Background: The current guidelines [ J Clin Oncol. 2020;38(5):496] recommend using the risk assessment model (RAM) Khorana score ≥2 [ Blood. 2008;111(10):4902-4907] for primary anticoagulant prophylaxis (PP) of venous thromboembolism (VTE) in patients treated for cancer in an outpatient setting. Recently, a novel RAM, the ONKOTEV score [ JAMA Network Open 2023;6(2):e230010] has been prospectively externally validated with good predictive performance. Like all other established RAMs, the Khorana Score and ONKOTEV models aim to assess the incidence of VTE accurately but do not integrate the expected benefits and potential harms of treatment to determine the clinical utility of the competing management alternatives. We set out to compare the clinical utility of the Khorana Score vs. the ONKOTEV predictive model to guide the PP of VTE in patients receiving anticancer treatment.
Methods: We used the ONKOTEV validation dataset (n=425). The ONKOTEV score included the Khorana Score (dichotomized as Khorana score >2 vs. ≤ 2) and three readily available clinical information (also dichotomized): history of previous VTE, the presence of metastatic disease, and clinical or radiological assessment of vascular or lymphatic compression by tumor. After assessing the models' performance (discrimination, calibration), we applied a generalized decision curve analysis (gDCA) to integrate the effects of treatments with the models' predictive accuracy. This, in turn, allowed us to develop individualized recommendations for using PP for VTE in patients receiving anticancer treatment. We calculated net benefits (NB) to compare three management strategies: don't provide any treatment vs. administer prophylaxis to all patients receiving anticancer treatment for VTE vs. use the Khorana or ONKOTEV model to guide PP of VTE. According to gDCA, the best treatment is the one with the highest NB. We obtained data on treatment effects from the meta-analysis of published randomized trials comparing direct oral anticoagulants (DOAC) and low-molecular-weight heparins (LMWH) vs. placebo [ Annals of Palliative Medicine. 2020;9(5):2970]. We estimated that DOAC reduced the risk of VTE by 57% [relative risk reduction (RRR) =0.57 (95%CI: 0.13 to 0.79)] and LMWH by 48% [RRR=0.48 (95%CI: 0.42 to 0.66)]. We assessed the major bleeding rate on DOAC as 0.01 (95%CI: 0.006 to 0.04) and 0.02 (95%CI: 0.01 to 0.03) for LMWH.
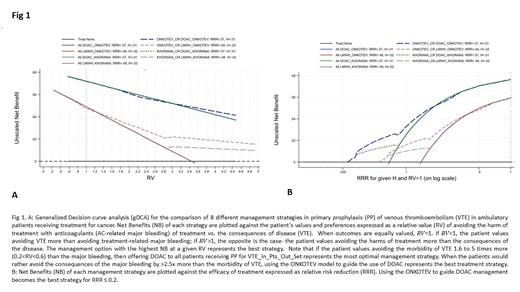
Results: Both RAMs had a satisfactory performance. Harell's C discrimination statistics was 0.71 for ONKOTEV and 0.66 for Khorana Score with good calibration properties (with intercept and the slope not statistically significantly different from 0 and 1, respectively). Fig 1A shows the results based on the default assumptions: if the patient values avoiding the morbidity of VTE 1.6 to 5 times more (0.2<RV<0.6) than the major bleeding, then offering DOAC to all patients receiving PP for VTE represents the most optimal management strategy. When the patients would rather avoid the consequences of the major bleeding by >2.5x more than the morbidity of VTE, using the ONKOTEV model to guide the administration of DOAC represents the best treatment strategy. If we assume that RRR ≤ 0.2, relying on the ONKOTEV model to guide the use of DOAC becomes the most optimal approach to PP of VTE in patients receiving anticancer treatment (Fig 1B).
Conclusion: ONKOTEV model is superior to Khorana Score in guiding individualized PP for VTE in ambulatory patients with cancer. It is particularly helpful when the patient values avoiding major bleeding more than the morbidity of VTE. Our analysis provides new recommendations for PP of VTE receiving outpatient treatment for cancer, calling for updating the current guidelines for PP of VTE in this setting.
Disclosures
Cella:IPSEN: Research Funding; Leo Pharma: Honoraria; BMS: Consultancy. Fazio:Hutchinson MediPharma: Honoraria; Merck: Honoraria; Beigene: Other: Financial Institutional interest; FIBROGEN: Other: Financial Institutional interest; IPSEN: Research Funding; Incyte: Other: Financial Institutional interest; 4SC: Other: Financial Institutional interest; AAA: Honoraria; NUCANA: Other: Financial Institutional interest; MSD: Other: Financial Institutional interest. Lordick:Amgen: Honoraria; Astellas Pharma: Honoraria; AstraZeneca: Honoraria; Bayer: Honoraria; Biontech: Honoraria; Daiichi Sankyo: Honoraria; Eli Lilly: Honoraria; Elsevier: Honoraria; Imedex: Honoraria; Incyte: Honoraria; Iomedico: Honoraria; Medscape: Honoraria; MedUpdate: Honoraria; Merck Serono: Honoraria; Merck Sharp & Dohme: Honoraria; Roche: Honoraria; Servier: Honoraria; Springer Nature: Honoraria; StreamedUp!: Honoraria; BMS: Other: Institutional, Research Funding.