Thrombin signaling in endothelial cells provides an important link between coagulation and inflammation. We report here that thrombin induces endogenous Egr-1 mRNA and Egr-1 promoter activity in primary human endothelial cells by approximately 6-fold and 3-fold, respectively. In transient transfection assays, deletion of the 3′ cluster of serum response elements (SREs), but not the 5′ cluster of SREs, resulted in a loss of thrombin response. When coupled to a heterologous core promoter, a region spanning the 3′ SRE cluster contained information for thrombin response, whereas a region spanning the 5′ SRE cluster had no such effect. A point mutation of the most proximal SRE (SRE-1), but not of the proximal Ets motif or upstream SREs, abrogated the response to thrombin. In electrophoretic mobility shift assays, nuclear extracts from thrombin-treated cells displayed increased binding of total and phosphorylated serum response factor (SRF) to SRE-1. Thrombin-mediated induction of Egr-1 was blocked by inhibitors of MEK1/2, but not by inhibitors of protein kinase C, phosphatidylinositol 3-kinase, or p38 mitogen-activated protein kinase (MAPK). Taken together, these data suggest that thrombin induces Egr-1 expression in endothelial cells by a MAPK-dependent mechanism that involves an interaction between SRF and SRE-1.
Introduction
Thrombin, a multifunctional serine protease derived from the zymogen prothrombin, is a key component of the coagulation cascade, serving to catalyze the conversion of fibrinogen to fibrin. In addition, thrombin plays a role in inflammation by activating a variety of cell types, including endothelial cells, smooth muscle cells, and leukocytes. Most of these cellular effects are mediated by protease-activated receptor-1 (PAR-1), a G-protein coupled receptor. Thrombin cleaves the receptor, unmasking a tethered ligand, which is then free to activate the receptor.1 2
Thrombin signaling in endothelial cells results in the early release of P-selectin,3 prostacyclin, and platelet-activating factor,4 and in the induction of multiple genes, including PAR-1,5 Flk-1/KDR, and Flt-1,6platelet-derived growth factor (PDGF),7 decay-accelerating factor,8 endothelin-1,9interleukin-8,10 monocyte chemotactic protein,11 E-selectin,7,10 vascular cell adhesion molecule-1 (VCAM-1),12 and intercellular adhesion molecule-1 (ICAM-1).12,13 Thrombin activation of endothelial cells results in increased permeability,14,15proliferation,16 angiogenesis,6 and leukocyte-endothelial cell interactions.7,12 17
Egr-1 (also known as zif268, TIS 8, NFGI-A, and Krox 24) is a member of the immediate-early gene family that includes c-fos, c-jun, and early growth response genes.18-23Egr-1, which encodes a serum-inducible zinc finger nuclear phosphoprotein, is rapidly induced in cultured cells by a wide variety of mitogenic and nonmitogenic stimuli. In endothelial cells, Egr-1 has been shown to be activated by acidic fibroblast growth factor,24 basic fibroblast growth factor,25,26 vascular endothelial growth factor (VEGF),27 epidermal growth factor (EGF),28,29 shear stress,30-32 cyclical strain,33 and hypoxia.34,35 In turn, the Egr-1 transcription factor may induce the expression of a wide range of target genes including PDGF-A,36 PDGF-B, tissue factor (TF),27 Flt-1,37 transforming growth factor (TGF)–β, tumor necrosis factor (TNF)–α, urokinase-type plasminogen activator (u-PA), and metalloproteinases.38Endothelial expression of Egr-1 is increased in response to injury.39 Under in vivo conditions, the administration of VEGF and EGF results in vascular bed–specific changes in Egr-1 expression.29 Moreover, elevated Egr-1 levels have been reported in human atherosclerotic lesions.40
We were interested in determining whether thrombin signaling in endothelial cells resulted in increased expression of Egr-1. In the present study, we show that thrombin induces Egr-1 protein and mRNA levels in primary human endothelial cells and that this effect is mediated by the inducible binding of the serum response factor (SRF) to the most proximal of the 5 serum response elements (SREs), namely SRE-1. These data suggest that SRF and Egr-1 be added to the growing list of thrombin-responsive transcription factors.
Materials and methods
Materials
Human thrombin, leech hirudin, thrombin receptor activation peptide (TRAP; SFLLRNPNDKYEPF) and rabbit polyclonal anti–β-actin antibody were obtained from Sigma (St Louis, MO). PD98059, SB203580, bisindolylmaleimide I (BIM), and LY294002 were obtained from Calbiochem (La Jolla, CA). FuGENE 6 transfection reagent was obtained from Roche Molecular Biochemicals (Indianapolis, IN). Antibodies against SRF, p-Elk-1, SAP-1a, Ets-1, Ets-2, p-ERK, and Egr-1 were obtained from Santa Cruz Biotechnology (Santa Cruz, CA). The antibody against phospho-SRF was a generous gift from Michael Greenberg (Harvard Medical School, Boston, MA).
Plasmid construction
The Egr-1 promoter constructs A-G and V were kindly provided by David Cohen (Oregon Health Sciences University, Portland, OR). These latter constructs contain variable lengths of theEgr-1 promoter coupled to the luciferase reporter gene in the promoterless vector pXP2.41 Egr-1-Luc, SRE-A-Luc, SRE-B-Luc, and SRE-C-Luc were generated as previously described.28 To generate mETS1, mSRE1, mSRE2, and mSRE1,2, the 1200-bp Egr-1 promoter was isolated as aKpnI/XhoI fragment from Egr-1-Luc and subcloned into the corresponding sites of the pBluescript vector. The resulting construct (Egr-1-pBlue) was used as a template for site-directed mutagenesis (Quick-Change Site-Directed Mutagenesis Kit, Stratagene, La Jolla, CA), according to the manufacturer's instructions. A 600-bpSfiI/XhoI fragment that spanned the mutated sites was used to replace the corresponding region in Egr-1-Luc. To generate mSRE3,4,5, a construct containing the mutated SRE-3 (generous gift from Michael Waters, University of Queensland, Australia) was used as a template to introduce a mutated SRE-4 by site-directed mutagenesis. A 202-bp StyI/SfiI fragment spanning the mutated SRE-3 and SRE-4 was isolated and used to replace the same region in Egr-1-pBlue. After introducing a mutation of SRE-5 into the resulting construct, theStyI/SfiI region was then used to replace the corresponding region in Egr-1-Luc, giving rise to mSRE3,4,5. Finally, to generate mSRE1,2,3,4,5, the 600-bp SfiI/XhoI fragment from mSRE1,2 was inserted into the corresponding sites in mSRE3,4,5. The following mutations were introduced: GGGCTTCCTGC→GGGCTTaaTGC (mETS-1), CCATATATGG→CCATAagTGG (mSRE-1), CCATATTAGG→CCATAgaAGG (mSRE-2), CCTTATATGG→CCTTAagTGG (mSRE-3), CCTTATTTGG→CCTTATTaat (mSRE-4), CCATATAAGG→CCATATgAaG (mSRE-5). All point mutations were confirmed by automated DNA sequencing.
Cell culture
Human pulmonary artery endothelial cells (HPAECs), human coronary artery endothelial cells (HCAECs), and human umbilical vein endothelial cells (HUVECs) (all are from Clonetics, San Diego, CA) were grown in endothelial growth medium–2-MV (EGM-2-MV; Clonetics) to 95% confluence, serum-starved overnight in endothelial basal medium (EBM)-2 medium containing 0.5% fetal bovine serum (FBS), and incubated in the absence or presence of thrombin at the doses and for the times indicated. In inhibition studies, serum-starved endothelial cells were preincubated with 20 μM PD98059, 1.5 U/mL-5 U/mL hirudin, 5 μM BIM, 10 μM LY294002, or 3 μM SB203580 for 10 minutes and then treated with thrombin for the times indicated.
RNA isolation and ribonuclease (RNase) protection assays
HCAECs or HPAECs were grown in 6-well plates, serum-starved overnight in EBM-2 medium containing 0.5% FBS, and treated with or without thrombin in the presence or absence of inhibitors, as described above. Cells were harvested for RNA by adding RNA-STAT reagent (TEL-TEST, Friendwood, TX) directly into the wells. Total RNA was extracted according to the manufacturer's instructions. For RNase protection assays, a 334-bp human Egr-1 RT-PCR product was subcloned into the pPCR-Script Amp SK(+) cloning vector using PCR-Script AMP Cloning Kit (Stratagene). The Egr-1 cDNA insert was verified by automated sequencing. The cDNA plasmid template for β-actin was purchased from Ambion (Austin, TX). α-[32P] UTP-labeled riboprobes were synthesized from the Egr-1 and β-actin cDNA templates, using T3 RNA polymerase (Ambion) and subsequently gel purified. RNase protection assays were carried out using the RPA III kit (Ambion). Briefly, the 2 riboprobes were mixed with 5 μg total cellular RNA in a volume of 10 μL and hybridized at 42°C overnight in hybridization buffer. The unhybridized RNAs were digested at 37°C for 30 minutes with RNase A/RNase T1 mix. The protected fragments were precipitated by adding the RNase Inactivation/Precipitation III solution and separated on a 4% acrylamide/8M urea gel. The gels were dried and autoradiographed. The intensity of the bands was analyzed by the National Institutes of Health (NIH) Image software. Statistical analyses were carried out using the Student t test.
Western blot analysis of Egr-1 protein expression
To prepare whole-cell protein extracts, cells were washed twice with ice-cold phosphate buffered saline (PBS), harvested with a cell scraper, and lysed in ice-cold lysis buffer containing 20 mM HEPES (N-2-hydroxyethylpiperazine-N'-2-ethanesulfonic acid), pH7.5, 150 mM NaCl, 1% NP-40, 0.1% sodium dodecyl sulfate (SDS), 1 mM EDTA (ethylenediaminetetraacetic acid), 1 mM dithiothreitol (DTT), 0.5 mM phenylmethylsulfonyl fluoride (PMSF), and 1X protease inhibitor cocktail (Boehringer Mannheim, Mannheim, Germany) for 1 hour. The resulting lysates were centrifuged at 10 000g for 20 minutes, and the supernatants were saved as whole-cell protein extracts. Twenty micrograms of protein was separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and electrotransferred to nitrocellulose membranes. Membranes were blocked with 2% nonfat dry milk in Tris (tris(hydroxymethyl)aminomethane)–buffered saline with 0.1% Tween 20 for 1 hour at room temperature. The blot was incubated with primary rabbit polyclonal anti–Egr-1 IgG (1:500 dilution) at room temperature for 1 hour, followed by secondary antibody goat-anti–rabbit horseradish peroxidase conjugate (1:1000 dilution) (Pierce, Rockford, IL). The blot was washed extensively between each incubation step. Peroxidase activity was visualized with an enhanced chemiluminescense substrate system (Amersham, Piscataway, NJ). The blots were subsequently stripped and reprobed with anti–β-actin antibody.
Transfections and analysis of luciferase activity
HPAECs, HCAECs, and HUVECs were transfected using FuGENE 6 reagent as instructed by the manufacturer. 1 × 105cells/well were seeded in 12-well plates 18 to 24 hours before transfection. A total of 0.5 μg of the reporter gene construct and 50 ng of a control plasmid containing the Renilla luciferase reporter gene under the control of a cytomegalovirus (CMV) enhancer/promoter (pRL-CMV; Promega, Madison, WI) was incubated with 2 μL FuGENE 6. After a 24-hour incubation with the transfection mix, cells were washed with PBS, serum-starved in EBM-2 medium containing 0.5% FBS for 18 hours, and treated in the absence or presence of thrombin and inhibitors for 6 hours. The cells were then lysed and assayed for luciferase activity using the Dual-luciferase reporter assay system (Promega). Light activity was measured in a luminometer (Lumat LB 9507; EG & G Berthold, Germany). All experiments were carried out in triplicate and repeated at least 4 times.
Nuclear extracts and electrophoretic mobility shift assays
Nuclear extracts were prepared by a modified Dignam procedure.42 Briefly, HPAECs were harvested by trypsinization, washed in ice-cold PBS, resuspended in one packed-cell volume of buffer A containing 10 mM HEPES (pH 7.9), 1.5 mM MgCl, 10 mM KCl, 50 mM NaF, 2 mM NaVO4, and 0.5 mM DTT and allowed to swell on ice for 15 minutes. The cells were lysed through a narrow-gauge hypodermic needle, and the cell homogenate was centrifuged at 12 000g for 20 seconds. The crude nuclear pellet was resuspended in 2/3 packed-cell volume of buffer C containing 20 mM HEPES, 25% vol/vol glycerol, 0.42 mM NaCl, 1.5 mM MgCl, 0.2 mM EDTA, 50 mM NaF, 2 mM NaVO4, 0.5 mM PMSF, and 0.5 mM DTT and incubated on ice for 30 minutes. The nuclear debris was pelleted by spinning for 5 minutes, and the supernatant dialyzed against buffer D containing 20 mM HEPES, 25% vol/vol glycerol, 0.1 M KCl, 0.2 mM EDTA, 50 mM NaF, 2 mM NaVO4, 0.5 mM PMSF, and 0.5 mM DTT. The oligonucleotides used for electrophoretic mobility shift assays are as follows: SRE-1 forward: 5′-TGCTTCCCATATATGGCCATGTA-3′, SRE-1 reverse: 5′-TACATGGCCATATATGGGAAGCA-3′, SRE-1 forward mutant: 5′-TGCTTCCCATAAGTGGCCATGTA-3′ (TA mutated to AG), SRE-1 reverse mutant: 5′-TACATGGCCACTTATGGGAAGCA-3′ (TA mutated to CT), TTCC forward mutant: 5′-TGCAACCCATATATGGCCATGTA-3′ (TT mutated to AA), TTCC reverse mutant: 5′-TACATGGCCATATATGGGTTGCA-3′ (AA mutated to TT), Ets consensus forward: 5′-GTGCCGGGGTAGGAAGTGGGCTGGG-3′, and Ets consensus reverse: 5′-CCCAGCCCACTTCCTACCCCGGCAC-3′. Ten picomoles of double-stranded oligonucleotides was end-labeled with γ-32P-adenosine triphosphate (ATP) and purified by G-50 spin columns (Amersham Pharmacia). Five micrograms of nuclear extracts was mixed with 1 μg of poly (dI-dC), 2 μL of 5X binding buffer containing 50 mM Tris HCl (pH 8.0), 750 mM KCl, 2.5 mM EDTA, 0.5% Triton-X100, 62.5% vol/vol glycerol, 1 mM DTT, and with 50 000 cpm radiolabeled probes with or without unlabeled competitor probes. For supershift assays, nuclear extracts were preincubated with antibodies against either SRF, phospho-SRF, Elk-1, p-Elk-1, SAP-1a, Ets-1, or Ets-2 for 30 minutes at room temperature before binding to the labeled probes. DNA-protein complexes were resolved on a 5% nondenaturing polyacrylamide gel containing 5% glycerol in 0.5X TBE (50 mM Tris, 50 mM boric acid, and 1 mM EDTA). The gels were dried and autoradiographed. Electrophoretic mobility shift assays were carried out in triplicate, using independent preparations of nuclear extracts. The signals were quantified with NIH Image software, and statistical analyses were carried out using the Student t test.
Immunolocalization studies
Cells were grown to confluence on a glass coverslip, serum-starved overnight, and treated in the absence or presence of 1.5 U/mL thrombin for the times indicated. After removal of culture medium, the cells were washed with PBS and then fixed with ice-cold methanol. After extensive washing in PBS, the cells were incubated with a blocking buffer containing 0.4% bovine serum albumin (BSA) and 0.1% saponin for 10 minutes, followed by primary antibody against human SRF 1:100 for 1 hour at room temperature. Following extensive washes in PBS, the cells were incubated with a fluorescein isothiocyanate (FITC)–conjugated goat-anti–rabbit secondary antibody (1:200 dilution) for 1 hour. The slides were washed in PBS, mounted in 4′,6-diamidino-2′-phenylindole dihydrochloride (DAPI)–containing Vectashield (Vector Laboratories, Burlingame, CA), and examined by fluorescence microscopy.
Results
Thrombin induces Egr-1 expression in primary human endothelial cells
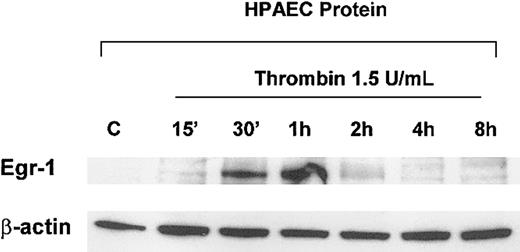
To determine the effect of thrombin on Egr-1 mRNA levels, early passage HPAECs were serum-starved in 0.5% FBS overnight and incubated in the absence or presence of 1.5 U/mL thrombin for 1 hour. As shown in Figure 1A, the addition of thrombin resulted in a 6-fold induction of Egr-1 mRNA, an effect that was mimicked by 10 ng/mL TRAP, a 14–amino acid peptide representing the new amino terminus of PAR-1 after thrombin cleavage. Preincubation with the thrombin protease inhibitor, hirudin, resulted in a dose-dependent inhibition of thrombin's effect on Egr-1 expression (Figure 1A). Thrombin-mediated induction of Egr-1 was both time- and dose-dependent in HPAECs and HCAECs (Figure 1B,C shows HPAECs). In general, Egr-1 mRNA induction was maximal at 30 minutes of treatment with 0.5 U/mL thrombin. Finally, in Western blot analyses, thrombin was shown to increase Egr-1 protein levels, with maximal levels occurring between 30 minutes and 1 hour (Figure 2). Taken together, these results indicate that thrombin induces Egr-1 mRNA and protein levels in primary human endothelial cells.
Thrombin induces Egr-1 mRNA in primary endothelial cells.
HPAECs were grown on 6-well plates and starved overnight in EBM-2 medium containing 0.5% FBS. The cells were treated in the absence or presence of thrombin at the doses and for the times indicated and subsequently harvested for total RNA. RNase protection assays were carried out with α-[32P] UTP–labeled Egr-1 and β-actin probes as described in “Materials and methods.” Densitometry was used to calculate the ratio of Egr-1 and β-actin signals. The results are expressed as fold induction relative to control untreated cells. The data represent the means and standard deviations from 3 independent experiments. (A) HPAECs were incubated in the absence (Control) or presence of 1.5 U/mL thrombin, 10 ng/mL TRAP, 5 U/mL hirudin, or 1.5 U/mL thrombin plus hirudin (1.5-5 U/mL) for 1 hour. *P < .01 relative to control untreated cells. (B) HPAECs were incubated in the absence (“C” indicates control) or presence of 1.5 U/mL thrombin for the times indicated. (Panel C) HPAECs were incubated in the absence (“C” indicates control) or presence of thrombin for 1 hour at the doses indicated.
Thrombin induces Egr-1 mRNA in primary endothelial cells.
HPAECs were grown on 6-well plates and starved overnight in EBM-2 medium containing 0.5% FBS. The cells were treated in the absence or presence of thrombin at the doses and for the times indicated and subsequently harvested for total RNA. RNase protection assays were carried out with α-[32P] UTP–labeled Egr-1 and β-actin probes as described in “Materials and methods.” Densitometry was used to calculate the ratio of Egr-1 and β-actin signals. The results are expressed as fold induction relative to control untreated cells. The data represent the means and standard deviations from 3 independent experiments. (A) HPAECs were incubated in the absence (Control) or presence of 1.5 U/mL thrombin, 10 ng/mL TRAP, 5 U/mL hirudin, or 1.5 U/mL thrombin plus hirudin (1.5-5 U/mL) for 1 hour. *P < .01 relative to control untreated cells. (B) HPAECs were incubated in the absence (“C” indicates control) or presence of 1.5 U/mL thrombin for the times indicated. (Panel C) HPAECs were incubated in the absence (“C” indicates control) or presence of thrombin for 1 hour at the doses indicated.
Thrombin induces Egr-1 protein in primary endothelial cells.
HPAECs were grown on 6-well plates and starved overnight in EBM-2 medium containing 0.5% FBS. The cells were treated in the absence (C) or presence of 1.5 U/mL thrombin for the times indicated and subsequently harvested for whole-cell protein extracts as described in “Materials and methods.” In Western blot analysis, 20 μg of cell lysates was separated by 10% SDS-PAGE, transferred to a nitrocellulose membrane, and probed with a polyclonal anti–Egr-1 antibody. The membranes were stripped and reprobed for β-actin to control for loading.
Thrombin induces Egr-1 protein in primary endothelial cells.
HPAECs were grown on 6-well plates and starved overnight in EBM-2 medium containing 0.5% FBS. The cells were treated in the absence (C) or presence of 1.5 U/mL thrombin for the times indicated and subsequently harvested for whole-cell protein extracts as described in “Materials and methods.” In Western blot analysis, 20 μg of cell lysates was separated by 10% SDS-PAGE, transferred to a nitrocellulose membrane, and probed with a polyclonal anti–Egr-1 antibody. The membranes were stripped and reprobed for β-actin to control for loading.
Thrombin induces Egr-1 promoter activity in primary human endothelial cells
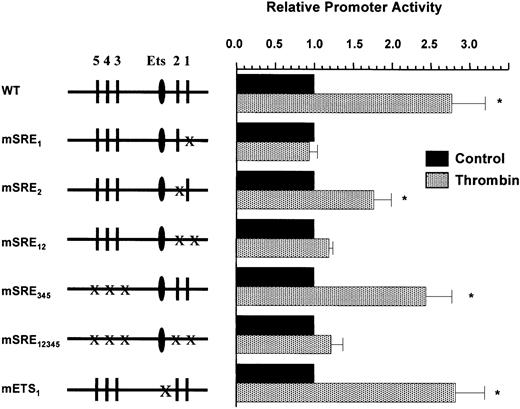
We next tested whether the Egr-1 promoter contained information for thrombin response. HCAECs, HPAECs, and HUVECs were transiently transfected with the 1200-bp Egr-1 promoter construct (Egr-1-Luc), serum-starved for 18 hours, incubated in the absence or presence of thrombin for 6 hours, and harvested for luciferase activity. Egr-1 promoter activity was induced in all 3 cell types by 2.5- to 3-fold (Figure3A). Since the transfection efficiency was greater in HUVECs compared with HPAECs or HCAECs, HUVECs were used in subsequent experiments. To delineate the promoter regions responsible for mediating the Egr-1 response to thrombin, a series of 5′ and internal deletion mutants of the Egr-1 promoter were transiently transfected into HUVECs (Figure 3B). Deletion of the upstream AP-1 binding sites (Egr-1-C) had no effect on thrombin induction. The deletion of regions containing the 3′ SRE cluster abolished thrombin response (Egr-1-B, Egr-1-D, and Egr-1-G), whereas the isolated deletion of 5′ SRE cluster had no such effect (Egr-1 E). When coupled to a heterologous core promoter, the region spanning the 5′ SRE cluster (−425 to −250) did not confer thrombin response (Figure 3C). In contrast, the fragment containing the 3′ SRE cluster (−125 to −70) alone or in combination with the 5′ SRE cluster was induced 2.4-fold by thrombin (Figure 3C). These data indicate that the region between −125 and −70 of the Egr-1 promoter contains information for thrombin response. To determine whether SRE-1 and/or SRE-2 was responsible for transducing the thrombin signal, we generated a series of SRE mutants and tested the resulting promoter-reporter gene constructs for thrombin response in transient transfection assays. As shown in Figure 4, constructs containing point mutations of all 5 SREs (mSRE1,2,3,4,5), the 2 proximal SREs (mSRE1,2), or SRE-1 alone (mSRE1) did not respond to thrombin treatment. A point mutation of SRE-2 (mSRE2) resulted in a slight decrease in thrombin response, whereas a mutation of the 5′ SRE cluster (mSRE3,4,5) had no effect on thrombin stimulation. Taken together, these findings strongly implicate SRE-1 as the thrombin-responsive element of theEgr-1 promoter. The 3′ SRE cluster has been shown to contain a single Ets motif at position −93, relative to the start site of transcription.43 To test whether this site was necessary for mediating the thrombin response, a mutation was introduced into the Ets motif, and the resulting plasmid was transfected into HUVECs. As shown in Figure 4, this construct retained thrombin responsiveness, suggesting that the proximal Ets site is not necessary for transducing the thrombin signal.
Thrombin induces Egr-1 promoter activity in primary endothelial cells.
Primary human endothelial cells were transiently transfected with 0.5 μg of Egr-1 promoter reporter gene constructs, serum-starved in EBM-2 medium containing 0.5% FBS for 18 hours, treated in the absence or presence of 1.5 U/mL thrombin for 6 hours, and then harvested for luciferase activity. The results show the means and standard deviations of luciferase light units (relative to control untreated cells) obtained in triplicate from 4 independent experiments. Luciferase light units were corrected for transfection efficiency as described in “Materials and methods.” (A) HPAECs, HCAECs, or HUVECs were transiently transfected with the full-length wild-type 1200-bp Egr-1-Luc plasmid. (B) HUVECs were transiently transfected with a series of 5′ or internal deletion mutants of the Egr-1promoter coupled to a luciferase reporter gene (pXP2). Sequences are numbered relative to the start site of transcription. The SREs are shown in bars. (C) HUVECs were transiently transfected with SRE heterologous promoter constructs, in whichEgr-1 promoter fragments encompassing the 5′ SRE cluster, 3′ SRE cluster, or both clusters were fused upstream of a minimalvon Willebrand factor (VWF) core promoter as previously described.28 For all panels, *P < .01, compared with control untreated cells.
Thrombin induces Egr-1 promoter activity in primary endothelial cells.
Primary human endothelial cells were transiently transfected with 0.5 μg of Egr-1 promoter reporter gene constructs, serum-starved in EBM-2 medium containing 0.5% FBS for 18 hours, treated in the absence or presence of 1.5 U/mL thrombin for 6 hours, and then harvested for luciferase activity. The results show the means and standard deviations of luciferase light units (relative to control untreated cells) obtained in triplicate from 4 independent experiments. Luciferase light units were corrected for transfection efficiency as described in “Materials and methods.” (A) HPAECs, HCAECs, or HUVECs were transiently transfected with the full-length wild-type 1200-bp Egr-1-Luc plasmid. (B) HUVECs were transiently transfected with a series of 5′ or internal deletion mutants of the Egr-1promoter coupled to a luciferase reporter gene (pXP2). Sequences are numbered relative to the start site of transcription. The SREs are shown in bars. (C) HUVECs were transiently transfected with SRE heterologous promoter constructs, in whichEgr-1 promoter fragments encompassing the 5′ SRE cluster, 3′ SRE cluster, or both clusters were fused upstream of a minimalvon Willebrand factor (VWF) core promoter as previously described.28 For all panels, *P < .01, compared with control untreated cells.
SRE-1 is necessary for mediating thrombin stimulation of the Egr-1 promoter in primary endothelial cells.
HUVECs were transiently transfected with a series of point mutants of the Egr-1 promoter coupled to a luciferase reporter gene (pGL2-basic) and exposed to 1.5 U/mL of thrombin for 6 hours. The results show the means and standard deviations of luciferase light units (relative to control untreated cells) obtained in triplicate from 4 independent experiments. Luciferase light units were corrected for transfection efficiency as described in “Materials and methods.” *P < .01, compared with control untreated cells.
SRE-1 is necessary for mediating thrombin stimulation of the Egr-1 promoter in primary endothelial cells.
HUVECs were transiently transfected with a series of point mutants of the Egr-1 promoter coupled to a luciferase reporter gene (pGL2-basic) and exposed to 1.5 U/mL of thrombin for 6 hours. The results show the means and standard deviations of luciferase light units (relative to control untreated cells) obtained in triplicate from 4 independent experiments. Luciferase light units were corrected for transfection efficiency as described in “Materials and methods.” *P < .01, compared with control untreated cells.
Thrombin induces the binding of SRF to SRE-1
Next, we used electrophoretic mobility shift assays to determine the effect of thrombin on DNA-protein interactions. Since SRE-1, but not the proximal Ets motif, was necessary for mediating the thrombin response in transient transfection assays, we focused on the SRE-1 sequence alone (Figure 5A, SRE-1 probe). As shown in Figure 5B, incubation of the probe with nuclear extracts from untreated and thrombin-treated HPAECs resulted in specific a DNA-protein complex (closed arrow, Figure 5B, lanes 1 and 13). The DNA-protein complex was inhibited by the addition of a 100-fold molar excess of unlabeled cold-competitor (Figure 5B, lanes 2 and 14), but not by the same concentration of unlabeled mutant SRE-1 (Figure 5B, lanes 3 and 15) or unlabeled consensus Ets probe (derived from the human Flt-1 promoter; Figure 5B, lanes 12 and 24). Of note, the SRE-1 site contains a potential overlapping Ets binding domain (TTCC; Figure 5A). However, the addition of 100-fold molar excess of unlabeled cold-competitor containing a mutation of the TTCC sequence (Figure 5B, lanes 4 and 16) resulted in a complete loss of DNA-protein complexes, arguing against the presence of a specific Ets DNA-protein complex.
Thrombin induces binding of SRF and phospho-SRF to SRE-1 in primary endothelial cells.
(A) Shown is a schematic representation of the probe sequences used in electrophoretic mobility shift assays. The consensus SRE motif and potential Ets site (TTCC) in the SRE-1 probe are underlined and bolded, respectively. The mutated bases in the SRE-1 and TTCC mutant probes are represented by asterisks (*). The Ets consensus probe (Ets site in bold) is based on sequence from the human Flt-1promoter.67 (B) Electrophoretic mobility shift assays were performed with γ-[32P]–labeled SRE-1 probe and 5 μg nuclear extract from serum-starved HUVECs treated in the absence (− Thrombin, lanes 1-12) or presence (+ Thrombin, lanes 13-24) of 1.5 units/mL thrombin. In competition assays, a 100-fold molar excess of unlabeled wild-type (lanes 2 and 14), SRE-1 mutant (lanes 3 and 15), TTCC mutant (lanes 4 and 16), or Ets consensus (lanes 12 and 24) probe was added to the reaction mixture. The closed arrow indicates specific DNA-protein complexes. The open arrows indicate nonspecific DNA-protein complexes. In supershift assays, nuclear extracts were preincubated with antibodies against SRF (lanes 5 and 17), phospho-SRF (lanes 6 and 18), Elk-1 (lanes 7 and 19), p-Elk-1 (lanes 8 and 20), SAP-1a (lanes 9 and 21), Ets-1 (lanes 10 and 22), or Ets-2 (lanes 11 and 23). The asterisk indicates the super-shifted complex.
Thrombin induces binding of SRF and phospho-SRF to SRE-1 in primary endothelial cells.
(A) Shown is a schematic representation of the probe sequences used in electrophoretic mobility shift assays. The consensus SRE motif and potential Ets site (TTCC) in the SRE-1 probe are underlined and bolded, respectively. The mutated bases in the SRE-1 and TTCC mutant probes are represented by asterisks (*). The Ets consensus probe (Ets site in bold) is based on sequence from the human Flt-1promoter.67 (B) Electrophoretic mobility shift assays were performed with γ-[32P]–labeled SRE-1 probe and 5 μg nuclear extract from serum-starved HUVECs treated in the absence (− Thrombin, lanes 1-12) or presence (+ Thrombin, lanes 13-24) of 1.5 units/mL thrombin. In competition assays, a 100-fold molar excess of unlabeled wild-type (lanes 2 and 14), SRE-1 mutant (lanes 3 and 15), TTCC mutant (lanes 4 and 16), or Ets consensus (lanes 12 and 24) probe was added to the reaction mixture. The closed arrow indicates specific DNA-protein complexes. The open arrows indicate nonspecific DNA-protein complexes. In supershift assays, nuclear extracts were preincubated with antibodies against SRF (lanes 5 and 17), phospho-SRF (lanes 6 and 18), Elk-1 (lanes 7 and 19), p-Elk-1 (lanes 8 and 20), SAP-1a (lanes 9 and 21), Ets-1 (lanes 10 and 22), or Ets-2 (lanes 11 and 23). The asterisk indicates the super-shifted complex.
The faster-migrating DNA-protein complexes were nonspecific (open arrows), as they were inhibited by the addition of wild-type and mutant self-competitors as well as competitor sequence from an unrelated gene (consensus Ets probe from the Flt-1 promoter). To determine the identity of the protein in the specific DNA-protein complex, the binding reactions were incubated with antibodies to SRF, phospho-SRF, Elk-1, p-Elk-1, SAP-1a, Ets-1, and Ets-2. Preincubation with antibodies to SRF (Figure 5B, lanes 5 and 17) and phospho-SRF (Figure 5B, lanes 6 and 18) resulted in a supershift of the specific DNA-protein complex (Figure 5B, asterisk), whereas the addition of IgG control (not shown) or antibodies against Elk-1 (Figure 5B, lanes 7 and 19), p-Elk-1 (Figure 5B, lanes 8 and 20), SAP-1a (Figure 5B, lanes 9 and 21), Ets-1 (Figure 5B, lanes 10 and 22), or Ets-2 (Figure 5B, lanes 11 and 23) did not produce any change in mobility.
Nuclear extracts from thrombin-treated endothelial cells displayed increased levels of total SRF binding (1.93 ± 0.17-fold,P < .05; Figure 5B, compare lanes 1 and 5 with lanes 13 and 17, respectively). Previous studies have shown that the inducible phosphorylation of SRF results in increased DNA binding avidity. To determine whether thrombin induces the binding of phosphorylated SRF to SRE-1, we carried out supershift assays with an anti–phospho-SRF antibody. The addition of this antibody resulted in a stronger supershift of the SRE-SRF complex in thrombin treated cells compared with untreated controls (1.94 ± 0.19-fold, P < .05; Figure 5B, compare lanes 6 and 18). Taken together, the data suggest that thrombin induces the binding of both total and phosphorylated SRF to SRE-1.
Thrombin-mediated induction of the Egr-1 gene and promoter is mediated by a MEK1/2-dependent MAPK pathway
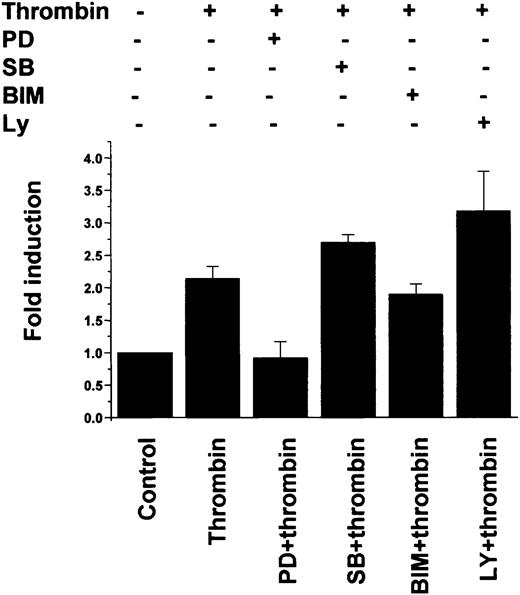
To delineate the signaling pathway involved in mediating the thrombin response, we carried out RNase protection assays of Egr-1 mRNA from untreated and thrombin-treated HPAECs preincubated in the absence or presence of chemical inhibitors of mitogen-activated protein kinase (MAPK), protein kinase C (PKC), and phosphatidylinositol 3-kinase (PI3K). Thrombin-mediated induction of Egr-1 expression was blocked by pretreatment with the MAPK kinase (MEK) inhibitor PD98059 (Figure6, PD), but not by the p38 MAPK inhibitor SB203580 (Figure 6, SB), the PKC inhibitor BIM (Figure 6, BIM), or the PI3K inhibitor LY294002 (Figure 6, LY). Similarly, in transient transfection assays, thrombin-mediated induction of the full-lengthEgr-1 promoter was abrogated by pretreatment with PD98059, but not with SB203580, BIM, or LY294002 (Figure7). These findings suggest that thrombin stimulation of the endogenous Egr-1 gene and the Egr-1promoter is mediated by an ERK1/2 MAPK-dependent, p38/PKC/PI3K-independent pathway.
Thrombin stimulation of Egr-1 mRNA in primary endothelial cells is mediated by an ERK1/2 MAPK–dependent pathway.
HPAECs were starved overnight in EBM-2 medium containing 0.5% FBS and then treated with or without 1.5 U/mL thrombin for 1 hour. Alternatively, the serum-starved cells were preincubated with 20 μM PD98059 (PD), 3 μM SB203580 (SB), 5 μM BIM, 10 μM LY294002 (LY), or dimethyl sulfoxide (DMSO) (1 μL as solvent control) for 10 minutes and then incubated for 1 hour with medium containing no thrombin or 1.5 U/mL thrombin. RNase protection assays were carried out with α-[32P] UTP-labeled Egr-1 and β-actin probes as described in “Materials and methods.” Densitometry was used to calculate the ratio of Egr-1 and β-actin signals. The results are expressed as fold induction relative to control untreated cells. The data represent the means and standard deviations from 3 independent experiments.
Thrombin stimulation of Egr-1 mRNA in primary endothelial cells is mediated by an ERK1/2 MAPK–dependent pathway.
HPAECs were starved overnight in EBM-2 medium containing 0.5% FBS and then treated with or without 1.5 U/mL thrombin for 1 hour. Alternatively, the serum-starved cells were preincubated with 20 μM PD98059 (PD), 3 μM SB203580 (SB), 5 μM BIM, 10 μM LY294002 (LY), or dimethyl sulfoxide (DMSO) (1 μL as solvent control) for 10 minutes and then incubated for 1 hour with medium containing no thrombin or 1.5 U/mL thrombin. RNase protection assays were carried out with α-[32P] UTP-labeled Egr-1 and β-actin probes as described in “Materials and methods.” Densitometry was used to calculate the ratio of Egr-1 and β-actin signals. The results are expressed as fold induction relative to control untreated cells. The data represent the means and standard deviations from 3 independent experiments.
Thrombin stimulation of Egr-1 promoter activity in primary endothelial cells is mediated by an ERK1/2 MAPK–dependent pathway.
HUVECs were transiently transfected with 0.5 μg of full-length wild-type 1200-bp Egr-1-Luc plasmid, serum-starved in 0.5% FBS for 18 hours, treated in the absence (Control) or presence of 1.5 U/mL thrombin for 6 hours, and harvested for luciferase activity. Where indicated, the transfected cells were preincubated with 20 μM PD98059 (PD), 3 μM SB203580 (SB), 5 μM BIM, or 10 μM LY294002 (LY) for 10 minutes. The results show the means and standard deviations of luciferase light units (relative to untreated cells) obtained in triplicate from 4 independent experiments. Luciferase light units were corrected for transfection efficiency as described in “Materials and methods.”
Thrombin stimulation of Egr-1 promoter activity in primary endothelial cells is mediated by an ERK1/2 MAPK–dependent pathway.
HUVECs were transiently transfected with 0.5 μg of full-length wild-type 1200-bp Egr-1-Luc plasmid, serum-starved in 0.5% FBS for 18 hours, treated in the absence (Control) or presence of 1.5 U/mL thrombin for 6 hours, and harvested for luciferase activity. Where indicated, the transfected cells were preincubated with 20 μM PD98059 (PD), 3 μM SB203580 (SB), 5 μM BIM, or 10 μM LY294002 (LY) for 10 minutes. The results show the means and standard deviations of luciferase light units (relative to untreated cells) obtained in triplicate from 4 independent experiments. Luciferase light units were corrected for transfection efficiency as described in “Materials and methods.”
Thrombin induces the nuclear translocation of SRF
In the last set of experiments, we used immunofluorescent studies to study the effect of thrombin on SRF localization in HPAECs. Thrombin treatment for 5 to 120 minutes resulted in the nuclear translocation of SRF, with maximal levels occurring at 30 minutes (Figure8 shows 5 minutes and 30 minutes). Together with our previous results, these data suggest that thrombin induces the nuclear translocation of SRF and that this effect is coupled to the transactivation of the Egr-1 promoter.
Thrombin induces the nuclear translocation of SRF in primary endothelial cells.
HPAECs were plated onto 4-well chamber slides, serum-starved overnight, and incubated in the absence (Control) or presence of 1.5 U/mL thrombin for the times indicated. The cells were fixed in ice-cold (4°C) methanol for 10 minutes, washed with PBS, and subsequently incubated with primary anti-SRF antibody, followed by FITC-conjugated second antibody at room temperature. The nuclei were stained with DAPI. Bar, 10 μm. All panels are shown at the same magnification.
Thrombin induces the nuclear translocation of SRF in primary endothelial cells.
HPAECs were plated onto 4-well chamber slides, serum-starved overnight, and incubated in the absence (Control) or presence of 1.5 U/mL thrombin for the times indicated. The cells were fixed in ice-cold (4°C) methanol for 10 minutes, washed with PBS, and subsequently incubated with primary anti-SRF antibody, followed by FITC-conjugated second antibody at room temperature. The nuclei were stained with DAPI. Bar, 10 μm. All panels are shown at the same magnification.
Discussion
The coagulation and inflammatory pathways are tightly linked. Inflammatory mediators induce the expression of tissue factor on circulating monocytes, thereby contributing to the initiation of coagulation. Moreover, cytokines and other inflammatory mediators activate endothelial cells, resulting in a local alteration of the hemostatic balance.44 45 Thrombin, once generated, may further activate endothelial cells and amplify the proinflammatory response.
Until recently, little was known about the mechanisms by which thrombin induces gene expression. Thrombin has subsequently been shown to stimulate PDGF-B chain expression via the inducible binding of a Y-box protein to a CCACCC consensus sequence in the upstream promoter region.46,47 A similar mechanism was proposed for the thrombin response of the endothelial protein C receptor.48Thrombin-mediated induction of ICAM-1 was demonstrated to involve the binding of p65 homodimers to a single NF-κB site in the upstream promoter region,13 while the effect of thrombin on VCAM-1 was shown to depend on tandem NF-κB and GATA motifs.49Finally, thrombin has been reported to increase the expression of the chemokine, 9E3/cCAF, through the induction of phosphorylated Elk-1-DNA protein interactions.50 Based on the results of the present study, we propose that both SRF and Egr-1 be added to the list of thrombin-responsive transcription factors.
The murine and human Egr-1 promoters contain 5 functional SREs, organized into 2 clusters: a 5′ cluster of 3 SREs and a 3′ cluster of 2 SREs. Collectively, the 5 SREs are responsible for transducing most, if not all, signals that activate Egr-1 expression. The 5′ SRE cluster has been shown to mediate the response to the majority of extracellular signals, including EGF,28 shear stress,30 growth hormone,51urea,41 hypotonicity, lipopolysaccharide (LPS),52 granulocyte colony-stimulating factor (G-CSF),53 and hypoxia.35 In nonendothelial cells, the 3′ cluster has been implicated in the response to glucose-induced depolarization54 and granulocyte-macrophage colony-stimulating factor (GM-CSF).55 56 To our knowledge, the present study is the first to uncover a role for the most proximal SRE (SRE-1) in mediating inducible Egr-1 expression in primary endothelial cells.
The SRE is an important converging point for signal transduction cascades. The SRF is a ubiquitous 67-kDa nuclear transcription factor that acts by binding to the SRE consensus site, CC(A/T)6GG. The prototypic SRE binds to a ternary complex that includes a homodimer of SRF and member of the ternary complex factors, including Elk-1, SAP-1, and SAP-2/ERP/Net. Previous studies have shown that the response of the 5′ cluster of SREs to extracellular signals is mediated by the coordinate action of SRF and ternary complex factor (TCF).30,35,51 52 In contrast, our results suggest that SRE-1 transduces the thrombin signal by an SRF-dependent, TCF-independent mechanism. For example, the mutation of SRE-1, but not the proximal Ets motif, resulted in a loss of thrombin responsiveness. Moreover, in electrophoretic mobility shift assays, thrombin resulted in the inducible binding of SRF and phospho-SRF to SRE-1 in the absence of TCF.
Consistent with our findings, a previous study of preadipocyte cells reported that SRE-1 is constitutively bound by SRF but not by TCF.51 Thrombin receptor activation in NIH3T3 cells resulted in an SRF-dependent, TCF-independent transactivation of the c-fos SRE.57 In pancreatic islet β-cells, depolarization has been reported to induce Egr-1 gene expression via the 3′ cluster of SREs.54 Moreover, the effect of depolarization on SRE-mediated gene transcription was shown to depend on SRF, but not Elk-1.58 Together with the published data, the results of the present study suggest that while signal transduction through the 5′ cluster of SREs requires the cooperative interaction of SRF and TCF, the response of the 3′ cluster is mediated by SRF alone.
Previous investigations have shown that phosphorylation of SRF at Ser-103 results in increased DNA binding and enhanced transcriptional activity.59,60 Several signaling pathways have been implicated in this process, including ERK1/2-dependent MAPKAP-K1,60 p38-dependent MAPKAP-K2,61 and CaM kinases.58 We demonstrated that thrombin induces the binding of phospho-SRF to SRE-1, suggesting that thrombin's actions may be mediated by the inducible phosphorylation of the transcription factor. Another possible mechanism by which thrombin mediates SRE-dependent induction of the Egr-1 gene is through the nuclear translocation of SRF, an effect that may be regulated independently of Ser-103 phosphorylation.62 The observation that thrombin induces the translocation of SRF from the cytoplasm to the nucleus is consistent with the previously reported effects of serum on the subcellular distribution of SRF.63
We have shown that thrombin-mediated induction of Egr-1 is dependent on MEK/ERK, but not p38 MAPK, PKC, or PI3K. Previous studies have established an important role for MEK/ERK in mediating Egr-1 response to other mediators, including LPS, growth factors, hormones, and TNF-α.28,52,64,65 PKC has been shown to play a role in mediating VEGF64 and hypoxia35 stimulation of Egr-1, while PI3K has been implicated in the SRF-dependent, TCF-independent induction of c-fos in 3T3-L1 cells.66Taken together, these data point to the existence of a large repertoire of signaling pathways that couple the extracellular environment to immediate early gene expression.
There is a growing appreciation that thrombin signaling contributes to a proinflammatory state. Egr-1 has been reported to induce a number of downstream genes, including TF, TNF-α, TGF-β, u-PA, and PDGF-B.39,52 The effect of Egr-1 on an individual target gene is likely to vary according to the cell type and extracellular signal.64 An important goal for future studies will be to characterize the nature of the Egr-1–responsive gene program in thrombin-treated endothelial cells and to determine the extent to which these gene products contribute to the activation phenotype.
Prepublished online as Blood First Edition Paper, June 28, 2002; DOI 10.1182/blood-2002-02-0415.
Supported by the National Institute of Health grant HL 65216-03.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
William C. Aird, Beth Israel Deaconess Medical Center, Molecular Medicine, RW-663, 330 Brookline Ave, Boston, MA 02215; e-mail: waird@caregroup.harvard.edu.

![Fig. 1. Thrombin induces Egr-1 mRNA in primary endothelial cells. / HPAECs were grown on 6-well plates and starved overnight in EBM-2 medium containing 0.5% FBS. The cells were treated in the absence or presence of thrombin at the doses and for the times indicated and subsequently harvested for total RNA. RNase protection assays were carried out with α-[32P] UTP–labeled Egr-1 and β-actin probes as described in “Materials and methods.” Densitometry was used to calculate the ratio of Egr-1 and β-actin signals. The results are expressed as fold induction relative to control untreated cells. The data represent the means and standard deviations from 3 independent experiments. (A) HPAECs were incubated in the absence (Control) or presence of 1.5 U/mL thrombin, 10 ng/mL TRAP, 5 U/mL hirudin, or 1.5 U/mL thrombin plus hirudin (1.5-5 U/mL) for 1 hour. *P < .01 relative to control untreated cells. (B) HPAECs were incubated in the absence (“C” indicates control) or presence of 1.5 U/mL thrombin for the times indicated. (Panel C) HPAECs were incubated in the absence (“C” indicates control) or presence of thrombin for 1 hour at the doses indicated.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/100/13/10.1182_blood-2002-02-0415/4/m_h82423529001.jpeg?Expires=1765971642&Signature=OM3AsYO2ATxDvameF6~Ou1dMWc3rT~JFMxEMZ6b~zk~QhTYSbutmLc7n~o8YiD1-7MTMayd6wDb2vIsazDpxMlIgH0YDGTHbe6EuGR2uLuE9BxvrvCaqkDkVSWrTn-~o3gbYq0Wi5~22Fi98yjFX5uslnMqa3MkUn0g86Y0AUPkzh4bvqzKn9I4aassbpZXNcEpprXsje9oxrtZbazwporeWOE3BfthOQCuJF-IWtpAiTPYHb0W3yUTEY7kFw97HTUWEHF-6rIcuKjjsEPYnbNJaK7Lu0WL~uBssogCcYUyd~XPxqLwUgenD9LYPF3ZN8xSMkE892abyVKZILfo3TA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Fig. 5. Thrombin induces binding of SRF and phospho-SRF to SRE-1 in primary endothelial cells. / (A) Shown is a schematic representation of the probe sequences used in electrophoretic mobility shift assays. The consensus SRE motif and potential Ets site (TTCC) in the SRE-1 probe are underlined and bolded, respectively. The mutated bases in the SRE-1 and TTCC mutant probes are represented by asterisks (*). The Ets consensus probe (Ets site in bold) is based on sequence from the human Flt-1promoter.67 (B) Electrophoretic mobility shift assays were performed with γ-[32P]–labeled SRE-1 probe and 5 μg nuclear extract from serum-starved HUVECs treated in the absence (− Thrombin, lanes 1-12) or presence (+ Thrombin, lanes 13-24) of 1.5 units/mL thrombin. In competition assays, a 100-fold molar excess of unlabeled wild-type (lanes 2 and 14), SRE-1 mutant (lanes 3 and 15), TTCC mutant (lanes 4 and 16), or Ets consensus (lanes 12 and 24) probe was added to the reaction mixture. The closed arrow indicates specific DNA-protein complexes. The open arrows indicate nonspecific DNA-protein complexes. In supershift assays, nuclear extracts were preincubated with antibodies against SRF (lanes 5 and 17), phospho-SRF (lanes 6 and 18), Elk-1 (lanes 7 and 19), p-Elk-1 (lanes 8 and 20), SAP-1a (lanes 9 and 21), Ets-1 (lanes 10 and 22), or Ets-2 (lanes 11 and 23). The asterisk indicates the super-shifted complex.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/100/13/10.1182_blood-2002-02-0415/4/m_h82423529005.jpeg?Expires=1765971642&Signature=LrryOIH33ZFHrf0YnU3OPkai20~Inp5PrHUltNU8l1WCdYB8OHApCBHt3CzEOCiZWU1S4zefT23qskdlmrco~trQh7e9DGW5OiirjtdjYO8LTnCHe5zRT08g9r-PYJ1srZgyOpVU6rkh2TpPfWQ-E0tuAfSK9YwAACAvxZRaSMwHjFsjKhvkL-XueN6EVlBLBSM8k4tLfnDFfCzHJHJcqEHoCxiQxbXDS7ecFn2wBiLTH1mP9wpLZR2MgLTtPQOieAxWjxRwys-fFWD3r7Q768czik7CQb-dMP8VOcHOk9fWIRcYss4YlRURIkrz1cZV2g33FANpEIBk39Mes06OFQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 6. Thrombin stimulation of Egr-1 mRNA in primary endothelial cells is mediated by an ERK1/2 MAPK–dependent pathway. / HPAECs were starved overnight in EBM-2 medium containing 0.5% FBS and then treated with or without 1.5 U/mL thrombin for 1 hour. Alternatively, the serum-starved cells were preincubated with 20 μM PD98059 (PD), 3 μM SB203580 (SB), 5 μM BIM, 10 μM LY294002 (LY), or dimethyl sulfoxide (DMSO) (1 μL as solvent control) for 10 minutes and then incubated for 1 hour with medium containing no thrombin or 1.5 U/mL thrombin. RNase protection assays were carried out with α-[32P] UTP-labeled Egr-1 and β-actin probes as described in “Materials and methods.” Densitometry was used to calculate the ratio of Egr-1 and β-actin signals. The results are expressed as fold induction relative to control untreated cells. The data represent the means and standard deviations from 3 independent experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/100/13/10.1182_blood-2002-02-0415/4/m_h82423529006.jpeg?Expires=1765971642&Signature=HA0~F6~7Y9Qk-DMQOMqiDyLhDVzweMV2Sdq6gnX6Hlv1rS5afL8NV8OMqmL~-ceZ8y2govOG3iIv0z-uX9XJ9jN7RDgAl5e-h8iiVJH9dtrwbg-sjdkTGgDG-RCGvPejUAg5~0-LTpjuJiHo93eNWWld~ZyRYKQfP1-AwHJTuejsSh8cUcCRVodLwCKqxhVCmb2OkfoBFWuYekkjwd0DWjUMkorul4MUDbumuhA96yho4d2Jm4ggHOpu5R0F9iHSgHrpyiSLhVaIc1F5S6Mhz8SH0rwwtjcgOmYhiTx2IN9CWSoozrH2WLgno26K7fhLYCTLmV-QuHVvy~CpPopa~Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal