In this issue of Blood, Nguyen et al have identified a novel mechanism to control ribosomal RNA (rRNA) synthesis.1
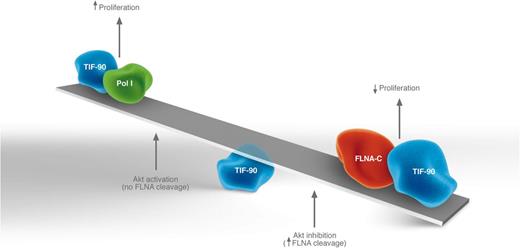
The relative amount of active TIF-90 complexes correlates with rRNA synthesis and thus proliferation. Akt activity affects the balance of complexes by modulating FLNA cleavage as described in the text. Given that no assays with purified protein have been carried out, it is unknown whether TIF-90 directly binds to either Pol I or FLNA-C, thus the illustration is not meant to imply direct interactions. Professional illustration by Luk Cox, Somersault1824.
The relative amount of active TIF-90 complexes correlates with rRNA synthesis and thus proliferation. Akt activity affects the balance of complexes by modulating FLNA cleavage as described in the text. Given that no assays with purified protein have been carried out, it is unknown whether TIF-90 directly binds to either Pol I or FLNA-C, thus the illustration is not meant to imply direct interactions. Professional illustration by Luk Cox, Somersault1824.
Inappropriate elevation of rRNA synthesis is typically associated with a broad variety of malignancies because it leads to increased ribosomal biogenesis, increased translation, and, thus, increased proliferation.2,3 Understanding new mechanistic aspects of this process is critical and could provide novel therapeutic strategies. The transcription initiation factor 1A (TIF-1A) plays a central role in the recruitment of Polymerase I (Pol I) to the rDNA promoter and thus is critical for rRNA synthesis.4 In Nguyen et al, the authors have discovered a novel splice variant of TIF-1A, TIF-90, and outline new modes of regulation.1 These elegant studies lead to novel insights into the control of ribosomal biogenesis, how it is connected to cellular signaling, and how these processes can be dysregulated in hematologic malignancy, specifically acute myeloid leukemia (AML).
The novel TIF-90 splice variant identified here has a 30 amino acid in-frame deletion relative to TIF-1A.1 The resulting TIF-90 protein is more strongly localized to the site of rRNA synthesis, the nucleolus. Consistently, it more strongly associates with Pol I and better promotes rRNA synthesis than TIF-1A. Consistent with the link between elevated rRNA synthesis and malignancy, TIF-90 and TIF-1A are highly expressed in primary AML specimens relative to normal bone marrow.
Previous studies had demonstrated that the actin-binding protein filamin A (FLNA) impaired rDNA transcription by inhibiting recruitment of Pol I machinery, including TIF-1A, to the rDNA promoter.5 Given these previous findings, Nguyen et al investigated whether FLNA could play a role in the control of TIF-90 activity. They found that a cleavage product of FLNA (FLNA-C) binds to TIF-90, apparently sequestering it from Pol I. This leads to reduced rRNA synthesis. These data suggest a model whereby the balance of rRNA synthesis is modulated by the competition of FLNA-C and Pol I for TIF-90 (see figure). More TIF-90–Pol I complexes mean more rRNA synthesis whereas more TIF-90–FLNA-C complexes mean a reduction in this.
The role of TIF-1A in rRNA synthesis is well established and is traditionally tightly linked to the phosphoinositide 3-kinase/Akt/mammalian target of rapamycin (mTOR) pathway with a particular emphasis on mTOR.2 For instance, mTOR through its effects on S6 kinase indirectly enhances Pol I activity through phosphorylation of its cofactors.2 Independently of mTOR, Akt cooperates with Myc to activate Pol I activity6 and, furthermore, Akt modulates casein kinase II phosphorylation leading to increased TIF-1A phosphorylation and elevated rRNA synthesis.7 Indeed, the mTOR inhibitor rapamycin does not inhibit rRNA synthesis to the same extent as Akt inhibition.6 Thus, Akt affects this process at multiple levels and independently of mTOR.
Given these findings, the authors examined the effects of Akt on TIF-90 activity. Interestingly, they found that Akt affects FLNA cleavage and thus the balance between active and inactive TIF-90 complexes (see figure). Specifically, Akt activation impaired FLNA cleavage allowing for more TIF-90–Pol I complexes to form whereas pharmacologic inhibition of Akt promoted FLNA cleavage leading to more TIF-90–FLNA-C complexes. Thus, the activation state of Akt, and not mTOR, modulated the balance between active and inactive TIF-90 complexes.
What are the implications for AML and other hematologic malignancies? Often there is a concern that targeting central processes such as ribosomal biogenesis or translation will cause overt toxicity. However, this does not seem to be the case, most likely because cancer cells are more sensitive to inhibition of these processes than normal cells due to oncogene addiction. Indeed, targeting of the translation factor eIF4E in refractory and relapsed AML led to clinical responses including remissions with minimal therapy-associated toxicities.8 rRNA synthesis itself may be targetable. For example, Pol I inhibitors are under development such as CX-5461.2 Indeed, many standard chemotherapies such as doxorubicin are known to inadvertently affect Pol I activity.2 The studies by Nguyen et al suggest that Akt inhibitors could be important tools for targeting elevated rRNA synthesis in AML. Clearly, Akt inhibitors would have pleiotropic effects, but perhaps it is the combination of these effects that will prove to be more efficacious than targeting any single event.
Many exciting questions remain to be answered. For instance, are the outcomes for AML patients with higher levels of TIF-90 worse than others? Similarly, for those patients with uncleaved vs cleaved FLNA? What are the mechanisms underpinning the alternative splicing leading to TIF-90 generation? How are these mechanisms dysregulated in AML? Can these be modified to reduce levels of TIF-90? Future studies will undoubtedly lead the way to understanding these aspects of rRNA synthesis and better reveal the intricate control of this critical cellular process.
Conflict-of-interest disclosure: The author declares no competing financial interests.