Abstract
Immunoglobulin (Ig) variable (V) region idiotypes (Id's) are highly tumor-specific antigens produced by B-lymphoma cells and are promising targets for immunotherapy. Id vaccination has proven effective in experimental mouse models and may possibly prevent recurrence of B lymphomas in humans. It has previously been shown that anti-Id antibodies protect against B-cell lymphoma in the absence of T cells. We here demonstrate in a T-cell–receptor transgenic mouse model that the contrary is also true: Id-specific CD4+ T cells can protect against Id+ B-lymphoma cells in the absence of B cells, antibodies, and CD8+ T cells. Moreover, Id-specific CD4+ T cells have a curative potential since they could be transferred as late as 17 days after subcutaneous tumor cell injection of severe combined immunodeficiency (SCID) mice and still abrogate tumor development in about 50% of mice. Such mice undergo an acute inflammatory swelling with infiltration of neutrophils at the site of tumor injection, which subsides over weeks, with some mice cured and delayed emergence of lymphomas in other mice. Adoptively transferred CD4+ T cells accumulated in the tumor and were activated (CD69+). In vitro experiments demonstrated that memory, but not naive, Id-specific CD4+ T cells kill Id+ B-lymphoma cells. The results show that Id-specific CD4+ T cells, in the absence of antibodies home to subcutaneous Id+ B lymphoma, become activated, induce inflammation, and prevent tumor development.
Introduction
Idiotypes (Id's) are antigenic determinants localized to the variable (V) regions of immunoglobulin (Ig). Because Ig V regions are extremely diverse, the monoclonal Ig produced by a malignant B-cell clone express unique Id's that may function as highly tumor-specific antigens.
Immunization with monoclonal Ig can induce Id-specific antibodies1 and Id-specific T cells,2 and early animal experiments suggested that both these humoral and cellular arms of adaptive immunity may have an antilymphoma effect.3-10 Id-vaccination has moved into phase 1 and 2 trials of follicular B-lymphoma patients with encouraging results.11-13
It has, however, been difficult to establish the relative contribution of antibodies and T cells to Id-specific protection against B lymphoma. Supporting a role of antibodies, passive treatment of patients with Id-specific antibodies (Abs) may have an anti–B-lymphoma effect,14 possibly due to signaling through lymphoma B-cell receptors (BCRs) and induction of apoptosis.15 Recently, it was shown that Id-specific antibodies alone protected mice against the mouse 38C13 B-cell lymphoma in the absence of T cells.16 The converse (ie, effect of Id-specific T cells in the absence of antibodies) has been shown in a myeloma model,17 but there is no conclusive evidence for B lymphomas. This is not a trivial distinction as B-cell lymphomas generally express major histocompatibility complex (MHC) class II molecules and surface Ig, both of which myeloma cells lack. Thus, B lymphomas could, in contrast to myeloma cells, be direct targets for both Id-specific CD4+ T cells and antibodies.
Id-specific T cells can be of the CD4+ type, recognizing Id-peptide on MHC class II molecules,18,19 or the CD8+ type, recognizing Id-peptide on class I molecules.20,21 However, the role of either of these 2 types of Id-specific T cells in fighting B lymphoma in vivo is poorly understood.
In a BALB/c mouse model, Id-specific CD4+ T cells have been shown to recognize a CDR3 peptide spanning residues 91 to 101 of a λ2 Ig light (L) chain. The particular λ2 chain is called λ2315 because it is derived from the MOPC315 plasmacytoma. The λ2315 contains unique amino acids in positions 94, 95, and 96, due to somatic mutations in the V-gene segment of MOPC315. T-cell recognition of the λ2315 Id heavily depends on these 3 CDR3 amino acids, and recognition is MHC class II (I-Ed) restricted.18,22,23 In order to establish a lymphoma model, the λ2315 gene was transfected into the BALB/c A20 B-lymphoma cells. Transfected cells (F9) expressed the λ2315 L chain together with the endogenous heavy (H) chain in the cell surface BCR. Moreover, F9 cells spontaneously processed λ2315 and presented the Id-peptide on their class II molecules to cloned Id-specific CD4+ cells.24,25 This finding was corroborated by elution of V-region peptides from B-lymphoma class II molecules.26 Thus, B-lymphoma cells constitutively express both serologically defined Id's (ie, surface Ig) and T-cell–defined Id's (ie, Id-peptides on class II molecules), each of which could be targets for Id-specific antibodies and CD4+ T cells, respectively.
Id+ F9 B-lymphoma cells were rejected by cloned Id-specific T cells in vivo in a Winn-type assay.6 Moreover, Id-specific T-cell–receptor (TCR) transgenic mice were protected against challenges with the Id+ B lymphoma.27 However, a role of antibodies or CD8+ T cells was not ruled out in these experiments. Moreover, as rearrangements of endogenous TCRα chains are common in TCR-transgenic mice, a considerable TCR repertoire for other antigens than Id's could have contributed to this protection.
To overcome these previous limitations, we have now used recombination-deficient Id-specific TCR transgenic mice that have CD4+ T cells that express only the transgenic Id-specific TCR but lack B cells and antibodies. We show that CD4+ Id-specific cells protect against challenges with B-lymphoma cells, moreover, we demonstrate that Id-specific CD4+ T cells have a therapeutic effect since they can prevent tumor occurrence in mice previously injected with lymphoma cells.
Materials and methods
Mice
Mice transgenic for an Id-specific TCR on a BALB/c background have been described,28 as have Id-specific TCR-transgenic mice homozygous for the scid mutation on a BALB/c IgH-congenic C.B-17 background.17 Recombinase activating 2 gene deficient (Rag2-/-) BALB/c mice were from Taconic (Bomholt, Ry, Denmark). Rag2 expression is necessary for recombination of endogenous TCR and Ig genes, therefore, Rag2-/- mice have a nonleaky severe combined immunodeficiency (SCID) phenotype. Hemizygous TCR-transgenic Rag2-/- were established in our animal facility by backcrossing homozygous TCR-transgenic BALB/c mice 2 times against Rag2-/- BALB/c. TCR-transgenic Rag2-/- mice expressed the transgenic αβTCR but not other TCR or Ig genes. TCR-transgenic SCID (or TCR-transgenic Rag2-/-) mice hemizygous for the TCR transgenes were bred by (SCID [or Rag2-/-] × TCR-transgenic SCID [or TCR-transgenic Rag2-/-]) crosses. Offspring (50% transgenic, 50% non-transgenic) were typed by staining of blood CD4+ lymphocytes using the clonotype-specific monoclonal antibody (mAb) GB113. Mice were of specific pathogen free (SPF) standard and bred behind a barrier. Experiments were performed according to governmental and institutional guidelines.
Adoptive transfer
CD4+ T cells and CD4- cells were purified from lymph nodes (LNs) of TCR-transgenic mice, or spleen and LNs of TCR-transgenic SCID mice, and transferred to SCID recipients. Purification of CD4+ T cells (> 95% pure) was done by positive selection using magnetic beads conjugated with an IgM anti-CD4 mAb and released from beads by use of an anti-Fab reagent (Dynabeads and Detachabead; Dynal, Oslo, Norway).17 The negatively selected cells from this procedure were used as a source of CD4- T cells.17 In protection-type experiments, the indicated number of purified cells was transferred intravenously to SCID recipients that were challenged 3 days later with the indicated number of tumor cells. In immunotherapy type experiments, SCID mice were first injected subcutaneously with tumor cells and then intravenously with purified CD4+ T cells at the indicated time points.
Tumor challenges, survival curves, and statistics
Adult (6-16 weeks old) mice were injected subcutaneously between the shoulder blades with the indicated number of tumor cells and monitored weekly for tumor development. Tumor sizes (diameter) were estimated by palpation and use of a caliper. A tumor of 3 mm in diameter was scored as a tumor take. Mice were killed when tumor size reached 20 mm. Tumor avoidance or survival curves and statistical comparison (log-rank test) were calculated by use of Graph Pad Prism 3.0 software (San Diego, CA).
B-lymphoma cell lines and ex vivo single-cell suspensions from tumors
F9A.15.3.19 (abbreviated F9) is a cell line derived from BALB/c (H-2d) A20 B-lymphoma cells transfected with the λ2315 gene.24 F9 cells express the λ2315 L chain together with the endogenous γ2a in the cell membrane, easily detectable by surface iodination and immunoprecipitation and by flow cytometry. It secretes small amounts (150-500 ng/mL in dense cultures) of λ2315 into supernatant (SN). It expresses Fas (CD95) as detected by the JO-2 mAb in flow cytometry and is readily killed by FasLigand (K.U.L. and B.B., unpublished data, June 2000). The F55B17.2 (F55) cell line represents A20 cells transfected with the empty vector alone (pSV2neo).25 To obtain single-cell suspensions from ex vivo subcutaneous lymphomas, tumors were minced and treated with collagenase and DNase as described.29
Id-specific CD4+ T cells
Naive Id-specific T cells were isolated from LNs and spleen of TCR-transgenic or TCR-transgenic SCID mice by magnetic beads as described.17 To obtain T cell helper 1 (Th1) or Th2 cells, LN cells from TCR-transgenic or TCR-transgenic SCID mice were set up in culture (5 × 105/mL) together with BALB/c spleen cells (2.5 × 106/mL), synthetic 91-107 Id-(λ2315) peptide (1 μg/mL), recombinant mouse interleukin 12 (IL-12) (2 ng/mL; R&D Systems, Minneapolis, MN), and anti–IL-4 (11B11 mAb, 1 μg/mL) for Th1 induction; or IL-4 (20 U/mL) for Th2 induction. T cells were expanded after several days and used after 1 or 2 10-day cycles. The Id (λ2315)–specific 7A10B2 Th1 clone has been described.18 Complete tissue culture medium was RPMI 1640 with 10% fetal calf serum (FCS; Gibco, Carlsbad, CA) and supplements.30
Antibodies and flow cytometry
The following commercially available mAbs were used, conjugated with either fluorescein isothiocyanate (FITC), phycoerythrin (PE), allophycocyanin, or biotin: CD4 (RM4-5), CD69 (H1.2F3), and CD45R (RA3-6B2) (PharMingen, San Diego, CA). The various fluorochrome-conjugated, isotype-matched control mAbs were from PharMingen. Transgenic-TCR–clonotype-specific GB113 mAbs and Vβ8-specific F23.1 mAbs were affinity-purified and biotin and FITC conjugated by standard procedures. Biotinylated Abs were detected with streptavidin (SA)–CyChrome or SA–peridin-chlorophyll protein (SA-PerCP; PharMingen). Combinations of the above-listed mAbs were used for double, triple, or quadruple stainings of cells essentially as described earlier.28 Samples were acquired on a FACScalibur (Becton Dickinson, Mountain View, CA) and analyzed by the WinMDI program (http://facs.scripps.edu/software.html).
Immunohistochemistry and microscopy
Stainings of tumor sections were performed by embedding organs in optimum cutting temperature (OCT) compound (Tissue-Tek; Miles, Elkhart, IN), and 5-μm frozen sections were mounted on l-polylysine–coated glass slides and air-dried overnight. The following biotinylated mAbs were added and detected by Streptavidin–Cy 3 (Amersham, Pittsburgh, PA): GB113,31 anti–Ly-6G (Gr-1, RB6-8C5; PharMingen), anti–Thy 1.2, (T24), anti-CD31 (MEC 13.3; PharMingen), and anti-CD11b (TIB128; American Type Culture Collection [ATTC], Manassas, VA). F9 and F55 tumor cells were stained with highly cross-absorbed Alexa Fluor 488 goat anti–mouse IgG, (Molecular Probes, Eugene, OR). Nuclei were stained with DAPI (4,6-diamino-2-phenylindoldi-hydrochloride; Molecular Probes). Pictures were made with a charge-coupled device camera (Hamamatsu, Ichinocho, Japan).
ELISA
The λ2315 Ig was measured in a sandwich enzyme-linked immunosorbent assay (ELISA) using 9A8 mAb (anti-Vλ1/Vλ2) as capture antibody and biotinylated 2B6 mAb (anti-Cλ2/Cλ3) as detection antibody. This assay detects free λ2315 as well as λ2315 associated with an H chain. Purified 36.1 mAb (IgG2a λ2315) produced by a hybridoma from a λ2315 transgenic mouse served as a standard (B.B., unpublished data, October 1999).
Apoptosis assays
Apoptosis was measured by a modified JAM assay.32 Tumor cells were cultured in the presence of [methyl-3H] thymidine (3H-TdR) for 24 hours. Assays were set up with radioactively labeled target cells (104/well) and titrated amounts of cytotoxic T cells. After overnight culture in 96-well plates, cells were harvested and counted on a TopCount NXT Microplate scintillation and luminescence counter (Packard, Boston, MA). Percent thymidine release was calculated as: (count control target cells) - (count apoptotic target cells) = (count control target cells) × 100. Apoptosis was also measured by annexin-V staining using Annexin-V–FLUOS Staining Kit (Roche Molecular Biochemicals, Basel, Switzerland).
Results
Id-specific TCR-transgenic SCID mice are protected against challenges with Id+ B-lymphoma cells
The λ2315-transfected Id+ A20 cells (F9) express λ2315 in their BCRs and secrete small amounts of λ2315 Ig. F9 cells spontaneously process λ2315 and present Id (λ2315) peptide bound to I-Ed class II molecules on their cell surface.24,25 In initial experiments, we observed that BALB/c mice were partially resistant to F9 while C.B-17 SCID mice readily developed tumors. This difference could be due to the normal B- and T-cell repertoire of BALB/c mice conferring a slight resistance against Id+ F9. One possibility is that amino acids (aa's) 106 to 114 of the endogenous H chain of A20, reported to be presented on H-2Kd to CD8+ T cells,33 could induce Id-specific CD8+ T cells in BALB/c mice. Alternatively, expression by F9 cells of the neomycin resistance gene encoding for a foreign phosphotransferase could induce a protective immune response in BALB/c mice.
For the reasons above, and in particular to avoid antibodies, we did most of the current experiments in immunodeficient, Id-specific TCR-transgenic SCID mice. Due to their recombination deficiency, TCR-transgenic SCID mice express very few endogenous Ig or TCR genes and contain hardly any B cells or antibodies or CD8+ T cells, whereas Id-specific CD4+ T cells dominate.17 Id+ F9 B-lymphoma cells were injected in titrated amounts subcutaneously between the shoulder blades of mice (Figure 1A). TCR-transgenic SCID mice were significantly protected against F9, compared with ordinary SCID mice, the extent of protection being dependent on the number of F9 cells injected. The protection was Id specific because TCR-transgenic SCID mice readily succumbed to A20 cells transfected with the empty pSV2neo vector (Id- F55 cells) (Figure 1A).
Id-specific TCR-transgenic SCID mice are protected against subcutaneous challenges with Id+ B lymphomas. (A) TCR-transgenic SCID mice and SCID littermates were challenged with the indicated titrated amounts of either A20 cells transfected with the λ2315 Ig L chain gene (F9) or A20 cells transfected with the empty pSV2neo vector alone (F55). A palpable tumor of 3 mm was scored as a tumor take. The lowest number of injected F9 cells, 1.25 × 10,3 was not tumorigenic. (B) Id+ λ2315 Ig in sera of mice injected 12 days earlier with 1.25 × 106 cells. Results were statistically significant (P < .0001), Mann-Whitney test.
Id-specific TCR-transgenic SCID mice are protected against subcutaneous challenges with Id+ B lymphomas. (A) TCR-transgenic SCID mice and SCID littermates were challenged with the indicated titrated amounts of either A20 cells transfected with the λ2315 Ig L chain gene (F9) or A20 cells transfected with the empty pSV2neo vector alone (F55). A palpable tumor of 3 mm was scored as a tumor take. The lowest number of injected F9 cells, 1.25 × 10,3 was not tumorigenic. (B) Id+ λ2315 Ig in sera of mice injected 12 days earlier with 1.25 × 106 cells. Results were statistically significant (P < .0001), Mann-Whitney test.
It is well known that B-lymphoma cells, including F9,24,25 secrete minor amounts of Ig. Because the experiments were performed in antibody-deficient scid-/- mice, it was possible to measure tumor-derived λ2315 in serum in the absence of any masking by endogenously produced λ2. At day 12, SCID mice injected with 1.25 × 106 F9 cells had an average serum λ2315 of about 7500 ng/mL. By comparison, TCR-transgenic SCID mice had much less λ2315 Ig in their sera (average 100 ng/mL) and 6 of 14 mice had undetectable levels (Figure 1B). Thus, the resistance of TCR-transgenic SCID mice against a tumor challenge with F9 (Figure 1A) was paralleled by a significant reduction in amounts of Id+ Ig in serum (Figure 1B).
Id-specific TCR-transgenic mice on a nonleaky Rag2-/- background are protected against Id+ B lymphoma
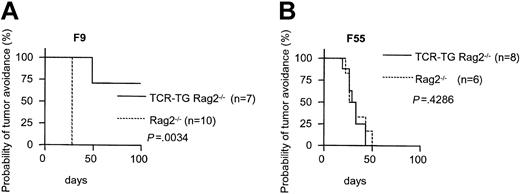
SCID mice are known to be leaky; in our TCR-transgenic SCID colony approximately 25% of adult mice had 10 to 400 μg/mL IgMκ in serum while the remainder were negative.17 We have also detected very low frequencies of endogenous TCR rearrangement and expression in these mice (L.A.N. and B.B., unpublished, December 1998). We therefore repeated the tumor challenge experiment, now injecting 1.25 × 105 cells, with Id-specific TCR-transgenic mice on a nonleaky Rag2-/- BALB/c background. Indeed, TCR-transgenic Rag2-/- mice were protected against F9 (Figure 2A) but not F55 (Figure 2B), while Rag2-/- succumbed to both. These experiments exclude the necessity of B cells or antibodies, or endogenous Ig or TCR rearrangements, to protection against Id+ B lymphoma.
Nonleaky Id-specific TCR-transgenic Rag2-/- mice are protected against subcutaneous challenges with Id+ B lymphomas. TCR-transgenic Rag2-/- mice and Rag2-/- littermates were challenged with either (A) Id+ F9 or (B) Id- F55 cells (1.25 × 105).
Nonleaky Id-specific TCR-transgenic Rag2-/- mice are protected against subcutaneous challenges with Id+ B lymphomas. TCR-transgenic Rag2-/- mice and Rag2-/- littermates were challenged with either (A) Id+ F9 or (B) Id- F55 cells (1.25 × 105).
Adoptively transferred Id-specific CD4+ T cells protect SCID mice against challenges with Id+ B-lymphoma cells
Although the TCR-transgenic SCID mice predominantly express CD4+ T cells, they have some CD4/CD8 double negative cells, and some very few CD8+ cells, that express the transgenic TCR.17 Albeit unlikely, these cells could have contributed to the tumor protection observed in TCR-transgenic SCID mice (Figure 1A). To test this possibility, we did a number of transfer experiments.
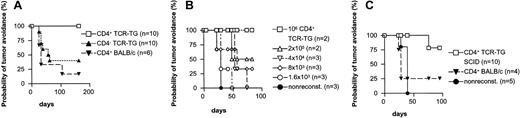
In the first set of experiments (Figure 3A-B), we used ordinary TCR-transgenic mice as donors for 2 reasons. First, their normal lymphocyte numbers facilitated purification of sufficient amounts of CD4+ T cells for transfer. Second, ordinary TCR-transgenic mice have relatively high numbers of CD8+ and CD4-8- T cells,17 which makes it easy to test any antilymphoma effect of these 2 populations. CD4+ T cells were highly purified (> 95%) and injected intravenously into SCID mice (2 × 106 cells per mouse). Another group of mice was injected with the leftover, negatively selected CD4- cells; this population consisted of a mixture of CD8+ and CD4-8- T cells, as well as B cells. A third group received purified CD4+ T cells from BALB/c mice. Three days later the reconstituted mice were challenged with F9 cells. As can be seen in Figure 3A, transgenic Id-specific CD4+ T cells conferred a significant protection against F9 compared with either transgenic CD4- cells or BALB/c CD4+ T cells.
Transfer of Id-specific CD4+ T cells confers tumor protection. (A) TCR-transgenic CD4+ cells, but not CD4- cells, protect. SCID mice were reconstituted with purified TCR-transgenic CD4+ or CD4- cells, or BALB/c CD4+ T cells, injected intravenously (2 × 106). Three days later, the mice were challenged with 1.25 × 105 Id+ F9 cells. TCR-transgenic CD4+ cells versus TCR-transgenic CD4- cells: P = .018. TCR-transgenic CD4+ cells versus BALB/c CD4+ cells: P = .0014. TCR transgenic CD4- versus BALB/c: P = 0.59. (B) Number of Id-specific CD4+ T cells required for transfer of protection. Groups of Rag2-/- mice were injected intravenously with the indicated titrated amounts of purified TCR-transgenic CD4+ T cells, or left nonreconstituted, and challenged 3 days later with 1.25 × 105 F9 cells. (C) Id-specific CD4+ T cells purified from recombination deficient TCR-transgenic SCID mice confer protection. Groups of SCID mice were reconstituted with 2.5 × 105 CD4+ cells purified from either TCR-transgenic SCID or BALB/c mice, or left nonreconstituted. Three days later, the mice were challenged with 1.25 × 105 F9 cells. TCR-transgenic SCID CD4+ cells versus BALB/c CD4+ cells: P = .014. BALB/c CD4+ versus nonreconstituted mice: P = .44.
Transfer of Id-specific CD4+ T cells confers tumor protection. (A) TCR-transgenic CD4+ cells, but not CD4- cells, protect. SCID mice were reconstituted with purified TCR-transgenic CD4+ or CD4- cells, or BALB/c CD4+ T cells, injected intravenously (2 × 106). Three days later, the mice were challenged with 1.25 × 105 Id+ F9 cells. TCR-transgenic CD4+ cells versus TCR-transgenic CD4- cells: P = .018. TCR-transgenic CD4+ cells versus BALB/c CD4+ cells: P = .0014. TCR transgenic CD4- versus BALB/c: P = 0.59. (B) Number of Id-specific CD4+ T cells required for transfer of protection. Groups of Rag2-/- mice were injected intravenously with the indicated titrated amounts of purified TCR-transgenic CD4+ T cells, or left nonreconstituted, and challenged 3 days later with 1.25 × 105 F9 cells. (C) Id-specific CD4+ T cells purified from recombination deficient TCR-transgenic SCID mice confer protection. Groups of SCID mice were reconstituted with 2.5 × 105 CD4+ cells purified from either TCR-transgenic SCID or BALB/c mice, or left nonreconstituted. Three days later, the mice were challenged with 1.25 × 105 F9 cells. TCR-transgenic SCID CD4+ cells versus BALB/c CD4+ cells: P = .014. BALB/c CD4+ versus nonreconstituted mice: P = .44.
To determine the number of T cells needed to protect against Id+ B-lymphoma cells, titrated amounts of TCR-transgenic CD4+ T cells in the range of 2 × 106 to 1.25 × 105 were transferred to SCID recipients prior to challenge with 1.25 × 105 F9 cells. None of the twelve mice developed tumors, indicating that transfer of more than 1.25 × 105 purified Id-specific CD4+ T cells suffice for complete protection (data not shown). In the next experiment TCR transgenic CD4+ T cells titrated over a wider range were transferred to Rag2-/- recipients (Figure 3B). All mice in the nonreconstituted control group acquired tumors within 28 days. Transfer of more than 8 × 103 cells retarded tumor development while partial to complete tumor avoidance required more than 2 × 105 cells. To exclude any influence of endogenous TCR genes, we repeated the experiment with recombination-deficient TCR-transgenic SCID mice. Purified CD4+ cells from such SPF-standard mice should be antigen inexperienced and thus naive. As can be seen from Figure 3C, 2.5 × 105 Id-specific CD4+ TCR-transgenic SCID cells conferred a significant protection. Hence, transfer of a few hundred thousand Id-specific CD4+ T cells prevent tumor occurrence while smaller amounts may retard development.
Immunotherapy with Id-specific CD4+ T cells
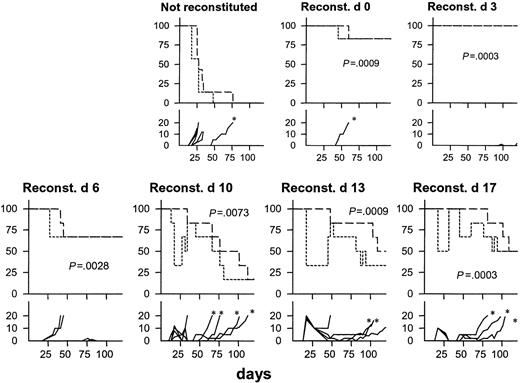
We next tested the curative potential of Id-specific TCR-transgenic CD4+ T cells in SCID mice previously injected with Id+ F9 cells. All nonreconstituted mice developed tumors, 6 of 7 within the first 35 days (Figure 4). By contrast, only 1 of 12 mice reconstituted with Id-specific CD4+ T cells on day 0 or day 3 developed a tumor within 120 days. Protection by transfer on day 6 was less impressive with 2 of 6 mice developing rapidly growing tumors within 30 days (Figure 4). When Id-specific CD4+ cell transfer was delayed until days 10, 13, or 17 after F9 injection, a striking phenomenon was observed: within a few days, most mice developed an acute swelling of up to 20 mm that is recorded as tumor occurrence in Figure 4 (bottom row). The swelling did not reflect tumor growth because it increased dramatically in size within a few days and then often receded to be unpalpable within some weeks. In a considerable number of long-term survivors, a nodule remnant could be palpated, possibly reflecting fibrosis. In 11 of 18 mice, late tumors (emerging after day 50) developed, marked by asterisks in Figure 4.
Curative potential of Id-specific CD4+ T cells. Groups of SCID mice (n = 6-7) were injected with 1.25 × 105 F9 cells at day 0 in all graphs. At the indicated time points subsequent to tumor cell injection, mice received 106 CD4+ T cells purified from TCR-transgenic mice. Percent tumor avoidance (short dashes; tumor ≥ 3 mm was defined as tumor take), % survival (long dashes), and tumor size in diameter (solid lines; mm) were recorded (split y-axis). * indicates late emergence of tumors (survival ≥ 60 days). P value in each graph represents comparison with nonreconstituted mice for survival.
Curative potential of Id-specific CD4+ T cells. Groups of SCID mice (n = 6-7) were injected with 1.25 × 105 F9 cells at day 0 in all graphs. At the indicated time points subsequent to tumor cell injection, mice received 106 CD4+ T cells purified from TCR-transgenic mice. Percent tumor avoidance (short dashes; tumor ≥ 3 mm was defined as tumor take), % survival (long dashes), and tumor size in diameter (solid lines; mm) were recorded (split y-axis). * indicates late emergence of tumors (survival ≥ 60 days). P value in each graph represents comparison with nonreconstituted mice for survival.
Id-specific CD4+ T cells accumulate and are activated in Id+ B-lymphoma tumors
The results in Figure 4 suggest that Id-specific CD4+ T cells find their way to the tumor and become activated with inflammation and tumor cell eradication as a result. To test this, SCID mice were first injected with F9 or F55 and were 13 days later injected with 106 Id-specific CD4+ T cells. At this time point, no tumors could be palpated. Five days later, 2 small F55 tumors (each 2 mm diameter, 4.2 mm3 volume, average 13.3 × 106 cells) and 2 larger F9 tumors (8 and 10 mm diameter, 270 and 520 mm3 volume, 28 × 106 and 34 × 106 cells) were observed. The F9 tumors were apparently inflammatory and edematous because the numbers of cells recovered were much less than that expected from their calculated volumes (F9/F55 cell ratio approximately 2:1; volume ratio approximately 100:1). In single-cell suspensions derived from the tumors, Id-specific CD4+ T cells were more frequent, more activated (CD69+), and had a more blastlike appearance in F9 compared with F55 tumors (Figure 5).
Id-specific CD4+ T cells accumulate and become activated in Id+ B-lymphoma tumors. SCID mice were injected with 1.25 × 105 F9 or F55 cells and 13 days later received 106 Id-specific CD4+ cells purified from TCR-transgenic mice. At this time point, none of the mice had tumors. Five days later, single-cell suspensions from 2 F55 tumors (∼2 mm) and 2 F9 tumors (8-10 mm) were analyzed by flow cytometry. (A) Number of Id-specific (GB113+) CD4+ T cells per 106 F9 and F55 tumor cells. (B) Number of activated (CD69+ GB113+) CD4+ cells per 106 F9 and F55 tumor cells. (C) Gated GB113+ CD4+ T cells in F9 and F55 tumor cell suspensions were analyzed for CD69 expression and size (forward scatter [FSC]).
Id-specific CD4+ T cells accumulate and become activated in Id+ B-lymphoma tumors. SCID mice were injected with 1.25 × 105 F9 or F55 cells and 13 days later received 106 Id-specific CD4+ cells purified from TCR-transgenic mice. At this time point, none of the mice had tumors. Five days later, single-cell suspensions from 2 F55 tumors (∼2 mm) and 2 F9 tumors (8-10 mm) were analyzed by flow cytometry. (A) Number of Id-specific (GB113+) CD4+ T cells per 106 F9 and F55 tumor cells. (B) Number of activated (CD69+ GB113+) CD4+ cells per 106 F9 and F55 tumor cells. (C) Gated GB113+ CD4+ T cells in F9 and F55 tumor cell suspensions were analyzed for CD69 expression and size (forward scatter [FSC]).
Infiltration of Id-specific T cells and neutrophils in inflamed Id+ B-lymphoma tumors
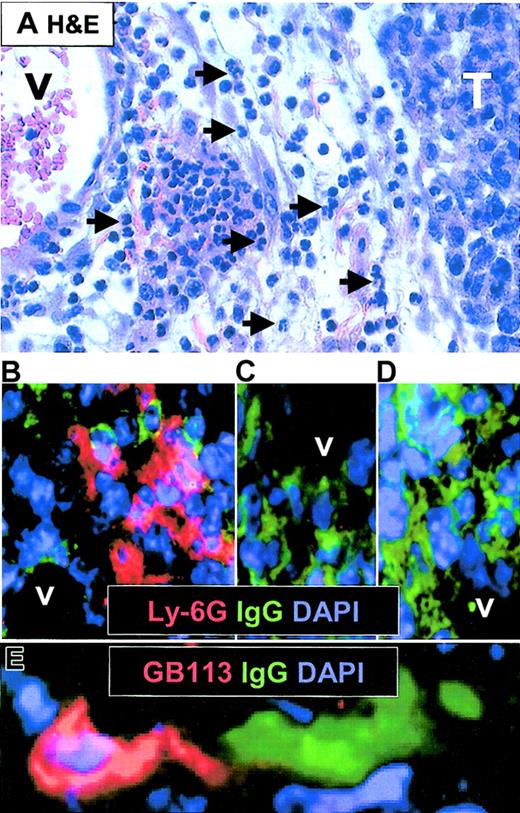
SCID mice were injected subcutaneously with F9 and F55 cells. When tumors were barely palpable (approximately 3 mm), mice did or did not receive Id-specific CD4+ T cells, and tumors were analyzed by immunohistochemistry 4 to 5 days later. For each combination of Id+ or Id- B lymphoma with or without Id-specific T cells, 2 to 4 animals were analyzed with similar results as that shown in Figure 6. Acutely inflamed Id+ F9 tumors from SCID mice that had received Id-specific CD4+ T cells were edematous and had perivascular leukocyte infiltrations dominated by polymorph nuclear (PMN) granulocytes (Figure 6A hematoxylin and eosin [H&E] stain). Immunohistochemistry confirmed the presence of Ly-6G+ PMN granulocytes in proximity to CD31+ vessels (Figure 6B and data not shown). Moreover, the tumor-infiltrating neutrophils were readily found in close contact to surface Ig (sIg)G+ F9 tumor cells (Figure 6B). No such infiltrates were found in Id+ F9 tumors in mice that had not received T cells (Figure 6C) or in Id- F55 tumors in recipients of T cells (Figure 6D). The occurrence of neutrophils as a sign of inflammation was therefore dependent on both injection of Id-specific T cells and Id expression of tumor cells. Further, we analyzed if Id-specific CD4+ T cells could be found in F9 tumors in T-cell–injected mice. Indeed, some few TCR transgenic GB113+ T cells were found infiltrating the F9 tumors, and in some cases they were found to be in direct contact with F9 cells (Figure 6E and data not shown).
Histologic analysis of inflamed F9 tumors after injection of Id-specific T cells. SCID mice with barely palpable subcutaneous F9 or F55 tumors were injected intravenously with purified Id-specific CD4+ T cells (1 × 106) and analyzed 4 to 5 days later. (A) Hematoxylin and eosin–stained section of paraffin-embedded acutely inflamed F9 (15 mm diameter) tumor. V indicates vessel; and T, tumor. Arrows mark some of the neutrophils infiltrating the perivascular edematous connective tissue. (B-C) Immunohistochemistry on cryosections of tumor tissue. Ly-6G (Gr-1) is shown in red, IgG in green, and nuclei (DAPI) in blue. Lymphoma cells express surface IgG2a and are therefore stained green. (B) F9 tumor in mice injected with T cells. (C) F9 tumor in mice not injected with T cells. (D) F55 tumor in mice injected with T cells. (E) Same tumor as in panel B, but stained for detection of Id-specific T cells (GB113, clonotype-specific, red) and IgG+ F9 cells. Original magnification, × 60.
Histologic analysis of inflamed F9 tumors after injection of Id-specific T cells. SCID mice with barely palpable subcutaneous F9 or F55 tumors were injected intravenously with purified Id-specific CD4+ T cells (1 × 106) and analyzed 4 to 5 days later. (A) Hematoxylin and eosin–stained section of paraffin-embedded acutely inflamed F9 (15 mm diameter) tumor. V indicates vessel; and T, tumor. Arrows mark some of the neutrophils infiltrating the perivascular edematous connective tissue. (B-C) Immunohistochemistry on cryosections of tumor tissue. Ly-6G (Gr-1) is shown in red, IgG in green, and nuclei (DAPI) in blue. Lymphoma cells express surface IgG2a and are therefore stained green. (B) F9 tumor in mice injected with T cells. (C) F9 tumor in mice not injected with T cells. (D) F55 tumor in mice injected with T cells. (E) Same tumor as in panel B, but stained for detection of Id-specific T cells (GB113, clonotype-specific, red) and IgG+ F9 cells. Original magnification, × 60.
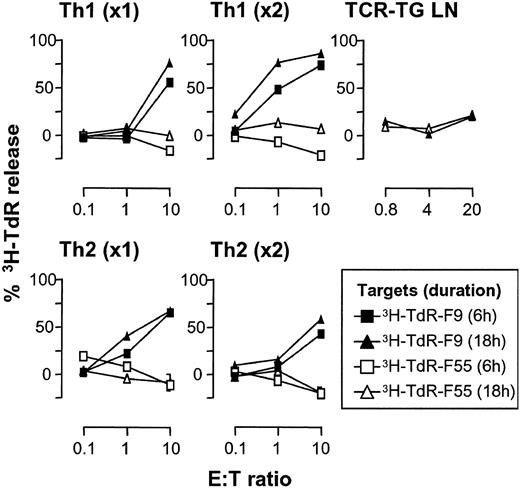
Memory but not naive Id-specific CD4+ T cells kill Id+ B-lymphoma cells
The experiments described in Figures 1, 2, 3, 4, 5, 6 suggest that Id-specific CD4+ T cells kill Id+ B-lymphoma cells in vivo. We proceeded to test this in vitro. F9 (Id+) and F55 (Id-) were tested in a 3H-TdR release assay (JAM assay) for susceptibility to killing by Id-specific CD4+ T cells (Figure 7). This assay measures release of 3H-TdR–labeled DNA fragments from cells, due to apoptosis. Previously unstimulated naive T cells from ordinary TCR-transgenic mice were unable to kill lymphoma cells (Figure 7), as were T cells from TCR-transgenic SCID mice or even purified CD4+ T cells from these sources (not shown). By contrast, previously stimulated, polarized T cells killed F9 but not F55 cells. Cytotoxicity of Th1-polarized cells increased with cycles of stimulation while Th2 cells reduced their cytotoxicity with repetitive stimulation. Similar results were obtained in another assay for apoptosis, annexin-V staining. An Id-specific Th1 clone, 7A10B2, induced apoptosis of F9 (Figure 8 arrow) but not F55 cells, whereas unstimulated purified Id-specific CD4+ cells purified ex vivo were unable to kill either (Figure 8).
Memory but not naive Id-specific T cells kill Id+ B-lymphoma cells by apoptosis. Id+ F9 and Id- F55 cells (104) were tested in a 6- or 18-hour 3H-Td release assay (JAM assay) for susceptibility to killing by unstimulated naive or Th1- or Th2-polarized Id-specific TCR-transgenic SCID splenocytes that had been stimulated in vitro for one (× 1) or 2 (× 2) 10-day cycles. TCR-transgenic lymph node cells served as a source of unstimulated naive CD4+ T cells.
Memory but not naive Id-specific T cells kill Id+ B-lymphoma cells by apoptosis. Id+ F9 and Id- F55 cells (104) were tested in a 6- or 18-hour 3H-Td release assay (JAM assay) for susceptibility to killing by unstimulated naive or Th1- or Th2-polarized Id-specific TCR-transgenic SCID splenocytes that had been stimulated in vitro for one (× 1) or 2 (× 2) 10-day cycles. TCR-transgenic lymph node cells served as a source of unstimulated naive CD4+ T cells.
Id+ B-lymphoma cells become annexin V+ when exposed to cloned Id-specific Th1 cells. Id+ F9 and Id- F55 B-lymphoma cells were incubated for 4 hours together with either an Id-specific Th1 cell clone (7A10B2), or magnetic bead–purified naive CD4+ T cells from a TCR-transgenic mouse (effector-target ratio [E/T] 2:1), followed by staining. Annexin-V staining of gated B220+ propidium iodide–negative lymphoma cells is shown as shaded histograms, open histograms represent annexin-V staining of lymphoma cells in the absence of T cells.
Id+ B-lymphoma cells become annexin V+ when exposed to cloned Id-specific Th1 cells. Id+ F9 and Id- F55 B-lymphoma cells were incubated for 4 hours together with either an Id-specific Th1 cell clone (7A10B2), or magnetic bead–purified naive CD4+ T cells from a TCR-transgenic mouse (effector-target ratio [E/T] 2:1), followed by staining. Annexin-V staining of gated B220+ propidium iodide–negative lymphoma cells is shown as shaded histograms, open histograms represent annexin-V staining of lymphoma cells in the absence of T cells.
Discussion
This study demonstrates that Id-specific CD4+ T cells protect against B-lymphoma cells in the complete absence of other T cells or antibodies. To obtain these results we have used Id-specific TCR-transgenic SCID mice and adoptive transfer of purified Id-specific CD4+ T cells into SCID or Rag2-/- mice. A similar strategy was recently taken with success in melanoma patients who first received nonmyeloablative conditioning treatment and then transfer of melanoma-specific T cells that expanded in the lympho-depleted patient.34 Our results let us conclude that transferal of a few hundred thousand Id-specific CD4+ T cells before tumor challenge confer protection against B lymphoma in the absence of other cell types of adaptive immunity. On a weight basis, this compares with 2 × 108 Id-specific CD4+ T cells needed for a human patient of about 60 kg. Taken together with other experiments in less defined but more physiologic systems,8,9,27,30,35-37 the case for an anti–B-lymphoma effect of CD4+ cells alone seems strong. Accordingly, Id-vaccination protocols should aim at also inducing Id-specific CD4+ T cells. There is increasing evidence for a role of CD4+ T cells also in other types of cancer (reviewed in Pardoll and Topalian38 ).
Importantly, Id-specific CD4+ T cells could be injected intravenously as late as 17 days after B-lymphoma cell injection and even so, prevent tumor growth in a considerable fraction of mice. This is remarkable since in the absence of rescuing T cells, most mice had to be killed within 30 days due to large tumor burdens. Clearly, Id-specific CD4+ T cells home to the subcutaneous B-lymphoma injection site, become activated, and induce an acute inflammation with infiltration of neutrophils, resulting in a burst in size from unpalpable to 5 to 20 mm in diameter within 4 to 6 days. The acute inflammation subsided over a few weeks. In a fraction (∼ 30%) of these mice treated with Id-specific CD4+ T cells 10 to 17 days after tumor cell injection, delayed cancer growth occurred with time. We are currently investigating if these escapee B-lymphoma cells have lost production of the λ2315 L chain, or are resistant to cytotoxicity mediated by CD4+ T cells, which would explain why they escape surveillance by Id-specific CD4+ T cells. Importantly, the curative potential of Id-specific CD4+ T cells could be of use in the clinic. For example, after Id-vaccination of patients with minimal residual disease, patient's Id-specific CD4+ T cells could be expanded in vitro and reinfused in high numbers. In fact, it has recently been demonstrated that transfer of 2 × 1010 to 13 × 1010 melanoma-specific T cells to lympho-depleted melanoma patients resulted in expansion of transferred T cells and objective tumor regression in about half of the patients.34 Probably, lympho-depleted patients and SCID mice are comparable, in the sense that both have empty lymphoid compartments that are readily populated by transferred lymphocytes. The number of Id-specific T cells used for obtaining therapeutic results in tumor-bearing SCID mice was about a tenth of the number of melanoma-specific T cells transferred to the melanoma patients on a weight basis.
A concern for application in the clinic is that in vitro expansion of Id-specific CD4+ T cells prior to infusion might create memory phenotype T cells that less efficiently home to sites of B-lymphoma growth. On the other hand, in the melanoma study,34 tumor-infiltrating mixtures of CD4+ and CD8+ lymphocytes were expanded in vitro with high amounts of IL-2 for 4 to 6 weeks. Nevertheless, such in vitro–expanded and –injected tumor infiltrating lymphocytes (TIL) could readily be identified in cutaneous and subcutaneous tumor nodules. In this context, it is relevant to point out that memory T cells may be divided into central memory (CCR7+) and effector memory T cells (CCR7-) based on their expression of CCR7, the receptor for the secondary lymphoid tissue chemokine (SLC).39 While the former recirculates between lymphoid organs by diapedesis through high endothelial venules, the latter may traverse (inflamed) flat endothelium in nonlymphoid sites. Thus, a mixture of central and effector memory cells might home to most sites where lymphomas usually grow.
How do Id-specific CD4+ T cells kill the B-lymphoma cells? They could do so directly or indirectly. As concerns direct killing, immunohistochemistry analysis revealed proximity of Id-specific T cells and Id+ tumor cells, but whether this reflects direct killing of B-lymphoma cells by T cells in vivo in the tumor remains to be demonstrated. In vitro, more conclusive data suggest that naive T cells are incapable of inducing tumor cell apoptosis of B-lymphoma cells but that previously stimulated T cells can do so. Surprisingly, not only Th1 cells were effective killers but so were Th2 cells, although the latter reduced their cytotoxicity with repetitious restimulation. Possibly, the cytotoxicity of Th2-polarized cells could be due to contaminating Th1 cells that gradually are lost on repetitive stimulation under Th2 conditions. Alternatively, Id-specific Th2 cells may be cytotoxic to B-lymphoma cells.6 In any case, the in vitro data suggest that in vivo, naive Id-specific CD4+ T cells require stimulation in order to attain cytotoxicity toward B-lymphoma cells. At least 2 possibilities exist for initial stimulation of naive Id-specific CD4+ T cells. First, Id+ B-lymphoma cells could directly stimulate Id-specific CD4+ T cells. Second, Id-primed dendritic cells at the tumor site and in draining lymph nodes could have this function, as described for a subcutaneous mouse plasmacytoma.29,40 We find the latter possibility most likely, because Id-specific TCR-transgenic mice are not protected against lymphoma cells and myeloma cells that do not secrete Id+ Ig, but retain it intracellularly. In fact, purified dendritic cells (DCs) from Id+ F9 subcutaneous tumors are far superior at stimulating Id-specific T cells than Id+ F9 lymphoma cells themselves (K.U.L. and B.B., unpublished data, May 2000).
The molecular mechanism by which Id-specific effector memory CD4+ T cells could directly kill B-lymphoma cells remains to be elucidated. One possibility is that FasLigand expressed on activated CD4+ T cells could bind Fas on B-lymphoma cells and induce apoptosis. Indeed, F9 cells express Fas, and we have obtained evidence that this is an important mechanism in vitro but less so in vivo (K.U.L. and B.B., unpublished data, November 2000).
As concerns indirect killing of B-lymphoma cells, it has been described that interferon (IFN)–γ produced by CD4+ T cells could inhibit tumor growth by destroying tumor vasculature.41 Consistent with this possibility, we have observed that activated Id-specific T cells in MOPC315 myeloma tumors produce IFNγ (A.C., B.B., unpublished data, January 2002). It has also been described that activated CD4+ T cells can recruit nonadaptive arms of immunity such as cytotoxic macrophages and eosinophils.42 Herein we describe that Id-specific CD4+ T cells induce inflammation and infiltration of neutrophils in Id+ B lymphomas. It might be that such an inflammatory reaction causes tumor cell death and explains the observed cure of B lymphoma. In summary, we find it likely that Id-specific T cells kill Id+ B-lymphoma cells by several mechanisms, some of them being direct and others indirect.
CD4+ cells in TCR-transgenic SCID mice have a naive phenotype that should make them extravasate through high endothelial venules (HEVs) and thus presumably recirculate between LNs. This raises a problem: how do naive T cells get in contact with B-lymphoma cells that are injected subcutaneously? One explanation could be that the B-lymphoma cells metastasize to draining LNs and activate naive T cells. An alternative explanation is that dendritic cells in the tumor pick up Id's from the locally secreted monoclonal Ig or from necrotic or apoptotic lymphoma cells, and that these Id-primed dendritic cells travel with afferent lymphatics to draining LNs and stimulate Id-specific naive T cells. Relevant to this, Id-primed dendritic cells have recently been demonstrated in subcutaneous plasmacytoma,29 and we also have data suggesting their existence in B-cell lymphoma (K.U.L., B.B., unpublished data, May 2000). In either case, stimulated Id-specific CD4+ LN T cells could become effector memory cells with a changed recirculation pattern43 so that they now might traverse flat endothelium of the subcutaneous tumor and kill the lymphoma cells.
The finding that antibodies and CD8+ cells are dispensable for protection against subcutaneous B-cell lymphoma in this model does in no way exclude an importance of CD8+ and B cells. In fact, in the 38C13 B-lymphoma model, anti-Id antibodies alone were sufficient for protection.16 Most likely, the different arms of adaptive immunity may contribute to Id-specific protection against B-cell lymphomas and in many instances work in synergy. However, the relative importance of Id-specific CD4+ T cells, CD8+ T cells, and antibodies (B cells) could differ for each individual tumor, depending on whether the BCR's V-region sequences fit MHC class I or class II molecules of that individual and on the susceptibility of the tumor cells to various killing mechanisms employed by T-cell subsets and antibodies.
Prepublished online as Blood First Edition Paper, March 20, 2003; DOI 10.1182/blood-2002-11-3381.
Supported by grants from The Norwegian Cancer Society and the Research Council of Norway.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
Suzanne Garman-Vik expertly prepared the manuscript.
K. U. Lundin and A. Corthay are fellows of the Norwegian Cancer Society.


![Figure 5. Id-specific CD4+ T cells accumulate and become activated in Id+ B-lymphoma tumors. SCID mice were injected with 1.25 × 105 F9 or F55 cells and 13 days later received 106 Id-specific CD4+ cells purified from TCR-transgenic mice. At this time point, none of the mice had tumors. Five days later, single-cell suspensions from 2 F55 tumors (∼2 mm) and 2 F9 tumors (8-10 mm) were analyzed by flow cytometry. (A) Number of Id-specific (GB113+) CD4+ T cells per 106 F9 and F55 tumor cells. (B) Number of activated (CD69+ GB113+) CD4+ cells per 106 F9 and F55 tumor cells. (C) Gated GB113+ CD4+ T cells in F9 and F55 tumor cell suspensions were analyzed for CD69 expression and size (forward scatter [FSC]).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/102/2/10.1182_blood-2002-11-3381/6/m_h81434629005.jpeg?Expires=1769104782&Signature=mlV-XhtTSAJ~uTdvpLRp25b4UmO4HQdOmVzuHnmkjz9Rv8s6EfkhhpgIok3SFAAmurAhkrhBf~wqcXPadMbC15AnOi6M7MAPzXkmwCVy2gCcxlZzKkP9XHe5XsNsRVeI6WlvZ1m1f0lW9CBxGLjmUts3MdnLyhbmXnTzgj79XDGMjjjhLQAbccQH84FDr3b8mWgNrjBogheoq36wACy2llM6EsP9QvO~93Tabj9yAyNA-f8rCQ-TL2Fz1tYfBDh1nJels3mzuhVwx09tTChqbmKrNZ2tSgSfCZfKY-cfURuzzgBFzMfLqcg6jc4dp6UDgZUqemQKoO99b3TuFE3ORQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 8. Id+ B-lymphoma cells become annexin V+ when exposed to cloned Id-specific Th1 cells. Id+ F9 and Id- F55 B-lymphoma cells were incubated for 4 hours together with either an Id-specific Th1 cell clone (7A10B2), or magnetic bead–purified naive CD4+ T cells from a TCR-transgenic mouse (effector-target ratio [E/T] 2:1), followed by staining. Annexin-V staining of gated B220+ propidium iodide–negative lymphoma cells is shown as shaded histograms, open histograms represent annexin-V staining of lymphoma cells in the absence of T cells.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/102/2/10.1182_blood-2002-11-3381/6/m_h81434629008.jpeg?Expires=1769104782&Signature=NSqt6Z0gNyP4ynxjye6ZtJaGAAKl0~zLTk5b3QuMPt7~IoH-D0JiOcV0BmixUVrwvGzLDkY~WpNkRRUA1OW~GF~DDgcCmsLqGWpAgGqEDSbtNgNvQn~1B5YXU7NhNtZiHXRNuufSB5RzAo1jQh42w22Mezs88CplK35OzuXJasgR86YKZ~aexfEGLCLK1dRrDGaIJUXs3sdj-xGxPCpCMqBHKDJwqrqcTbNy6Q2vrby~8Yrj19ulavTQAHq-Yavc5-abQQAuLbZLKnN8eiiR-WUnVodfpHPyGc~qR~zBm8k2ziZllSe4ksD3qmboEM1Nyrw0LIgt1rEsneTU1tQWpA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal