Abstract
Background: Ibrutinib (ibr), a first-in-class, once-daily Bruton's tyrosine kinase inhibitor, is approved by the US FDA for treatment of patients (pts) with chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) including pts with del17p. The phase 1b/2 PCYC-1102 trial showed single-agent efficacy and tolerability in treatment-naïve (TN; O'Brien, Lancet Oncol 2014) and relapsed/refractory (R/R) CLL/SLL (Byrd, N Engl J Med 2013). We report efficacy and safety results of the longest follow-up to date for ibr-treated pts.
Methods: Pts received 420 or 840 mg ibr QD until disease progression (PD) or unacceptable toxicity. Overall response rate (ORR) including partial response (PR) with lymphocytosis (PR-L) was assessed using updated iwCLL criteria. Responses were assessed by risk groups: unmutated IGVH, complex karyotype (CK; ≥3 unrelated chromosomal abnormalities by stimulated cytogenetics assessed by a reference lab), and in hierarchical order for del17p, then del11q. In the long-term extension study PCYC-1103, grade ≥3 adverse events (AEs), serious AEs, and AEs requiring dose reduction or discontinuation were collected.
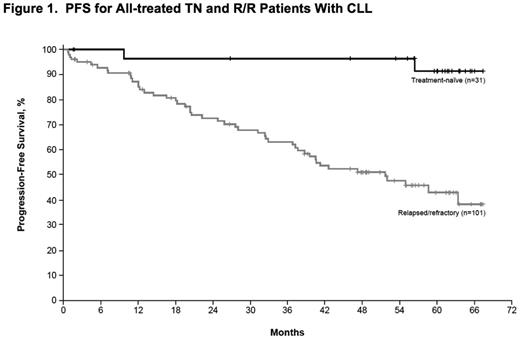
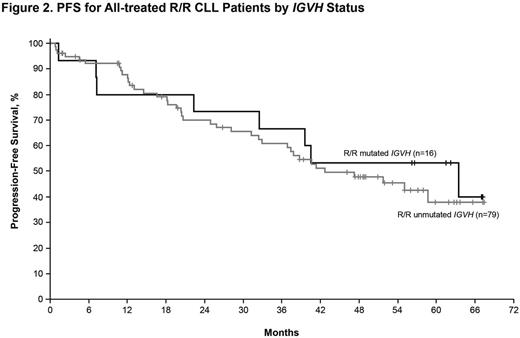
Results: Median age of the 132 pts with CLL/SLL (31 TN, 101 R/R) was 68 y (range, 37-84) with 43% ≥70 y. Baseline CK was observed in 41/112 (37%) of pts. Among R/R pts, 34 (34%) had del17p, 35 (35%) del11q, and 79 (78%) unmutated IGVH. R/R pts had a median of 4 prior therapies (range, 1-12). Median time on study was 46 m (range, 0-67) for all-treated pts, 60 m (range, 0-67.4) for TN pts, and 39 m (range, 0-67) for R/R pts. The ORR (per investigator) was 86% (complete response [CR], 14%) for all-treated pts (TN: 84% [CR, 29%], R/R: 86% [CR, 10%]). Median progression-free survival (PFS) was not reached (NR) for TN and 52 m for R/R pts with 60 m estimated PFS rates of 92% and 43%, respectively (Figure 1). In R/R pts, median PFS was 55 m (95% confidence intervals [CI], 31-not estimable [NE]) for pts with del11q, 26 m (95% CI,18-37) for pts with del17p, and NR (95% CI, 40-NE) for pts without del17p, del11q, trisomy 12, or del13q. Median PFS was 33 m (95% CI, 22-NE) and NR for pts with and without CK, and 43 m (95% CI, 32-NE) and 63 m (95% CI, 7-NE) for pts with unmutated and mutated IGVH, respectively(Figure 2). Among R/R pts, median PFS was 63 m (95% CI, 37-NE) for pts with 1-2 prior regimens (n=27, 3 pts with 1 prior therapy) and 59 m (95% CI, 22-NE) and 39 m (95% CI, 26-NE) for pts with 3 and ≥4 prior regimens, respectively. Median duration of response was NR for TN pts and 45 m for R/R pts. Pts estimated to be alive at 60 m were: TN, 92%; all R/R, 57%; R/R del17p, 32%; R/R del 11q, 61%; R/R unmutated IGVH, 55%. Among all treated pts, onset of grade ≥3 treatment-emergent AEs was highest in the first year and decreased during subsequent years. With about 5 years of follow-up, the most frequent grade ≥3 AEs were hypertension (26%), pneumonia (22%), neutropenia (17%), and atrial fibrillation (9%). Study treatment was discontinued due to AEs in 27 pts (20%) and disease progression in 34 pts (26%). Of all treated pts, 38% remain on ibr treatment on study including 65% of TN pts and 30% of R/R pts.
Conclusions: Single-agent ibrutinib continues to show durable responses in pts with TN or R/R CLL/SLL including those with del17p, del11q, or unmutated IGVH. With extended treatment, CRs were observed in 29% of TN and 10% of R/R pts, having evolved over time. Ibrutinib provided better PFS outcomes if administered earlier in therapy than in the third-line or beyond. Those without CK experienced more favorable PFS and OS than those with CK. Ibrutinib was well tolerated with the onset of AEs decreasing over time, allowing for extended dosing for 65% of TN and 30% of R/R pts who continue treatment.
O'Brien:Janssen: Consultancy, Honoraria; Pharmacyclics, LLC, an AbbVie Company: Consultancy, Honoraria, Research Funding. Furman:Pharmacyclics, LLC, an AbbVie Company: Consultancy, Honoraria, Speakers Bureau. Coutre:Janssen: Consultancy, Research Funding; Pharmacyclics, LLC, an AbbVie Company: Consultancy, Research Funding; AbbVie: Research Funding. Flinn:Janssen: Research Funding; Pharmacyclics LLC, an AbbVie Company: Research Funding; Gilead Sciences: Research Funding; ARIAD: Research Funding; RainTree Oncology Services: Equity Ownership. Burger:Pharmacyclics, LLC, an AbbVie Company: Research Funding; Gilead: Research Funding; Portola: Consultancy; Janssen: Consultancy, Other: Travel, Accommodations, Expenses; Roche: Other: Travel, Accommodations, Expenses. Sharman:Gilead: Research Funding; TG Therapeutics: Research Funding; Acerta: Research Funding; Seattle Genetics: Research Funding; Pharmacyclics: Research Funding; Celgene: Research Funding. Wierda:Abbvie: Research Funding; Genentech: Research Funding; Novartis: Research Funding; Acerta: Research Funding; Gilead: Research Funding. Jones:Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pharmacyclics, LLC, an AbbVie Company: Membership on an entity's Board of Directors or advisory committees, Research Funding. Luan:AbbVie: Equity Ownership; Pharmacyclics, LLC, an AbbVie Company: Employment, Other: Travel, Accommodations, Expenses. James:AbbVie: Equity Ownership; Pharmacyclics, LLC, an AbbVie Company: Employment. Chu:Pharmacyclics, LLC, an AbbVie Company: Employment; AbbVie: Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract