally irradiated mice. In summary, our data suggest that podocalyxin is indeed a highly restricted marker of long-term repopulating HSC in adult BM and that it is present on these cells throughout development. In light of its role in blocking cell adhesion in vitro and its prominent expression by hematopoietic precursors when hematopoietic tissues are actively seeded, we propose that podocalyxin may act as an invasive factor to make these cells more mobile and aid in the seeding of new hematopoietic compartments.
Materials and methods
Mice
Wild-type (wt), Ly5.1–enhanced green fluorescent protein (Ly5.1-EGFP), podxl+/-, cd34-/-, and podxl+/-cd34-/- mouse strains (all in a C57BL/6 background) were bred and maintained at The Biomedical Research Centre (BRC), University of British Columbia (UBC). Gestational age of embryos was determined by the date of copulation and morphologic landmarks as previously described.21 Mice used in phenylhydrazine (PHZ) experiments were all more than 8 weeks old.
Cell preparation
Single-cell suspensions were prepared from YS, FTL, SPL, and BM by drawing cells through a 25-gauge needle and then expelling them through a 70 μm nylon mesh. Cells were suspended in either phosphate-buffered saline (PBS) containing 0.05% NaN3 and 10% fetal calf serum (FCS) or in Hanks balanced salt solution (HBSS) without phenol red (Sigma Chemical, St Louis, MO), supplemented with 2% FCS.
Antibodies
The rat monoclonal antibody against podocalyxin (PCLP1) has been described previously.14 The immunoglobulin G1 (IgG1) control (Cedarlane, Hornby, ON) was used at 5 μg/mL to confirm the specificity of PCLP1 antibody, and in some cases podxl-/- tissues were used to verify the absence of nonspecific background staining. All other antibodies were obtained from BD PharMingen (San Diego, CA) and used as recommended by the manufacturer: biotinylated antibodies to platelet endothelial cell adhesion molecule-1 (PECAM-1)/CD31 (vascular endothelia), Ter119 (erythroid cells), Gr1 (8C5 [granulocytes]), B220 (6B2 [B lineage cells]), Mac1 (M1/70 [myelomonocytic cells]), and CD34 (RAM34 [progenitor cells]) and phycoerythrin (PE)–conjugated antibodies to Sca-1 (E13 [progenitor cells]), c-kit (2B8 [progenitor cells]), CD71 (C2 [proliferating cells and erythroblasts]), and CD45 (panhematopoietic).
Immunohistochemistry
Immunoperoxidase staining of frozen embryo sections was performed essentially as described previously.13 Antibodies were used at the following dilutions/concentrations: PCLP1 (1:1000 14 ), PECAM-1/CD31 (2.5 μg/mL [BD PharMingen]), and control IgG1 (5 μg/mL). Secondary antibodies were used as recommended by the manufacturer (Vectastain-ABC kit; Vector Laboratories, Burlingame, CA). Peroxidase activity was detected with diaminobenzidine (0.5 mg/mL) and 0.03% H2O2 in H2O followed by methyl green counterstain; coverslips were mounted with Permount (Fisher Scientific, Hampton, NH). An Olympus BH-2 microscope (10 ×/0.3 aperture Olympus objective), a Sensys1401E digital camera (Roper Scientific, Duluth, GA), and MetaVue Software (Universal Imaging, Downing-town, PA) were used for acquisition of images.
Flow cytometry and cell sorting
For single-, dual-, and triple-color immunofluorescence analyses, 106 primary cells were preincubated with 5% goat serum in PBS containing 10% FCS and stained for PCLP1 followed by goat antirat IgG Alexa-fluor 488–coupled antibody (Molecular Probes, Eugene, OR). After extensive washes and blocking in 2% rat serum, biotin-conjugated monoclonal antibodies followed by allophycocyanin (APC)–coupled streptavidin (BD PharMingen) and PE-conjugated monoclonal antibodies were added to the cells as described previously.22 All flow cytometric analyses were performed using a FACSCalibur (Becton Dickinson, San Jose, CA). 7-Amino actinomycin (7AAD) and light scatter properties were used to eliminate dead cells. Analyses were performed using CellQuest software (Becton Dickinson). Sorting of PCLP1+ cells was performed on a FACSVantage (Becton Dickinson). After 2 rounds of sorting, single cells were deposited in a 96-well flat-bottom culture plate using the CloneCyte device and software (Becton Dickinson). For in vivo injection, 104 sorted cells were collected and diluted to 1000 cells per 200 μL HBSS before injection into the tail vein of recipient mice for spleen colony-forming unit (CFU-S) assays.
Hematopoietic reconstitution
Cell sorting of PCLP1+ cells was performed on a FACSVantage cell sorter (Becton Dickinson). After 2 rounds of sorting, 10 to 500 Ly5.1 LSK cells expressing high levels of PCLP1 antigen or LSK cells negative for this antigen were deposited in a 96-well flat-bottom culture plate using the CloneCyte device and software (Becton Dickinson). About 300 000 Sca-1–depleted Ly5.2-GFP helper BM cells were added to the donor cells before tail vein injection into lethally irradiated Ly5.2 recipients. Reconstituted mice were periodically bled via the tail vein to monitor reconstitution by donor-marked (Ly5.1+) progenitors. Serial transplantations were performed after 4 months for animals presenting more than 10% BM chimerism. Total BM (106 cells) from the original recipients was reinjected into the tail vein of lethally irradiated Ly5.2 secondary and tertiary recipients. All flow cytometric analyses were performed using a FACSCalibur flow cytometer (Becton Dickinson). 7AAD-positive cells and cells with high obtuse scatter or nonphysiologically low forward scatter were excluded from all sorts and analyses to eliminate dead cells and debris.
Migration assays
FTL cells (E15) from wt, podxl-/-, or podxl-/-cd34-/- were either labeled with carboxyfluorescein diacetate succinimidyl ester (CFSE) at 5 μM per 107 cells per milliliter or carboxylic acid SNARF-1 acetate succinimidyl ester (CSA-SE) at 10 μM per 107 cells per milliliter (Molecular Probes) as indicated; the cells were then washed and mixed at an equal ratio before transplantation. Cells were recovered from the BM and SPL 16 hours after injection and were analyzed for the presence of either CFSE or CSA-SE by flow cytometry by acquiring 106 cells per sample using a FACSCalibur cytometer.
PHZ treatment
Hemolytic anemia was induced with PHZ (60 mg/kg body weight; Sigma Chemical) injected intraperitoneally for 2 consecutive days.23 The first day of injection was considered as time zero, and mice were killed at various times afterward as described. BM and SPL were removed under sterile conditions for further analyses.
Hematologic parameters
Blood was collected in heparinized microcapillaries (40 μL) from the tail vein of adult animals. Hematocrit (Hct) and reticulocyte counts were determined manually. At least 1000 cells were counted for each determination.
Reverse transcription–polymerase chain reaction (RT-PCR) analysis
Total RNA used for cDNA amplification was prepared with Trizol (Gibco, Gaithersburg, MD) following the manufacturer's protocol. For reverse transcription–polymerase chain reaction (RT-PCR) analysis, 2 μg RNA was converted into cDNA in a 20 μL reaction using the Thermoscript RT-PCR System (Gibco) primed with oligo-dT12-18. Two microliter aliquots of cDNA served as template for PCR using Taq-DNA polymerase (Gibco) and specific primers for podxl (forward primer: GAGGATTTGTGCACTCTACATGTG; reverse primer: TACTCGAGTGGGTTGTCATGGTAACC), cd34 (forward primer: TAGCACAGAACTTCCCAGCAAAC; reverse primer: CTCAGATCACAGTTCTGTGTCAGC), endgl (forward primer: ACGGGACGCTCTTTGTAGTGC; reverse primer: ATGCGGCCGCTCACAGGTGAGTGTCCTCCTC), and hypoxanthine-phosphoribosyl-transferase (hprt) used as a loading control (forward primer: CTCGAAGTGTTGGATACAGG; reverse primer: TGGCCTATAGGCTCATAGTG). PCR comprised 30 cycles of 94°C for 45 seconds, 58°C for 55 seconds, and 72°C for 2 minutes. PCR products were separated on a 2% agarose gel.
Results
Podocalyxin is expressed by all 3 embryonic germ layers during gastrulation
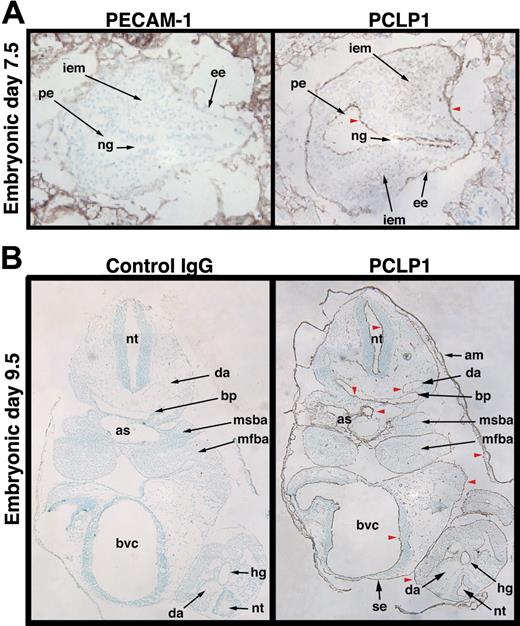
Previous reports in avians and mice have suggested that podocalyxin is expressed by the most primitive embryonic and adult hematopoietic progenitors12 and by hemangioblasts,14 respectively. To characterize the spatial localization of murine podocalyxin during embryogenesis and before the appearance of hematopoietic cells, we analyzed its expression during gastrulation. Immunoperoxidase staining of transverse sections in the distal region of the embryonic pole of 7.5 dpc mouse embryos with the antipodocalyxin antibody, PCLP1, revealed expression by the first intraembryonic mesoderm (Figure 1A). Thus, the mesodermal precursors that are thought to give rise to hemangioblasts (precursors for hematopoietic cells and vascular endothelia) express podocalyxin at the primitive streak stage. More surprisingly, we found that podocalyxin was not restricted to the mesoderm but was also expressed on the apical face of the neuroepithelium in the primitive ectoderm and by the primitive endoderm surrounding the embryos. This suggests that podocalyxin may be expressed by cells of the “epiblast,” which is reported to be the source of all 3 germ layers.24 Staining of the same-stage embryos with PECAM-1/CD31 showed no reactivity. After axial rotation of the embryos and inversion of the germ layers (9.5 dpc), podocalyxin was widely expressed by “boundary elements” (vasculature, mesothelial linings, and the luminal face of newly formed cavities) as has previously been reported in avians (Figure 1B; McNagny et al12 ).
Podocalyxin is expressed by all 3 germ layers and boundary elements during development. Frozen sections of embryos recovered at E7.5 (A) and E9.5 (B) were stained with antibodies to podocalyxin (PCLP1) and PECAM-1 (CD31) or control antibody and counterstained with methyl green. Red arrowheads highlight cavities whose luminal/boundary faces are coated with podocalyxin. These include the primitive ectoderm (pe), embryonic endoderm (ee), amnion (am), the branchial pouches (bp), the dorsal aorta (da), aortic sac (as), bulboventricular canal (bvc), surface ectoderm (se), and neuroepithelium of the neural tube (nt). Other labeled structures include the following: ng, neural groove; iem, intraembryonic mesoderm; msba, mandibular component of second branchial arch; mfba, mandibular component of first branchial arch, hg, hindgut. Original magnification × 10 (see “Materials and methods”).
Podocalyxin is expressed by all 3 germ layers and boundary elements during development. Frozen sections of embryos recovered at E7.5 (A) and E9.5 (B) were stained with antibodies to podocalyxin (PCLP1) and PECAM-1 (CD31) or control antibody and counterstained with methyl green. Red arrowheads highlight cavities whose luminal/boundary faces are coated with podocalyxin. These include the primitive ectoderm (pe), embryonic endoderm (ee), amnion (am), the branchial pouches (bp), the dorsal aorta (da), aortic sac (as), bulboventricular canal (bvc), surface ectoderm (se), and neuroepithelium of the neural tube (nt). Other labeled structures include the following: ng, neural groove; iem, intraembryonic mesoderm; msba, mandibular component of second branchial arch; mfba, mandibular component of first branchial arch, hg, hindgut. Original magnification × 10 (see “Materials and methods”).
Podocalyxin is transiently expressed in all hematopoietic compartments during embryonic and perinatal life
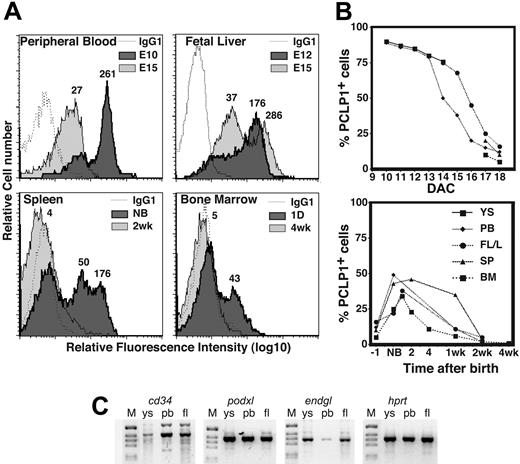
Although the highest levels of staining were observed on mesothelial cells and boundary elements, podocalyxin was also expressed at lower levels by hematopoietic cells within the vasculature. To more accurately assess the expression levels and distribution of podocalyxin on hematopoietic subsets, we used the more sensitive technique of immunofluorescence and flow cytometry. Cell suspensions from YS, peripheral blood (PB), FTL, SPL, and BM were analyzed at various stages of embryogenesis (Figure 2). Surprisingly, while podocalyxin expression is only detectable on a very small population of cells in adult hematopoietic compartments (see “Steady state podocalyxin expression in adult BM is restricted to a subpopulation of long-term repopulating HSCs”), it is highly expressed in all hematopoietic compartments during early embryonic life, and the expression level gradually decreases during ontogeny (Figure 2A). Furthermore, the frequency of PCLP1+ cells also decreases in all hematopoietic compartments during development. Essentially, all cells in day 10 YS and PB (97% ± 3%) are PCLP1+, and the level of expression gradually decreases over the next 5 days of development. Similarly, podocalyxin is expressed by 75% ± 5% of day 15 FTL cells and, again, the frequency of positive cells gradually decreases over the next 3 days of development (Figure 2B). Finally, both fetal BM and SPL acquire detectable populations of PCLP1+ cells (9% ± 4% and 17% ± 7%, respectively) as these tissues become hematopoietically active (E17) and, again, the frequency of positive cells gradually decreases to reach its nadir just prior to birth, with virtually all hematopoietic cells becoming PCLP1- (Figure 2B and data not shown). Strikingly, immediately after birth we observe a burst of podocalyxin expression by postnatal hematopoietic tissues reaching 52% of the total cells in the SPL and more than 30% of the cells in BM (Figure 2A-B). This, again, is followed by a gradual decline over the next 2 weeks of postnatal life until podocalyxin expression is detectable only on a rare subpopulation in the SPL and BM of adult mice. No reactivity was detected in age-matched podxl-/- mice, confirming the specificity of the PCLP1 antibody for podocalyxin and not other CD34 family members (Doyonnas et al13 and data not shown).
Podocalyxin is expressed by all primitive hematopoietic cells and then declines with development. (A) FACS profiles showing expression of PCLP1 by hematopoietic tissues at various stages of development (1 of 3 independent experiments). Mean relative fluorescence intensity (RFI) for each population is indicated numerically above each peak. (B) Graph showing the percentage of PCLP1+ cells in tissues during development. DAC indicates days after coitus. (C) Expression of CD34-related transcripts in YS, PB, and FTL of E15 embryos; podxl, endgl, cd34, and hprt indicate RT-PCR–amplified products for podocalyxin, endoglycan, CD34, and hypoxanthine-guanine phosphoribosyl transferase, respectively. The RT-PCR products are shown in a negative image of an agarose gel stained with ethidium bromide. M indicates molecular weight marker.
Podocalyxin is expressed by all primitive hematopoietic cells and then declines with development. (A) FACS profiles showing expression of PCLP1 by hematopoietic tissues at various stages of development (1 of 3 independent experiments). Mean relative fluorescence intensity (RFI) for each population is indicated numerically above each peak. (B) Graph showing the percentage of PCLP1+ cells in tissues during development. DAC indicates days after coitus. (C) Expression of CD34-related transcripts in YS, PB, and FTL of E15 embryos; podxl, endgl, cd34, and hprt indicate RT-PCR–amplified products for podocalyxin, endoglycan, CD34, and hypoxanthine-guanine phosphoribosyl transferase, respectively. The RT-PCR products are shown in a negative image of an agarose gel stained with ethidium bromide. M indicates molecular weight marker.
To independently confirm podocalyxin expression during embryonic development, we performed RT-PCR analysis to detect its mRNA (Figure 2C) and that of the related molecules, CD34 and endoglycan.13 All 3 molecules are expressed by YS, fetal blood, and FTL (Figure 2C) and therefore could potentially compensate for each other's loss, because it has previously been shown that deletion of podocalyxin leads to up-regulation of CD34 mRNA.13 The relative amount of podocalyxin mRNA, however, is about 10-fold and 20-fold higher than the amount of CD34 or endoglycan mRNA, respectively, in E15 YS and FTL by quantitative RT-PCR (data not shown).
In summary, our data suggest that podocalyxin expression corresponds to the onset of hematopoietic activity and the seeding of new hematopoietic compartments and that its expression gradually declines (in terms of both levels and frequency of positive cells) after hematopoietic tissue colonization.
Podocalyxin is expressed by primitive erythroid precursors and by primitive and definitive hematopoietic progenitors during embryonic and perinatal development
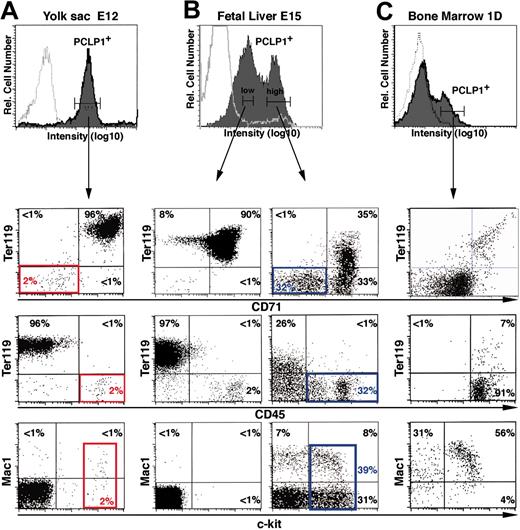
Most hematopoietic cells circulating between E10 and E12 are primitive, nucleated red blood cells of YS origin. The fact that nearly all the circulating cells are PCLP1+ suggests that most of the nucleated red blood cells express PCLP1. This was confirmed by 2-color immunofluorescence: Most PCLP1+ cells (96%) from the YS at E12 are double-positive for the erythroid markers Ter119 and CD71 (Figure 3A, lower panels). However, at this stage, there is also a small population of PCLP1+ cells that is CD71- and Ter119- but positive for the stem cell factor receptor c-kit and the panhematopoietic marker CD45 (Figure 3A; red boxes). Functional analyses by colony-forming assays in vitro and CFU-S assays in vivo showed that these cells give rise to erythroid (74%) and myeloid colonies (26%) (Table 1). This suggests that, in addition to its expression by primitive erythrocytes at 12 dpc, podocalyxin is also expressed by primitive myeloid or multilineage progenitors. Moreover, no in vitro or in vivo colonies develop from PCLP1-/low cells in the YS, suggesting that all primitive hematopoietic progenitors express podocalyxin (Table 1 and data not shown).
Colony-forming potential of PCLP1+ or PCLP1- cells from YS
. | CFCs* . | . | . | . | . | . | CFUs-S† . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| E12 YS . | No. of wells . | No. of colonies . | E . | GM . | MK . | Mix . | No. of colonies . | |||||
| PCLP1-Ter119- | 192 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| PCLP1+Ter119- | 192 | 31 | 23 | 8 | 0 | 0 | 5 | |||||
. | CFCs* . | . | . | . | . | . | CFUs-S† . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| E12 YS . | No. of wells . | No. of colonies . | E . | GM . | MK . | Mix . | No. of colonies . | |||||
| PCLP1-Ter119- | 192 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| PCLP1+Ter119- | 192 | 31 | 23 | 8 | 0 | 0 | 5 | |||||
The results are expressed as a sum of 2 to 3 separate analyses.
CFCs, colony-forming cells; E, erythroid colonies; GM, granulocyte or macrophage colonies; MK, megakaryocyte colonies; and Mix, mixed colonies.
In vitro methylcellulose CFCs from single cells seeded in 96-well plates. Number of wells analyzed and number of wells with 1 CFC is indicated
In vivo CFUs-S 12 days after injection of 1000 cells in irradiated recipients
Podocalyxin becomes progressively more restricted to hematopoietic progenitors during embryonic development. E12 YS (A), E15 FTL (B), and 1-day BM (C) were stained with anti-PCLP1 plus antirat IgG–Alexa-fluor 488, followed by Ter119-biotin plus streptavidin-APC and CD71-PE, CD45-PE, or Mac-1–PE. PCLP1low and PCLP1high cells were gated as indicated, and coexpression of hematopoietic markers in each population was assessed by 3-color FACS analyses. Solid curves indicate specific staining with the indicated antibodies. Open curves indicate isotype-matched control stains. The “gated” percentages of cells in each quadrant are indicated. Red boxes indicate colony-forming cells from E12 YS. Blue boxes indicate the hematopoietic precursor fraction of 15-day FTL.
Podocalyxin becomes progressively more restricted to hematopoietic progenitors during embryonic development. E12 YS (A), E15 FTL (B), and 1-day BM (C) were stained with anti-PCLP1 plus antirat IgG–Alexa-fluor 488, followed by Ter119-biotin plus streptavidin-APC and CD71-PE, CD45-PE, or Mac-1–PE. PCLP1low and PCLP1high cells were gated as indicated, and coexpression of hematopoietic markers in each population was assessed by 3-color FACS analyses. Solid curves indicate specific staining with the indicated antibodies. Open curves indicate isotype-matched control stains. The “gated” percentages of cells in each quadrant are indicated. Red boxes indicate colony-forming cells from E12 YS. Blue boxes indicate the hematopoietic precursor fraction of 15-day FTL.
While YS cells are mainly PCLP1high at E12 (Figure 3A), 15-day FTL exhibits 2 distinct populations of PCLP1+ cells: one PCLP1high population representing about 30% of the total cells and a second PCLP1low population representing more than 50% of the total cells (Figure 3B). Again, most PCLP1low cells at E15 are c-kit-/CD45-/Ter119+/CD71+ (90%), suggesting that they represent either previously formed primitive erythrocytes or newly formed definitive erythroblasts arising from the FTL. In contrast to the PCLP1+/CD45- hemangioblasts present in the AGM,14 32% of the PCLP1high cells found in the FTL are CD45+ and therefore already committed to a hematopoietic cell fate (Figure 3B, bottom panel). In addition, while the entire PCLP1low population is of erythroid origin, the PCLP1high population contains all cells expressing c-kit (39% of PCLP1high cells; Figure 3B, blue box), suggesting that this compartment contains all definitive hematopoietic precursors.25 Interestingly, these PCLP1high cells are negative for all mature markers (B220, CD3, and Gr-1) with the single exception of Mac-1 (Figure 3B), which is known to mark early hematopoietic progenitors/stem cells at this stage of development.26 We conclude that in FTL at E15, the PCLP1high population contains definitive embryonic hematopoietic progenitors.
To test if hematopoietic progenitors after birth continue to express podocalyxin, we examined the cell surface phenotype of PCLP1+ cells in the BM (Figure 3C) and SPL (data not shown) of perinatal mice. Although most PCLP1+ cells (80% to 85%) express CD71low, only a low percentage (less than 10%) express the late erythroid marker Ter119 in neonatal life. Moreover, at this stage, PCLP1+ cells are negative for most hematopoietic lineage markers (B220, Gr-1, and CD3), but many (60% to 70%) are c-kit+ (Figure 3C). We conclude that podocalyxin is expressed by erythroblasts and early progenitors in perinatal mice.
Steady-state podocalyxin expression in adult BM is restricted to a subpopulation of long-term repopulating HSCs
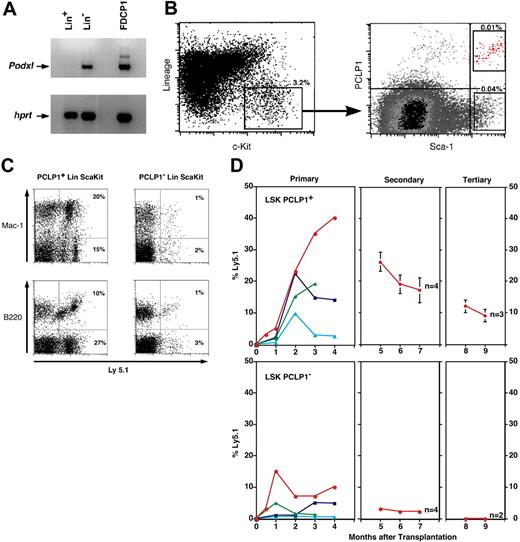
To test whether podocalyxin is a marker of adult murine HSCs, we examined its expression by cells with the appropriate cell surface phenotype. BM cells from 1-week-old mice were sorted into Lin- and Lin+ fractions and examined for PCLP1 expression by RT-PCR (Figure 4A). Using this method, we determined that podocalyxin is expressed exclusively by the Lin- fraction of BM known to contain HSCs. The Lin- population from adult mice (4 to 8 weeks) was then further fractionated on the basis of expression of the HSC antigens, Sca-1 and c-kit. This population, representing about 0.1% of the adult BM, is highly enriched in cells with long-term reconstituting capacity.27 Staining of adult LSK BM cells with anti-PCLP1 revealed both PCLP1+ (20%) and PCLP1- (80%) fractions (Figure 4B). Thus, in neonates and 4- to 8-week-old mice, podocalyxin is selectively expressed by a subset of BM cells with a surface phenotype consistent with HSCs.
We next tested for HSC function within LSK PCLP1+ or LSK PCLP1- fractions by transplantation of these cells into lethally irradiated mice. Low numbers of these cells were isolated by fluorescence-activated cell sorting (FACS) from the BM of Ly5.1 mice and transplanted into lethally irradiated congenic Ly5.2 mice. To ensure that these mice survived the short-term, posttransplantation leukopenia following irradiation, they were coinjected with host-type (Ly5.2) radioprotective GFP+ Sca-1- BM helper cells that have previously been shown to lack long-term repopulating capacity. Approximately 70% (21 of 30) of the recipients transplanted with between 10 and 500 PCLP1+ LSK cells showed significant levels of hematopoietic cell chimerism (more than 0.1%) in the PB for more than 4 months after transplantation. Animals exhibiting more than 1% engraftment are shown in Table 2 (12 mice) and Figure 4D (the top 4 mice for each group). In these animals, both myeloid and lymphoid lineages developed as determined by expression of Mac-1 and B220 on donor-derived cells, thus demonstrating multilineage reconstituting capabilities of LSK PCLP1+ HSCs (Figure 4C and first panel of 4D; Table 2). To further confirm the long-term reconstituting potential of these PCLP1+ cells, we analyzed their ability to contribute to the hematopoietic compartments of secondary and tertiary recipients in serial transplantation experiments (Figure 4D). A total of 106 BM cells from primary recipients with the highest chimerism were transplanted into lethally irradiated Ly5.2 secondary recipients. All mice analyzed after secondary and tertiary transplantation from PCLP1+ LSK HSCs had donor-derived myeloid and lymphoid PB cells (data not shown). We conclude that in BM, podocalyxin is expressed by cells with HSC activity.
The percentage of donor-derived Ly5.1+, myeloid (Mac1+ and Gr1+), and lymphoid (B220+ and CD3+) cells in the blood of mice 8 weeks after reconstitution with PCLP1+ or PCLP1- LSK cells
No. of Ly5.1+ cells injected . | No. of recipient mice . | Donor-derived myeloid cells, % . | Donor-derived lymphoid cells, % . |
|---|---|---|---|
| PCLP1+ LSK | |||
| 500 | 1 | 3 | 20 |
| 50 | 1 | 1 | 18 |
| 10 | 4 | 2-7 | 0.5-20 |
| PCLP1- LSK | |||
| 500 | 2 | 0.2-1 | 0.8-5 |
| 50 | 1 | 0.3 | 1.3 |
| 10 | 3 | 1-2 | 0.5-1 |
| Controls* | 3 | 0 | 0 |
No. of Ly5.1+ cells injected . | No. of recipient mice . | Donor-derived myeloid cells, % . | Donor-derived lymphoid cells, % . |
|---|---|---|---|
| PCLP1+ LSK | |||
| 500 | 1 | 3 | 20 |
| 50 | 1 | 1 | 18 |
| 10 | 4 | 2-7 | 0.5-20 |
| PCLP1- LSK | |||
| 500 | 2 | 0.2-1 | 0.8-5 |
| 50 | 1 | 0.3 | 1.3 |
| 10 | 3 | 1-2 | 0.5-1 |
| Controls* | 3 | 0 | 0 |
Control animals did not receive Ly5.1+ cells
PCLP1+ cells in adult BM are a subpopulation of HSCs. (A) RT-PCR analyses of Lin+ (stem cell depleted) and Lin- (stem cell enriched) BM from 1-week-old mice and the PCLP1+ cell line, FDCP-1. (B) FACS profiles showing expression of PCLP1 by a subset of the LSK HSC population in adult BM. Percentages indicate total frequency in BM. (C) Donor-derived lymphoid (B220) and myeloid (Mac-1) composition of BM 16 weeks after reconstitution of lethally irradiated mice with 500 cells of either PCLP1+ or PCLP1- LSK BM cells. Ly5.1+ cells are donor derived. (D) Percentage (determined by frequency of Ly5.1+ staining) of donor-derived cells in the blood of mice reconstituted with PCLP1+ and PCLP1- LSK cells after primary, secondary, and tertiary transplantation. In primary recipients, each colored line represents an individual mouse reconstituted with either 500, 50, or 10 enriched stem cells. In each case, 106 BM cells from the most highly reconstituted primary recipients (red) were used for subsequent transplantations in secondary and tertiary recipients.
PCLP1+ cells in adult BM are a subpopulation of HSCs. (A) RT-PCR analyses of Lin+ (stem cell depleted) and Lin- (stem cell enriched) BM from 1-week-old mice and the PCLP1+ cell line, FDCP-1. (B) FACS profiles showing expression of PCLP1 by a subset of the LSK HSC population in adult BM. Percentages indicate total frequency in BM. (C) Donor-derived lymphoid (B220) and myeloid (Mac-1) composition of BM 16 weeks after reconstitution of lethally irradiated mice with 500 cells of either PCLP1+ or PCLP1- LSK BM cells. Ly5.1+ cells are donor derived. (D) Percentage (determined by frequency of Ly5.1+ staining) of donor-derived cells in the blood of mice reconstituted with PCLP1+ and PCLP1- LSK cells after primary, secondary, and tertiary transplantation. In primary recipients, each colored line represents an individual mouse reconstituted with either 500, 50, or 10 enriched stem cells. In each case, 106 BM cells from the most highly reconstituted primary recipients (red) were used for subsequent transplantations in secondary and tertiary recipients.
Podocalyxin expression on adult erythrocyte precursors is reactivated by hemolytic anemia
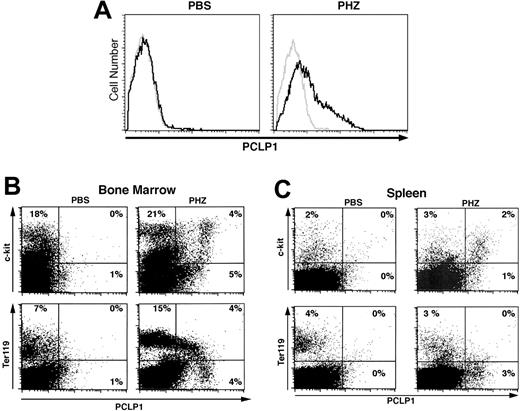
Because PCLP1 expression correlates with rapid expansion of erythroid progenitors during development, we tested whether its expression could be reactivated in adult erythroid cells by hemolytic anemia. Adult mice were injected with PHZ to induce profound anemia,28 and SPL and BM cells were monitored for PCLP1 expression at daily intervals during the recovery phase when erythropoiesis is enhanced (Table 3). During normal erythropoiesis, progenitors progress through a very well-defined set of maturational steps: from proerythroblasts (Ter119low/CD71high), to basophilic erythroblasts (Ter119high/CD71high), to polychromatophilic erythroblasts (Ter119high/CD71med), and finally to orthochromatophilic erythroblasts (Ter119high/CD71low).29 In nonanemic mice, most Ter119high cells are late orthochromatophilic erythroblasts (Ter119high/CD71low). We found that the PCLP1+ cells, which normally represent less than 1% of the cells in adult BM, increase rapidly after PHZ treatment and reach a peak frequency of 24% ± 4% of the total cells on the third day, well before the appearance of reticulocytes in the PB (Table 3 and Figure 5A). Moreover, we observed the expansion of PCLP1+ c-kit+ cells in BM just prior to the expansion of Ter119low/CD71high proerythroblasts (Figure 5B). Three-color staining revealed that PCLP1+/c-kit+ cells are predominantly Ter119- (more than 90%), while the bulk of the Ter119+/PCLP1low cells are c-kit- (more than 85%, data not shown). This suggests that podocalyxin expression is rapidly increased on very early c-kit+ progenitors that can potentially emigrate from the BM to enhance extramedullary erythropoiesis. Although podocalyxin expression is largely restricted to BM, a low frequency of PCLP1+ cells is also present in the SPLs of anemic animals (Figure 5C).
PCLP1/podocalyxin expression is reactivated by PHZ-induced hemolytic anemia, and its deficiency induces accumulation of erythrocyte precursors within the hematopoietic compartments
. | Hematocrit, % . | Reticulocyte index,* % . | PCLP1+ cells in BM, % . |
|---|---|---|---|
| PBS-treated mice | |||
| Day 2 | 45 ± 5 | 2 ± 1 | 1 ± 1 |
| PHZ-treated mice | |||
| Day 2† | 32 ± 3 | 4 ± 3 | 11 ± 2 |
| Day 3 | 23 ± 4 | 9 ± 3 | 24 ± 4 |
| Day 6 | 41 ± 6 | 67 ± 5 | 19 ± 3 |
| Day 9 | 52 ± 4 | 12 ± 2 | 3 ± 1 |
. | Hematocrit, % . | Reticulocyte index,* % . | PCLP1+ cells in BM, % . |
|---|---|---|---|
| PBS-treated mice | |||
| Day 2 | 45 ± 5 | 2 ± 1 | 1 ± 1 |
| PHZ-treated mice | |||
| Day 2† | 32 ± 3 | 4 ± 3 | 11 ± 2 |
| Day 3 | 23 ± 4 | 9 ± 3 | 24 ± 4 |
| Day 6 | 41 ± 6 | 67 ± 5 | 19 ± 3 |
| Day 9 | 52 ± 4 | 12 ± 2 | 3 ± 1 |
The results are expressed as the mean ± SD of 3 separate analyses.
Reticulocyte index is expressed as corrected reticulocyte count. Corrected reticulocyte count (%) = reticulocyte count (%) × 45 (normal hematocrit rate)
Twenty-four hours after second injection of PHZ
Podocalyxin and CD34 are required for efficient short-term homing to hematopoietic tissues
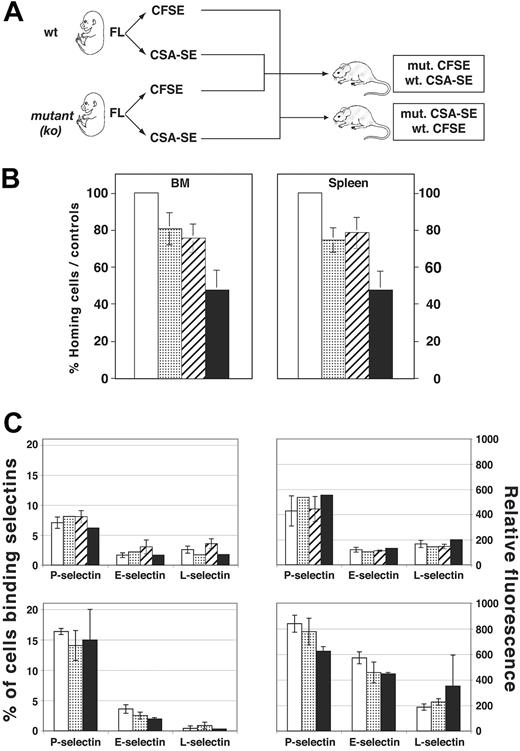
Recently, we and others have shown that podocalyxin and CD34 play an important role in blocking hematopoietic and epithelial cell adhesion and cell-cell contact.19,20,30 Based on these observations and the fact that podocalyxin is abundantly expressed by embryonic hematopoietic cells as they seed new tissues, we postulated that these molecules may play a role in facilitating the exit and entry of hematopoietic cells into new environments. To test this hypothesis, we isolated FTL cells from wt, cd34-/-, podxl-/-, and podxl-/-cd34-/- animals and assayed them for short-term homing to BM. Briefly, wt or mutant cells were labeled with the fluorochromes CFSE (green) or CSA-SE (red) mixed in equal proportions and injected into lethally irradiated recipient mice (Figure 6A). It has been demonstrated previously that the number of cells that home to BM in murine transplantation experiments plateaus between 3 hours and 24 hours, with no effect on apoptosis and expansion.31,32 We therefore analyzed recipient mice 16 hours after transplantation for the relative proportion of labeled cells in the SPL and BM. To rule out the possibility of selective dye toxicity, experiments were performed in duplicate and the fluorescent tags were reversed. The results of these analyses are summarized in Figure 6B. Although FTL progenitors of each genetic background (wt, cd34-/-, podxl-/-, and cd34-/-podxl-/-) were detectable in the BM and SPL of recipient mice, wt cells exhibited the most robust migration to both hematopoietic organs. Based on the number of cells injected and the frequency of cells recovered, we estimate that 12% of input cells were recovered in representative experiments with wt cells. After normalization to the number of wt cells harvested, the relative proportions of donor cells harvested from the BM and SPL of the recipients was determined. The cd34-/- or podxl-/- FTL cells are 20% to 30% less efficient in migrating to the SPL and BM of recipient mice (Figure 6B). Loss of both CD34 and podocalyxin had an additive effect, and these cells are less efficient in BM and SPL homing (Figure 6B).
PHZ-induced anemia reactivates podocalyxin expression in adult erythroid cells. (A) Representative FACS histograms showing PCLP1+ cells in BM of adult mice 3 days after injection with either PBS (left) or PHZ (60 mg/kg; right). Cells were stained with PCLP1 (black line) or control mAb (gray line) and counterstained with Alexa-fluor 488–conjugated antirat IgG. (B-C) Coexpression of podocalyxin with hematopoietic progenitor and erythroid markers on BM cells from PBS- and PHZ-injected mice 2 days after the first injection. BM (B) and SPL (C) cells were stained with anti-PCLP1 as in panel A and counterstained with biotinylated Ter119 plus streptavidin-PE or c-kit–PE. Percentages are determined according to quadrant markers.
PHZ-induced anemia reactivates podocalyxin expression in adult erythroid cells. (A) Representative FACS histograms showing PCLP1+ cells in BM of adult mice 3 days after injection with either PBS (left) or PHZ (60 mg/kg; right). Cells were stained with PCLP1 (black line) or control mAb (gray line) and counterstained with Alexa-fluor 488–conjugated antirat IgG. (B-C) Coexpression of podocalyxin with hematopoietic progenitor and erythroid markers on BM cells from PBS- and PHZ-injected mice 2 days after the first injection. BM (B) and SPL (C) cells were stained with anti-PCLP1 as in panel A and counterstained with biotinylated Ter119 plus streptavidin-PE or c-kit–PE. Percentages are determined according to quadrant markers.
Impaired short-term homing in vivo despite identical selectin ligand expression. (A) Experimental design for short-term homing experiment. Mutant 15-day FTL cells were isolated, genotyped, and labeled with either CFSE or CSA-SE. These were mixed in a 1:1 ratio with wt cells labeled with the opposite fluorescent tracker prior to intravenous injection into irradiated recipients. Relative frequency of CFSE- and CSA-SE–labeled cells in BM and SPL was determined 16 hours later by FACS. In control experiments, the fluorescent labels were reversed to rule out dye toxicity. (B) Percent homing efficiency of wt or mutant FTL cells to SPL (right panel) or BM (left panel). The wt cells were assigned a frequency of 100%. Results from CFSE and CSA-SE homing were pooled together and standard deviation calculated for the number of mice analyzed (n = 4 per genotype). (C-D) The percentage and mean RFI of E-, P-, and L-selectin binding to wt, cd34-/-, podxl-/-, or podxl-/-cd34-/- FTL cells. Shown are results from 2 independent experiments. (B-D) □ indicates wt; ▦, cd34-/; ▨, podxl-/-; and ▪, podxl-/-cd34-/-.
Impaired short-term homing in vivo despite identical selectin ligand expression. (A) Experimental design for short-term homing experiment. Mutant 15-day FTL cells were isolated, genotyped, and labeled with either CFSE or CSA-SE. These were mixed in a 1:1 ratio with wt cells labeled with the opposite fluorescent tracker prior to intravenous injection into irradiated recipients. Relative frequency of CFSE- and CSA-SE–labeled cells in BM and SPL was determined 16 hours later by FACS. In control experiments, the fluorescent labels were reversed to rule out dye toxicity. (B) Percent homing efficiency of wt or mutant FTL cells to SPL (right panel) or BM (left panel). The wt cells were assigned a frequency of 100%. Results from CFSE and CSA-SE homing were pooled together and standard deviation calculated for the number of mice analyzed (n = 4 per genotype). (C-D) The percentage and mean RFI of E-, P-, and L-selectin binding to wt, cd34-/-, podxl-/-, or podxl-/-cd34-/- FTL cells. Shown are results from 2 independent experiments. (B-D) □ indicates wt; ▦, cd34-/; ▨, podxl-/-; and ▪, podxl-/-cd34-/-.
Previously, it has been shown that, when expressed by high endothelial venules, podocalyxin and CD34 are glycosylation-dependent adhesive ligands for L-selectin on migrating leukocytes. Although the ability of L-selectin to bind to CD34 and podocalyxin is dependent on an unusual lymph node–specific glycosylation that has not been observed on most vasculature or hematopoietic cells, it remained a formal possibility that the defects observed in progenitor cell homing could be due to loss of these molecules as selectin ligands. To rule out this possibility, FTL from wt, cd34-/-, podxl-/-, and podxl-/-cd34-/- embryos were stained with recombinant, soluble forms of P-, E-, and L-selectin, the only molecules known to play a role in carbohydrate-dependent adhesion of hematopoietic cells. The results are shown in Figure 6C-D. Whether stained individually or with a cocktail of soluble selectins, we found no significant difference in the frequency or the level of selectin ligand expression by wt or mutant FTL cells. We conclude that the short-term homing defects observed with cd34-/-, podxl-/-, and podxl-/-cd34-/- cells are not due to the loss of these molecules as selectin ligands and are more likely a result of these molecules providing a “Teflon” effect in preventing nonspecific adhesion.
Interestingly, although hematopoietic progenitors from double-deficient mice seem to be highly impaired in their ability to migrate and home to hematopoietic tissues, they retain their competence for normal differentiation into mature hematopoietic lineage cells. Indeed, we have found that injection of between 2.5 × 105 and 106 double-deficient FTL cells is sufficient to fully reconstitute lethally irradiated recipient mice. Likewise, we have seen no detectable differences in the frequencies or subsets of hematopoietic lineage cells in the FTL of single- or double-deficient embryos (Doyonnas et al13 and data not shown). Thus, despite their impaired short-term migration, the data suggest that, when provided in sufficient numbers and given sufficient time, these cells are able to find the appropriate niches for proliferation and maturation. In summary, our preliminary data suggest that CD34 and podocalyxin have overlapping roles in aiding the short-term migration of hematopoietic progenitors.
Discussion
During ontogeny, sequential changes in the anatomic sites of hematopoiesis are believed to occur through the migration of hematopoietic stem and progenitor cells from the extraembryonic YS and/or the AGM into the FTL, the SPL, and the BM of developing embryos. Evidence suggests that most migrating embryonic hematopoietic progenitors are contained within the cell fraction expressing the sialomucin, CD34.33,34 Despite the use of this molecule as a selective marker for hematopoietic precursor cells, deletion of the encoding gene in mice does not appear to significantly perturb the migration of hematopoietic progenitors during embryogenesis or in adult life.35,36 We and others have reported the discovery of a new family of CD34-related molecules,12,13,16,18 and gene-targeting studies have suggested that members of this class of sialomucins may functionally compensate for each other's loss in tissues where they are coexpressed.13 In the present study, we have analyzed the hematopoietic expression of one of these new family members, podocalyxin, during ontogeny. The results suggest that podocalyxin, in contrast to CD34, is expressed by all 3 embryonic germ layers during gastrulation and that its expression is maintained on both primitive and definitive embryonic hematopoietic progenitors. Moreover, we find that podocalyxin is also expressed by nucleated red blood cells and their precursors.
At the earliest stages of development (E8 to E12), podocalyxin is expressed by essentially all circulating hematopoietic cells (97% ± 3%), including all cells capable of forming colonies in vitro and in CFU-S assays. Previous reports have suggested that these cells express the progenitor cell antigens CD34, Sca-1, and c-kit; consistent with this, we find that all cells expressing these markers coexpress podocalyxin. Similarly, we find that early hematopoietic progenitors in the FTL, SPL, and BM also express podocalyxin. By 2 weeks after birth, podocalyxin is nearly undetectable in BM, while a sizable population retains expression of CD34 and c-kit (more than 5%) and, as published previously, these cells contain the majority of colony-forming units. Here we show that the few remaining PCLP1+ cells (less than 0.5%) in adult BM contain long-term repopulating HSCs with the capacity to reconstitute lethally irradiated primary, secondary, and tertiary recipients. All of the experiments described here were performed with mice not older than 8 weeks. Notably, CD34 has been shown to be a marker of long-term repopulating cells only until 10 weeks of age.37 Subsequently, most long-term repopulating cells are CD34-. CD34, however, is reexpressed by these cells after mobilization with G-CSF or after transplantation (reviewed by Ogawa38 and Zanjani et al39 ). Thus, the data suggest that CD34 expression probably reflects the activation/cell cycle status of long-term repopulating cells. Interestingly, we have found that podocalyxin continues to be expressed by the LSK population in the BM of older mice (more than 10 weeks), although we have not yet shown that these cells have long-term repopulating activity. Thus, the mechanisms that regulate the expression of podocalyxin and CD34 by stem cells may differ.
In addition to its expression by hematopoietic progenitors, we have shown by cytologic, flow cytometric, and functional analyses that most cells expressing podocalyxin during embryonic and neonatal life are affiliated with the erythroid lineage. Thus, podocalyxin marks essentially all nucleated erythroid cells at the earliest stages of hematopoietic development and the bulk of the definitive erythroid progenitors in FTL and neonatal BM and SPL. Our data suggest that expression of podocalyxin by these cells (as well as hematopoietic progenitors) correlates closely with high rates of erythropoiesis and with the expansion and seeding of erythroid progenitors to new hematopoietic microenvironments. Thus, 10-day YS, 15-day FTL, and newborn BM and SPL each exhibit a burst of podocalyxin expression by erythroid progenitors that declines as erythropoiesis shifts to a new anatomic site or reaches steady state. Moreover, we have shown that podocalyxin expression is rapidly reactivated by erythropoietic stress.
Interestingly, erythropoiesis during development is in many ways analogous to stress-induced erythropoiesis, because embryos sustain a much higher rate of red cell production to compensate for their lack of a reservoir of erythropoietic progenitors.40,41 It therefore appears that podocalyxin is expressed by early erythroblasts only in situations where maximal red cell output is essential. Several genes have been described that appear to be critical for fetal erythropoiesis but not for steady-state erythropoiesis in adults. These include the transcription factors activating transcription factor 4 (ATF4), adenoviral E2 gene promoter-binding factor 4 (E2F4), and signal transducer and activator of transcription 5a/5b (Stat5a/5b).42-44 Inactivation of each of these genes results in severe anemia during neonatal and fetal life but relatively normal erythropoiesis in adults. Interestingly, most of these deficiencies lead to increased sensitivity to chemically induced anemia, further suggesting a deficiency in response to erythropoietic stress. It will now be interesting to see if the expression of any of these factors correlates with the high level of podocalyxin expression on embryonic erythroid cells or the lower level observed on PHZ-treated erythroid cells. Alternatively these differences could correlate with turnover rate of this antigen on embryonic versus adult erythroid cells. The induction of podocalyxin expression does not appear to be hypoxia inducible, because preliminary data showed treatment of PCLP1+ cell lines (Baf3 or FDCP-1) or primary BM cells with desferroxamine or CoCl had no effect on podocalyxin expression (although they did augment CD71 expression). Likewise, we were unable to detect a change in expression in mice treated with 5-fluorouracil (5-FU). Thus, the mechanism of podocalyxin up-regulation during anemia will require a more detailed analysis of the gene's regulatory elements.
How do CD34-related proteins aid in hematopoietic cell migration? Recently it has been shown that ectopic expression of podocalyxin in adherent cells leads to a profound block in cell-cell adhesion.20 For example, overexpression in CHO and MDCK cells (in vitro) was found to result in an expression level–dependent block in homotypic aggregation and to inhibit the formation of cell-cell tight junctions, respectively.20 Likewise, we have found that podocalyxin is selectively up-regulated in metastatic breast cancers with poor outcome and that ectopic expression in more benign breast cell lines leads to loss of polarity and cell-cell junctions.19 Conversely, we have shown that deletion of the podocalyxin-encoding gene in mice leads to perinatal lethality due to excessive cell-cell adhesion in a number of embryonic structures. For example, podocalyxin loss from the developing mesothelium leads to excessive adhesion between the embryonic intestine and the umbilical cord, resulting in herniation of the gut in many embryos. In addition, all mice lacking podocalyxin die perinatally due to high blood pressure and kidney failure from excessive adhesion and failure to remodel tight junctions between kidney podocytes.13 Finally, we have recently shown that mast cells selectively express CD34, and deletion of the encoding gene leads to homotypic mast cell aggregation.30 Thus, all of the available functional data suggest that podocalyxin and CD34 play a direct role in blocking cell-cell adhesion.
Based on these observations and the abundant expression of podocalyxin by boundary elements early in development (Figure 1) we propose that, on hematopoietic progenitors, podocalyxin and CD34 perform a similar function and act as molecular Teflon to enhance the ability of these cells to cross endothelial barriers and enter/exit primary hematopoietic tissues. This is supported by short-term homing assays that reveal a gene dose–dependent impairment in the ability of cells to enter the BM. That these molecules function as invasive factors rather than specific homing receptors is supported by the observation that wt and mutant cells show an identical ability to bind selectins, which are the only known receptors for these molecules.
Although CD34 and podocalyxin are both expressed by hematopoietic progenitors, only podocalyxin is expressed by most erythroid cells during embryonic development and in response to anemia. We postulate that the high-level expression in YS and subsequently FTL may reflect a role in the migration of erythroid progenitors and more mature erythroblasts through the endothelial barriers in these tissues and into new sites of erythropoietic activity. In the YS most of the inner cells of the blood island progressively lose their intercellular attachments as they differentiate into primitive erythroblasts and then leave these anatomic sites. Likewise, in the FTL, hematopoietic precursors cross the endothelial barrier of the vasculature to reach the PB, where they can circulate freely and later seed the developing BM and SPL. Finally, severe anemia leads to an efflux of erythroid progenitor cells from the BM and to the establishment of extramedullary sites of erythropoiesis.44,46 Although these phenomena have been well documented, the molecular mechanisms governing the release of cells from these sites has not been clarified. It will now be interesting to see if podocalyxin, by acting as molecular Teflon, plays an important role in this process, and this work is in progress.
Prepublished online as Blood First Edition Paper, February 8, 2005; DOI 10.1182/blood-2004-10-4077.
Supported by Canadian Institutes of Health Research (CIHR) grant MT-15477 (K.M.M.), a Grant-in Aid from the Heart and Stroke Foundation of B.C. & Yukon, a fellowship from the National Sciences and Engineering Research Council of Canada (NSERC) (J.S.N.), the Michael Smith Foundation for Health Research (E.D.), and the Heart and Stroke Foundation of B.C. & Yukon (E.D.). K.M.M. is a Scholar of the Canadian Institutes of Health Research and the Michael Smith Foundation for Health Research.
R.D. and J.S.N. contributed equally to this study.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
The authors thank Lea Wong and Samantha Kleczkowski for their help with animal care, Yanet Valdez for her excellent technical support, and Andy Johnson for expert assistance in flow cytometric analyses. We also thank Helen Merkens for motivating discussions. Helpful suggestions and critical evaluation of this manuscript were provided by Drs Thomas Graf, Hermann Ziltener, John Schrader, and Fabio Rossi.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal