Introduction: Methotrexate (MTX) is a weak acid that is approximately 50% bound to serum albumin. Although data are limited, recent publications have shown higher MTX concentrations in patients with low albumin levels. It has been proposed that hypoalbuminemia alters MTX clearance through two distinct mechanisms: 1) increased formation of intracellular MTX polyglutamates, and 2) development of third-space fluid collections in the absence of appropriate colloidal osmotic pressure. The purpose of our study was to evaluate the effect of albumin on MTX clearance in cancer patients.
Methods: This single center, retrospective analysis included hospitalized adult and pediatric patients receiving high-dose MTX therapy (≥ 1 g/m2). The study was approved by the local IRB. Adult patients' records were reviewed from January 2015 to December 2018, and pediatric patients' records were reviewed from August 2016 to December 2018. Patients were included in the study more than once if they had a repeat but separate hospital encounter. The primary outcome measure was the average time to MTX clearance in patients with normal (3.5 g/dL to 5 g/dL) versus low (< 3.5 g/dL) serum albumin levels. MTX clearance was defined as the first recorded time that the level was ≤ 0.1 μM/L. Secondary outcomes included MTX levels at 24, 48, and 72 hours; rate of delayed clearance; rate of toxicities; hospital length of stay; and other factors that contribute to delayed clearance. Group comparisons for continuous data were made using Student's t-test or Mann-Whitney U test. Chi-square or Fisher's exact test was used for categorical data. To evaluate the relationship between time to MTX clearance and toxicities, Cox proportional hazards models were used. Analyses were conducted separately for pediatric and adult patients. P values < 0.05 were considered statistically significant.
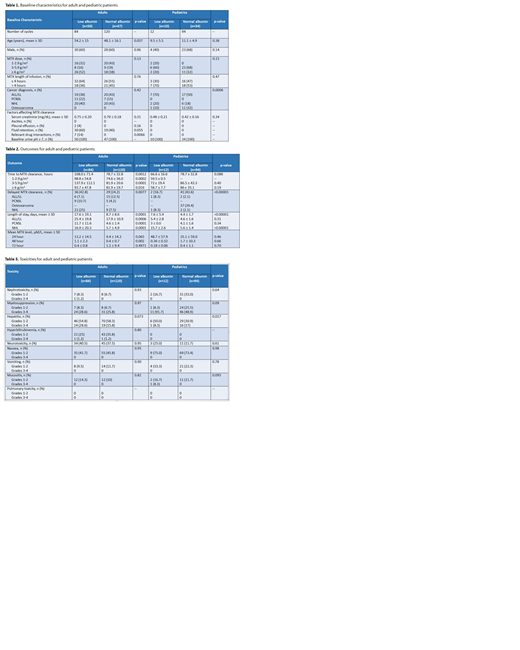
Results: Of 510 encounters initially screened, 204 adult patient encounters and 106 pediatric patient encounters were included. Among the adults and pediatric patients, 52% and 23% had hypoalbuminemia, respectively. In both cohorts, baseline characteristics were largely similar between the groups (Table 1). The most common cancer diagnosis among adults was lymphoma (60%). High-dose MTX ± rituximab (58%) and HyperCVAD (39%) were the most common protocols used in the adult population. The majority of pediatric patients had leukemia (55%). Results are shown in Table 2.
Adult outcomes: MTX clearance was prolonged in patients with hypoalbuminemia when compared to those with normal albumin levels (108 hours vs 78.7 hours; p=0.0012) with a significantly higher percentage of the former group experiencing delayed clearance (42.8% vs 24.2%; p=0.00077). Mean length of stay was longer among hypoalbuminemic patients (17.6 days vs 8.7 days; p=0.0001). There were no statistically significant differences in rate of toxicities (Table 3). However, there was a trend towards an increased rate of grades 3-4 hepatitis among patients with low albumin levels (28.6% vs 15.8%; p=0.073). Further analysis utilizing a Cox proportional hazards model revealed that a shorter infusion time (HR 0.4, 95% CI 0.22-0.75) had a protective effect on MTX clearance. Conversely, MTX doses of 1-2.9 g/m2 (HR 3.26, 95% CI 1.52-6.97) were associated with delayed excretion of MTX. Age (HR 0.94, 95% CI 0.83-1.06), hypoalbuminemia (HR 0.94, 95% CI 0.49-1.79) and a MTX dose of 3-5.9 g/m2 (HR 1.57, 95% CI 0.64-3.88) were not significant predictors of delayed clearance.
Pediatric outcomes: There was no statistically significant difference between the two groups with regard to mean time to MTX clearance. Overall mean length of stay was 7.6 days in the low albumin group and 4.4 days in the normal albumin group (p<0.00001). There were no differences in mean MTX levels at 24, 48, or 72 hours. As seen in Table 3, hypoalbuminemic patients had an increased rate of hepatitis (58.3% vs 47.9%; p= 0.017). Using the Cox analysis, the most significant predictors of delayed MTX clearance were serum creatinine and age.
Conclusion: Adult patients with hypoalbuminemia had prolonged MTX excretion but this observation did not translate into an increased rate of toxicities. No difference in clearance time was detected between the two pediatric groups. To our knowledge, this is the largest study to demonstrate that high-dose MTX in combination with aggressive supportive care can be safely administered to patients with low albumin levels.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.