In this issue of Blood, Montoro et al describe the influence of TP53 gene mutations and its allelic burden in the largest cohort of patients with myelodysplastic syndromes with the 5q deletion [MDS-del(5q)] published so far.1
The identification of MDS-del(5q) as a distinct entity within the myelodysplastic syndromes was recognized in 2001 by the creation of a separate category in the World Health Organization (WHO) classification. This disease is usually associated with a good prognosis if bone marrow blasts are <5% and the cytogenetic abnormality is isolated (or at least not part of a complex karyotype). The underlying biology has been defined, and it has a unique sensitivity to lenalidomide.2 With the advent of high-throughput sequencing, the presence of TP53 mutations in MDS-del(5q) has been identified. Although initially described in patients at the time of disease progression, it now has been found during the chronic phase of the disease. Jädersten et al first identified the presence of very small mutated TP53 clones in 18% of patients with MDS-del(5q) and established a link between these clones and the risk of disease progression, although conflicting results were also reported.3 A major international study by Bernard et al showed that, in a large MDS population including all types of MDS, only biallelic inactivation of TP53 had a prognostic impact on the outcome, whereas monoallelic inactivation of this gene had no impact.4 In that series, the effect of monoallelic TP53m on overall survival was not changed in patients with del(5q). Finally, the recent WHO 2022 and International Consensus Classification (ICC) 2022 guidelines have recognized as a distinct entity patients with MDS carrying the TP53 mutation,5,6 with a rather poor prognosis. It is unfortunate that the definitions of these entities differ between the 2 classifications. The WHO classification recognizes the existence of a group of patients with MDS with biallelic inactivation of TP53, whereas the ICC categorizes groups of patients with more or less than 10% blasts. The challenge is therefore to balance 2 distinct forms of MDS: MDS-del(5q) with a good prognosis and MDS with TP53 inactivation with a poor prognosis, knowing that there is overlap between the 2 forms. In their study, Montoro et al aim to clarify this issue and establish criteria dedicated to MDS-del(5q) in identifying the prognostic impact of TP53 mutations.
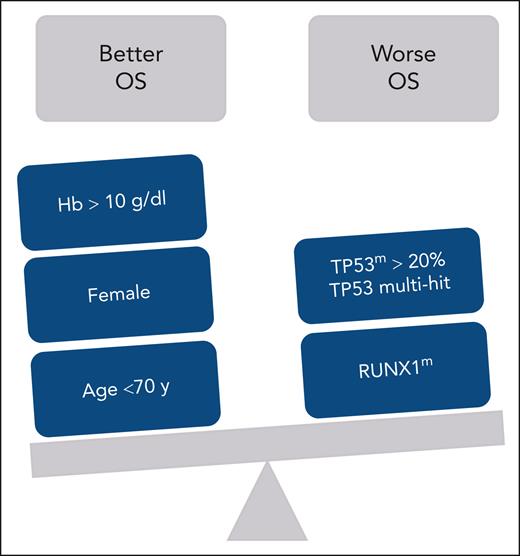
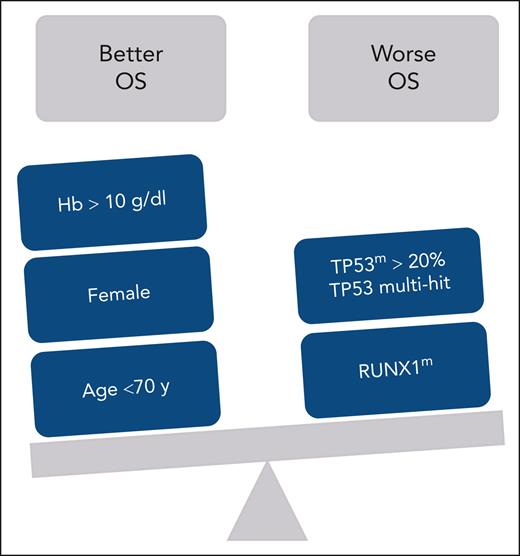
Based on a very large, retrospective international cohort of 682 patients with MDS-del(5q), with a median follow-up of >6 years, the authors have identified a TP53 mutation in 18.9% of cases, confirming exactly the data of Jädersten et al.3 However, looking at whether the mutations were monoallelic or multihit, it appears that the vast majority were monoallelic (76% of patients with TP53 mutation) and that only 4.5% of the whole cohort of patients with the MDS-del(5q) had TP53 multihit mutations, which impacted the risk of acute myeloid leukemia (AML) evolution, but, surprisingly, not survival. Given the low incidence of multihit mutations, the authors looked for an alternative threshold and identified that patients with the TP53-monoallelic mutation with variant allele frequency (VAF) >20% had a risk of death and AML evolution comparable with that seen with TP53-multihit MDS-del(5q). Thus, patients with the TP53-monoallelic mutation with VAF >20% had a 32.2% risk of AML evolution, similar to the 40.4% observed among patients with multihit mutations. It is also of interest that survival was also influenced (see figure), in the multivariable analysis, by the RUNX1 mutation, and that the risk of AML evolution was genetically impacted not only by the presence of TP53m with VAF >20% or multihit mutation but also by SF3B1 mutation, confirming previous work.7
Factors influencing OS in patients with myelodysplastic syndromes with the 5q deletion. Hb, hemoglobin; OS, overall survival.
Factors influencing OS in patients with myelodysplastic syndromes with the 5q deletion. Hb, hemoglobin; OS, overall survival.
A few questions remain, including several that are important for treatment recommendations. It will probably be highly useful to know, from sequential data, the rate of growth of TP53 clones and the clinical, biological, or therapeutic factors likely to influence the rate of growth. What is the length of time between the emergence of a small TP53 clone that has no impact on the risk of AML and its subsequent growth to a threshold that predicts transformation into acute leukemia? At what point is it reasonable to offer stem cell transplantation, neither too early to avoid the toxicity of the procedure nor too late after transformation? Another remaining task is evaluation of the role of early treatment intervention with lenalidomide to potentially reduce the risk of transformation. Thus, the results of the Sintra-REV trial assessing the role of lenalidomide in non–transfusion-dependent anemia in MDS-del(5q) are highly anticipated.8 Finally, having identified a specific at-risk population is a major step forward. The 20% VAF threshold will probably deserve to be integrated into our diagnostic and prognostic classification tools for MDS-del(5q). This is currently not the case in the WHO or ICC classifications. From a therapeutic perspective, beyond stem cell transplantation for patients with MDS and at risk of death or AML evolution, we hope in the future to have specific targeted therapy for those with the TP53 mutation, so that their poor prognosis can finally be overcome. Patients with MDS-del(5q) with the TP53 mutation represent an ideal population for studying any such therapeutic intervention.
Conflict-of-interest disclosure: L.A. has served on the advisory boards of Bristol Myers Squibb (BMS), AbbVie, Takeda, Novartis, Roche, and Jazz and has received institutional research support from BMS, Novartis, and AbbVie.