Introduction
During the management of hemoglobinopathies, including sickle cell disease (SCD) and thalassemia, red blood cell (RBC) transfusions are crucial in treating symptomatic anemia. Repeated transfusions can lead to iron accumulation in soft tissues, causing inflammation, susceptibility to infections, and damage to vital organs. Currently treatment of iron overload with iron chelation therapy, does not always guarantee optimal and lasting responses.
Erythrocytapheresis, an automated method for RBC exchange, has shown promising results in reducing iron overload in patients with SCD and adult patients with β-thalassemia. Studies show limited use in children and young adults, due to optimal flow and venous access problems.
At the Antwerp University Hospital, erythrocytapheresis has been adopted as the standard of care in managing patients with both SCD and β-thalassemia major who develop iron overload, with the goals of reducing ferritin levels and preventing permanent complications. This study aimed to evaluate the efficacy and cost-effectiveness ratio associated with the inclusion of erythrocytapheresis in the overall management of these patients.
Methods
This retrospective study analyzed patients diagnosed with SCD or β-thalassemia major between 2015 and 2022, who were treated for transfusion related iron overload with iron chelation therapy with serum ferritin and hemoglobin (Hb) levels as indicators.
The case cohort were treated with erythrocytapheresis alone or in combination with iron chelation therapy, whilst the control cohort was treated with oral iron chelation therapy alone.
Long-term venous access was established in all patients utilizing one of two methods: 1) two high pressure intravenous 14-gauge catheters (Port-a-Cath®), one placed in the subclavian vein, and one placed in the inguinal vein 2) or a surgical arteriovenous fistula created in the cubital fossa. Erythrocytapheresis was performed with the Spectra Optia® system (Caridian BCT, Inc)
Data to evaluate erythrocytapheresis therapy outcomes were collected at five different time points, until one year post commencing erythrocytapheresis therapy.
Results
Of the 19 patients, 11 (57,9%) were males with a M:F ratio of 1:0.7. The median age was 13 years (range 1 to 27 years). The cohort consisted of 10 (52,6%) patients with SCD, eight (42,1%) with β-thalassemia major and one (5,3%) with sickle-thalassemia.
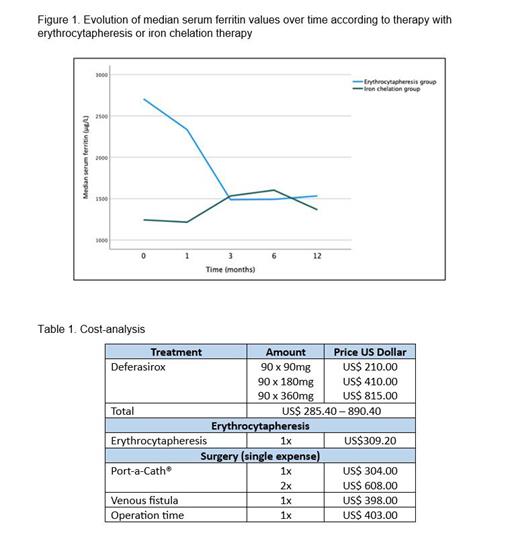
The cohort treated with erythrocytapheresis showed a significant decrease in serum ferritin ( p<0.001) whilst maintain steady Hb levels ( p<0.001). The cohort treated with oral iron chelation therapy alone, no significant decrease in serum ferritin was found ( p=0.156) but a decrease in Hb levels over time ( p=0.07). Comparing the evolution of median serum ferritin between therapy with erythrocytapheresis and iron chelation therapy showed a statistically significant difference ( p=0.008) ( Fig. 1). Patients with β-thalassemia major receiving erythrocytapheresis showed a greater reduction in serum ferritin compared to patients without ( p=0.036). A difference could not be shown between the erythrocytapheresis and iron chelation single therapies (p=0.100).
Optimal continuous flow was achieved with two small Port-a-Cath® systems. A cost benefit analysis showed that the cost of a single erythrocytapheresis is comparable to the lowest dosage of Deferasirox ( Table 1). Erythrocytapheresis could be a financially viable treatment method for children and young adults independent of the resource setting.
Conclusion
Currently iron chelation therapy is not always sufficient in patients with hemoglobinopathies treated for iron overload. Patients treated with erythrocytapheresis showed more stable and higher hemoglobin levels and a more significant decline in serum ferritin levels compared to iron chelation therapy alone. This study showed that erythrocytapheresis can also be used in children with β-thalassemia major. Further studies are needed to optimize erythrocytapheresis in children and extrapolate these findings to a larger population.
Disclosures
No relevant conflicts of interest to declare.