Introduction:
Dasatinib plus prednisone is a standard induction approach for adults with Philadelphia Chromosome positive (Ph+) ALL known to result in high remission rates with minimal toxicity, however optimal post-remission therapy has not been determined. Given the excellent outcomes observed in recent clinical studies using ponatinib in adult Ph+ ALL, we explored a strategy of switching the TKI to ponatinib and consolidating with sequential blinatumomab (blina) and chemotherapy after dasatinib + prednisone induction. In contrast to recent studies of blina + TKI wherein patients have received up to 3-5 cycles of blina, we limited the number of blina cycles to 2 to minimize cost and logistical burden. We also administered systemic chemotherapy after blina with the goal of decreasing risk of extramedullary relapse, which has been observed in studies of blina-based “chemotherapy-free” regimens.
Methods:
Since June 2021, all adult patients with Ph+ ALL (n=14) at our institution were switched to ponatinib (30mg/d then 15mg/d once complete molecular remission [CMR] achieved) after dasatinib + prednisone induction. Concurrent with TKI, patients could also receive blina for one or two cycles (at discretion of treating clinician), followed by up to four cycles of consolidative chemotherapy alternating between high-dose methotrexate (1.5g/m 2) and intermediate-dose cytarabine (1g/m 2 x 6 doses). Patients not proceeding to stem cell transplant continue ponatinib monotherapy for up to 5 years, and 12 doses of intrathecal methotrexate 15mg are given throughout the total course of treatment. Prior to June 2021, our institutional approach was to recommend allogeneic SCT in CR1 for all transplant candidates immediately after dasatinib + prednisone induction. We reviewed charts of patients with newly diagnosed Ph+ ALL at University of Colorado Hospital from 2014-2023 to compare outcomes of patients treated before 06/2021 (“dasatinib/SCT” cohort) and after 06/2021 (“ponatinib” cohort).
Results:
From 6/2021-02/2023, 14 patients (median age 54) with newly diagnosed Ph+ ALL were switched to ponatinib after dasatinib + prednisone induction. After induction, 12 patients received blina (median 1 cycle; range 1-2) and consolidation chemotherapy (median 4 cycles; range 3-4). Blina could not be obtained for 2 patients who instead received chemotherapy only for 6 cycles. 3 patients (21%) underwent allogeneic SCT in CR1 due to persistent detectable bcr/abl transcripts by qPCR, and all 3 continued ponatinib as maintenance after SCT. With a median follow-up of 15 months (range: 4-24 months) no relapses have occurred and all patients remain alive at last follow-up. No patients have discontinued ponatinib due to intolerance. Chemotherapy plus ponatinib was well-tolerated after blina, with all except for one patient having received all intended cycles (one patient stopped chemotherapy after 3 cycles due to excessive hematologic toxicity).
Between 11/2014-03/2021, 36 patients (median age 50) with Ph+ ALL were treated with dasatinib + prednisone induction, after which 31 (86%) underwent allogeneic SCT in CR1. All non-transplant candidates continued TKI alone. With a median follow-up of 37 months (range: 4-102 months), 11 relapses have occurred, 9 of which occurred after SCT. Among 13 patients who died during follow-up, 7 deaths were due to relapsed/refractory ALL and 6 were due to treatment-related mortality.
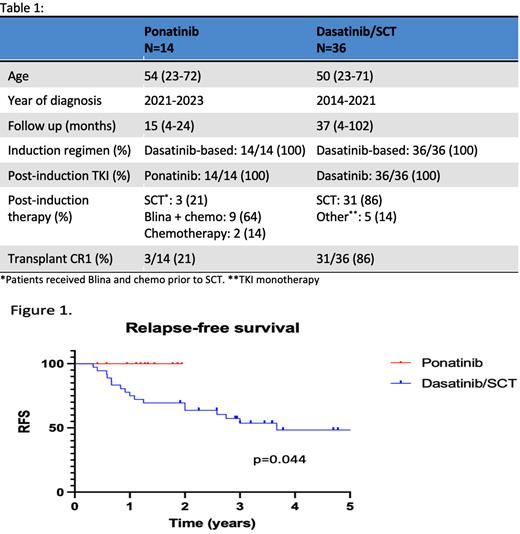
Comparing outcomes between the two cohorts, relapse-free survival is improved in the more recent (ponatinib) cohort compared to the earlier (dasatinib/SCT) cohort (p=0.044, log-rank test) (Figure 1) despite a lower rate of allogeneic SCT in CR1 (21% vs 86%). No difference in overall survival (p=0.12) has been observed (not shown).
Conclusions:
For adults with Ph+ ALL, switching to ponatinib after dasatinib-based induction and consolidating with blina and chemotherapy is a safe and potentially effective strategy. The optimal number of blina cycles given post-remission is unclear, though we observed favorable outcomes in patients receiving a median of only 1 cycle, and maximum of 2 cycles. Favorable outcomes despite the low rate of allogeneic SCT (21%) seen in our cohort suggest that SCT may be safely avoided for some patients who receive ponatinib plus blina and chemotherapy-based consolidation.
OffLabel Disclosure:
Schwartz:Jazz Pharmaceuticals: Consultancy; Novartis: Consultancy. McMahon:Kura Oncology: Membership on an entity's Board of Directors or advisory committees; Syros Pharmaceuticals: Research Funding; Syndax Pharmaceuticals: Research Funding; Arcellx: Membership on an entity's Board of Directors or advisory committees. Pollyea:Teva, Karyopharm, Bristol Myers Squibb, and AbbVie.: Research Funding; AbbVie, Bristol Myers Squibb, Syros, Novartis, Beigene, Bergen Bio, Arcellx, Jazz, Genentech, Immunogen, AstraZeneca, Kura, Ryvu, Magenta, Qihan, Zentalis, Medivir, Hibercell, LINK, Daiichi Sankyo, Aptevo, Rigel, Sumitomo, Adicet, Seres, Gilead, OncoVerit: Consultancy. Smith:OncoVerity: Current Employment, Current holder of stock options in a privately-held company; AML JV: Consultancy. Jordan:AML JV: Consultancy.
Ponatinib frontline in Ph+ ALL Blinatumomab consolidation in Ph+ ALL