Abstract
Abstract 1952
Graft versus host disease (GVHD) remains a major cause of morbidity and mortality in allogeneic hematopoietic cell transplantation (HCT) despite current prophylaxis strategies. Methotrexate (MTX) in combination with a calcineurin inhibitor is the current standard, however can lead to significant toxicity. Mycophenolate mofetil (MMF) is successfully combined with a calcineurin inhibitor in reduced intensity HCT, but data on its use in myeloablative transplants is limited. In 2004 we reported a prospective study evaluating the use of cyclosporine (CSA) and MMF versus CSA and short course MTX following myeloablative transplantation from matched sibling donors. We found that patients in the MMF group had less mucositis and faster hematopoietic recovery, however were not able to detect significant differences in relapse or survival. These observations prompted us to look at our larger experience with MMF based regimens in myeloablative HCT.
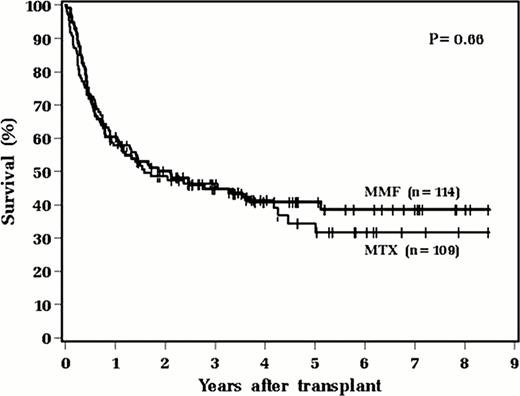
From May 2003 until April 2011, we retrospectively identified 223 adult patients who underwent myeloablative allogeneic HCT from an 8/8 HLA matched unrelated (MUD) or sibling donor (MSD). One hundred fourteen patients received a MMF-based GVHD regimen for HCT from a MSD and 109 patients received MTX-based prevention for transplantation from a MUD. The two groups were similar in patient characteristics such as age, gender, disease, disease status, and CMV status but differed in time to transplant and preparative regimen primarily due to the differences of donor type between the two groups. In regards to preparative regimen, 94 (82.5%) patients in the MMF group received cyclosphosphamide in combination with busulfan (BuCy) compared with 73 (67.0%) patients in the MTX group, (p<0.001). Other preparative regimens included Cy in combination with total body irradiation (TBI), (n=14, 12.8% in the MTX group compared with n=1, 0.9% in the MMF group); and TBI with etoposide (VP-16), (n=22, 20.2% in the MTX group versus n=19, 16.7% in the MMF group). MMF was given at 500mg three times daily, however MTX dosing changed in mid-2009 from 5mg/m2 (days 1, 3, 6, 11) to 15mg/m2 day 1, followed by 10mg/m2 days 3, 6, 11. Baseline variables were compared between MMF and MTX using the Wilcoxon rank sum test or Chi square test and outcomes were compared using the log-rank test or Pepe-Mori test. Patients receiving MMF experienced less short-term toxicities, including faster time to hematopoietic recovery (median 11 days versus 17 days with MTX, p<0.001) as well as shorter hospital stay (median 26 days versus 34 days, p<0.001). Of 110 patients with recorded WHO mucositis scores, there was significantly less grade 3 and 4 mucositis with MMF compared to MTX (p=0.019). In this larger analysis, we again did not detect significant differences in incidence or degree of acute (p=0.90) or chronic GVHD (p=0.12), relapse (p=0.39), or overall survival (p=0.66) between MMF and MTX in univariate analysis. While there was a suggestion toward higher non-relapse mortality with MTX-based regimens in keeping with increased toxicity, this was not statistically significant (p=0.08).
Results from this retrospective analysis demonstrates that the use of MMF in myeloablative sibling donor transplantation results in similar outcomes with significantly less toxicity compared to MTX in unrelated donor transplantation and supports the use of MMF as a less toxic substitute for MTX in GVHD prevention for myeloablative sibling donor transplants. Given the obvious differences between sibling and unrelated donor transplants, further prospective study evaluating and comparing the use of MMF with MTX in myeloablative unrelated donor transplants are warranted.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.