Abstract
Background - In many adult protocols for acute myeloid leukemia (AML), the standard of care is to perform a lumbar puncture (LP) at the time of diagnosis only if there are central nervous system (CNS) symptoms present, and if absent, the LP is performed after a period of systemic therapy in order to avoid contamination of the cerebrospinal fluid (CSF) with circulating blasts in the case of a traumatic LP, or is not done at all. This differs from the practice in pediatric AML protocols where LP is performed as part of the diagnostic workup and intrathecal (IT) cytarabine given at the time of the diagnostic LP, as well as during therapy, similar to the pediatric ALL approach. We sought to determine the effect of an initial traumatic LP in a large cohort of pediatric patients with de novo AML.
Methods - Treatment protocols COG AAML03P1 and AAML0531 enrolled 1344 patients with diagnostic LP and outcome data available. In these protocols, patients identified to be CNS2 (0-5 WBC with blasts present) or CNS3 (>5 WBC with blasts or CNS symptoms at diagnosis) prior to systemic therapy received additional IT therapy, while CNS1 (no blasts) patients received IT therapy only at the beginning of most courses of chemotherapy. Patients were analyzed for CNS status as well as diagnostic LP red cell count. A diagnostic LP with greater than 100 red blood cells was considered traumatic. The effect of traumatic LP on outcome was analyzed.
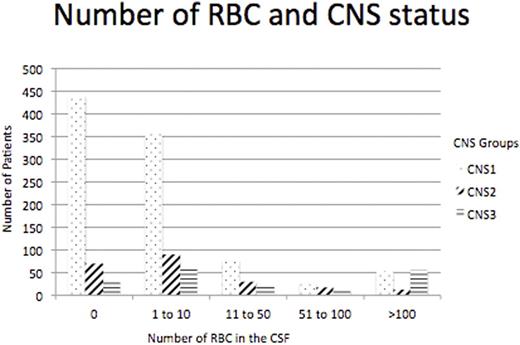
Results - Among these patients, 949 were CNS1, 217 were CNS2 and 178 were CNS3. In looking at percentage of patients with more than 100 RBC in the CSF, there were 52 (5.48%) CNS1, 12 (5.53%) CNS2 and 55 (30.9%) CNS3 (three group comparison, p<0.001). As well, in patients with 6 to 100 RBC in the CSF in each CNS group there was a significant difference with 150 (15.81%) CNS1, 64 (29.49%) CNS2 and 45 (25.28%) CNS3 patients, (p<0.001). Figure 1 shows the number of patients in each CNS group with number of RBC in the diagnostic LP. The number of RBC in the CSF did not correlate with the degree of peripheral blood hyperleukocytosis.
The outcomes of patients with traumatic initial LP showed that patients who had a traumatic initial LP did not have a significantly different OS (HR 1.14, p=0.405) or EFS (HR 1.22, p=0.132) from study entry, nor RR (HR 1.06, p=0.769) or TRM (HR 1.30, p=0.510) compared to those with an atraumatic LP. This was similarly seen in the subgroups, CNS1, CNS2, and CNS3. In examining patients with 0 RBC in their initial CSF, OS was similar among the CNS1, CNS2 and CNS3 patients (62.6%, 62.7% and 66.4% respectively, p=0.974). The OS was also similar for patients with <100 RBC (64.5% ± 2.8%) and >100 RBC (63.0% ±9.1%). In examining outcomes of CNS3 patients with a traumatic tap, there were no significant differences in OS, EFS, or RR among those with traumatic tap and those without. Overall though, multivariable analyses showed that CNS3 patients had significantly worse EFS from study entry compared to CNS1 and CNS2 patients (HR 1.58, p<0.001) due to a higher RR (HR 1.64, p=0.003). The CNS3 patients with traumatic LP had similar OS and EFS compared to those without traumatic LP (OS 63.5% ± 15.2% vs 60.8% ± 11.4% respectively, and EFS 43.4% ± 15.3% vs 35.5% ± 10.9% respectively).
Conclusion - There were significantly more CNS3 patients with >100 RBC in the CSF than in the other 2 CNS groups. Outcome for these patients with traumatic LP vs those with atraumatic LP however were not different for EFS, OS or RR for all 3 CNS groups. It is likely that more than half of the CNS3 patients received extra IT therapy due to traumatic LP. In the majority of cases these IT therapies were given with sedation, potentially unnecessarily. The concern though that a traumatic initial LP contributes to worse outcome in CNS3 patients is not warranted. Delaying initial LP to align with adult practice, when peripheral blasts are cleared, would not change prognosis, but may lessen the number of patients who need additional IT therapy from traumatic LPs, thus sparing a significant proportion of children unnecessary procedures and CNS directed chemotherapy.
Number of Red Blood Cells in the CSF, CNS Status and Number of Patients in Each Grouping
Number of Red Blood Cells in the CSF, CNS Status and Number of Patients in Each Grouping
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.