Background
Augustson et al (2005) showed that myeloma patients diagnosed and managed in the early‐mid 2000s are at a higher risk of infections compared to those diagnosed prior to this period. This has been attributed, in part, to a changing treatment paradigm, including the use of proteasome inhibitors (PI) and immunomodulatory drugs (IMiDs) for longer periods which confer different infection risks. Furthermore, infection related outcomes are poorly reported in elderly co-morbid patients due to lack of representation in clinical trials. Assessment of infection related morbidity and mortality in transplant ineligible NDMM patients during routine care requires evaluation, so future strategies can be devised to limit these complications.
Methods
We performed a single institution retrospective study of 200 transplant ineligible newly diagnosed myeloma patients treated with UK standard of care (2009-2018) to assess infection related outcomes over a 12 month period from treatment initiation.
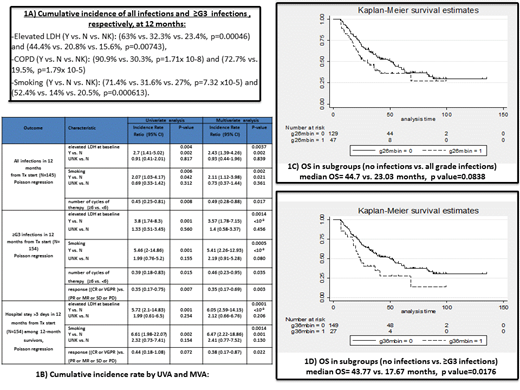
Cumulative incidence of infections at 12 months were compared between the following subgroups using Gray test : age (<75 vs. ≥75), sex (M vs. F), immunoparesis at baseline (N vs. Y), elevated LDH (Y vs. N), ISS (3 vs. <3), Charlson co-morbidity index (CCI) (0-2 vs. 3-4 vs. ≥5), diabetes (Y vs. N), COPD (Y vs. N), renal impairment (Y vs. N), choice of therapy (IMiD vs. PI vs. Chemo), combination (doublet vs. triplet), dose attenuation (Y vs. N), PCP prophylaxis (N vs. Y) and antifungal prophylaxis (N vs. Y).
Using Poisson regression, univariate (UVA) and multivariate analyses (MVA) were conducted to assess factors associated with increased 12 month incidence rates of: all infections, ≥G3 infections, as well as significant inpatient admission (>3 days).
A landmark analysis was conducted to compare overall survival of patients who experienced infections in the first 6 months versus infection-free patients.
Results
The median age of total cohort was 75 years (range 40-94), of whom 54% had IgG subtype, and 44% had ISS 3 staging. CCI categories were: 0-2 (20.5%), 3-4 (48%) and ≥5 (28%). Patients received a median of 6 cycles of therapy (range 1-39): IMiD-based (69%), PI-based (20%) and chemo-based (11%). Median follow up was 67.7 months. Median OS and PFS were 33.5 and 9.2 months, respectively.
There were 116 documented infections, of which 72 were ≥G3. Two thirds occurred in the first 6 months. Median time to first episode was 70 days (range 7-343) for all infections and 70 days (range 7-338) for ≥G3. Cumulative incidence of infections in the total cohort for all infections and ≥G3 infections were 33% (95% CI 26.6-39.6%) and 22% (95% CI 16.5-28%), respectively. Median number of all infections and ≥G3 infections per patient was: 1 (range 1-7) and 1 (range 1-5), respectively. Episodes occurred during induction (49.1%), remission (17.2%), progressive disease (13.8%) and during 2nd line therapy (19.8%). Thirty seven episodes occurred in winter seasonal period (Dec-Mar). Most common infection sites were respiratory tract (n=61) and genitourinary tract (n=22). Fifty six episodes required a significant inpatient stay (>3 days), of which 2 required ICU admissions. Cumulative 12-month infection-mortality was 4% (95% CI 0.019-0.074).
Baseline characteristics did not account for a significant difference in cumulative incidence of all infections and ≥G3 infections except for: elevated LDH, COPD and smoking status (Figure 1A).
By MVA, elevated LDH, smoking and number of cycles of therapy (≥6 vs. <6) were independent factors associated with increased rate of all grade infections. Elevated LDH, smoking, number of cycles and response ((CR or VGPR) vs. (PR or MR or SD or PD) were independent factors associated with increased rate of ≥G3 infections. Elevated LDH, smoking and response were independent factors associated with significant infection-related inpatient stay (Figure 1B).
Median OS was lower in the subgroup of patients who experienced infections in the first 6 months compared to those who did not, for both all grade infections (borderline statistical significance) and ≥G3 infections (statistical significance), (Figures 1C and 1D).
Conclusions
This study demonstrates significant infection related morbidity and hospital admissions within a year of treatment initiation in an elderly routine care cohort. Whilst this study identified independent predictors of infections, strategies should be put in place to modify these risk factors
Djebbari:Novartis: Honoraria; Celgene: Honoraria, Other: support with conference attendance, Research Funding; Takeda: Honoraria, Other: support with conference attendance; Amgen: Honoraria. Ramasamy:Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Oncopeptides: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Research Grants ; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Research Grants ; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Research Grants ; Abbvie: Honoraria, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.