Abstract
Migration of hematopoietic stem cells through the blood, across the endothelial vasculature to different organs and to their bone marrow (BM) niches, requires active navigation, a process termed homing. Homing is a rapid process and is the first and essential step in clinical stem cell transplantation. Similarly, homing is required for seeding of the fetal BM by hematopoietic progenitors during development. Homing has physiological roles in adult BM homeostasis, which are amplified during stress-induced recruitment of leukocytes from the BM reservoir and during stem cell mobilization, as part of host defense and repair. Homing is thought to be a coordinated, multistep process, which involves signaling by stromal-derived factor 1 (SDF-1) and stem cell factor (SCF), activation of lymphocyte function–associated antigen 1 (LFA-1), very late antigen 4/5 (VLA-4/5) and CD44, cytoskeleton rearrangement, membrane type 1 (MT1)–matrix metalloproteinase (MMP) activation and secretion of MMP2/9. Rolling and firm adhesion of progenitors to endothelial cells in small marrow sinusoids under blood flow is followed by trans-endothelial migration across the physical endothelium/extracellular matrix (ECM) barrier. Stem cells finalize their homing uniquely, by selective access and anchorage to their specialized niches in the extravascular space of the endosteum region and in periarterial sites. This review is focused on mechanisms and key regulators of human stem cell homing to the BM in experimental animal models and clinical transplantation protocols.
Introduction
The hallmark of hematopoietic stem cells (HSCs) is their migration and repopulation potential (self-renewal and multilineage differentiation capacities). Progenitors isolated from the murine embryo, which lack directional migration and homing in adult mice that received transplants, are nondefinitive, cannot engraft the bone marrow (BM), and fail to rescue lethally irradiated recipients. These progenitors require in vitro stimulation with stromal cells, which convert them into functional, BM-repopulating stem cells endowed with migration and homing potential.1 Interestingly, migration of mobilized human CD34+ cells toward a gradient of the chemokine stromal derived factor-1 (SDF-1) in vitro correlates with their in vivo repopulation potential in patients who received autologous transplants and in immune-deficient nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice.2-4
Most clinical and experimental stem cell transplantation protocols use intravenous (iv) injections of recipients previously conditioned with DNA-damaging agents such as total body irradiation (TBI) and/or chemotherapy. These agents induce massive cell death of cycling hematopoietic cells in the bone marrow, spleen, and blood and damage the physiological BM endothelium barrier. Tissue damage leads to a dramatic increase in the levels of secreted chemokines, cytokines, and proteolytic enzymes in many organs as part of the regeneration and repair process, which have profound impacts on stem cell migration and repopulation. DNA damage and stress also lead to proliferation and mobilization of quiescent stem cells from the BM to the circulation (reviewed in Cottler-Fox et al5 ). For example, cyclophosphamide is used clinically to induce stem cell mobilization and to destroy malignant cells prior to stem cell transplantation. Yet, DNA damage aimed at eliminating malignant cells in patients also increases secretion of survival, migration, and angiogenic factors such as SDF-1. This chemokine can increase CXC chemokine receptor 4+ (CXCR4+) malignant stem cell survival, proliferation, invasion, and metastasis, leading to the spreading of tumors in a broad range of cancers including breast, prostate, and brain tumors as well as leukemias.6-9 Interactions between stem cells and their BM microenvironment, which are broken during cell mobilization, should be re-established to enable efficient BM homing and, moreover, retention. In genetically marked parabiotic mice with a shared circulation, granulocyte–colony-stimulating factor (G-CSF)–induced mobilization also dramatically increases the levels of stem cell engraftment in the partner bone marrow.10 Thus, stem cell mobilization, homing, and repopulation are sequential events with physiological roles.
Finally, there is lack of clarity with regards to overlapping definitions of homing to the bone marrow, engraftment, and repopulation. While homing is essential for successful engraftment and repopulation, it is not sufficient. In order to clarify the differences between these processes, we provide our definitions.
Homing
Homing is the first—and a fairly rapid—process (measured in hours and no longer than 1-2 days) in which circulating hematopoietic cells actively cross the blood/BM endothelium barrier and lodge at least transiently in the BM compartment by activation of adhesion interactions prior to their proliferation. Aging neutrophils express high CXCR4 levels and home to the BM prior to their apoptotic cell death.11 Stem cells also home to other organs (especially in response to stress signals, transmitted in response to alarm situations such as total body irradiation [TBI]) and can first migrate to the spleen prior to BM homing.12,13
Engraftment and repopulation
Short-term engraftment (ranging from weeks to a few months) is initiated by differentiating progenitors. Durable long-term multilineage engraftment (many months in mice, and years in patients) is carried out by stem cells following their unique homing to their specialized niches.14 Hematopoietic stem cells require the BM microenvironment, which regulates their migration, proliferation, and differentiation in order to maintain active hematopoiesis throughout life.15,16
Similarities
The presence of donor-type hematopoietic cells in the BM is the outcome of both homing and engraftment/repopulation. Both processes are partially regulated by common mechanisms of migration and adhesion (eg, SDF-1/CXCR4 signaling, very late antigen 4 [VLA-4]/vascular cell adhesion molecule 1 [VCAM-1] and CD44/hyaluronic acid [HA] interactions).
Differences
Homing is a rapid process, which can lead to transient retention and does not require cell division, while engraftment does. Many cell types, including long-term repopulating human CD34+/CD38- stem cells, short-term repopulating CD34+/CD38+ progenitors, and mature, specialized T cells and neutrophils, can home to the BM, but only stem cells, which can home to their endosteal niches, initiate long-term repopulation. Murine BM homing cells, which successfully adhere to the recipient stroma, are viable, most probably due to protection from apoptosis.17 Lineage- immature cells demonstrate stronger adhesion properties (30-fold) than Lineage+ cells.18 Homing does not require host preconditioning with DNA damage, while durable engraftment with high levels of repopulation by transplanted stem cells is largely dependent on this procedure and the time interval between TBI and transplantation. For example, significantly increased survival rates of lethally irradiated mice that received transplants 24 and 48 hours after TBI with graded doses of syngeneic BM cells were documented, compared with mice that received transplants immediately after TBI.19 Of interest, this time frame is in correlation with increased secretion of SDF-1 and stem cell factor (SCF) by the irradiated murine marrow and spleen,20,21 suggesting that these and other factors influence repopulation levels of transplanted cells as documented for human CD34+/CD38-/low progenitors in NOD/SCID mice that received transplants immediately after TBI, or 24 and 48 hours later.20
The BM architecture, DNA damage, homing, and repopulation
The BM endothelium is the first anchoring site for homing cells, presenting adhesion molecules and stimulating chemokines. Human and mouse BM share common structures, but species-dependent differences can also be observed. The small blood vessels in both human and murine BM, the sinusoids in which trans-endothelial migration is thought to take place, are composed of specialized cell structures that regulate cell trafficking (Figure 1A and C, and Esser et al22 ). TBI and cyclophosphamide disrupt the physical BM endothelium barrier, allowing erythrocyte passage from the circulation to the extravascular hematopoietic space of treated but not control mice, despite their discontinuous BM endothelium with small gaps.23-25 We documented disruption of necrotic irradiated endothelium, which requires repair in the BM of conditioned patients (Figure 1D). G-CSF–induced mobilization increases the permeability and creates larger gaps between murine BM endothelial cells.26 The chemokine SDF-1 is expressed by immature human osteoblasts in the endosteum region.20 This chemokine and its receptor CXCR4, constitutively expressed by human and murine BM endothelium,20,27 are essential for stem cell seeding of the murine BM during fetal development and for definitive repopulation in adult mice that received transplants (reviewed in Cottler-Fox et al5 ). However, murine CXCR4 knockout (KO) cells can home to the BM, and the reduced retention of CXCR4 (KO) stem cells in the BM of primary but not serially transplanted recipients can be corrected by increasing the cell dose.28-30 SDF-1/CXCR4 expression is regulated by hypoxia inducible factor-1 (HIF-1), and since the BM is partially hypoxic,27 this pair is functionally expressed by many cell types including immature and maturing hematopoietic cells, stromal cells lining the stem cell rich endosteum region, and human Stro1+ mesenchymal progenitors.20,31 TBI induces increased SDF-1 and SCF secretion within 24 to 48 hours in the murine BM and spleen.5,20,21 SDF-1 secreted to the circulation from the injured murine liver can functionally pass the endothelium barrier in a CXCR4-dependent manner into the BM and recruit hematopoietic progenitors to the circulation.13 In addition, intravenously injected human SDF-1 increased homing of human mobilized and cord blood (CB) CD34+-enriched cells to the BM and spleen of nonirradiated B-2 microglobulin knock-out (B2mnull) NOD/SCID mice.33 Interestingly, 2 hours after injection, murine stem cells were observed adherent to specialized SDF-1–expressing microdomains of the BM.34 Other organs also induce HIF-1 activation and a shift in SDF-1 expression by endothelial cells in the liver,13 heart,35 and brain36 during stress. These changes, together with the secretion of other chemokines, cytokines, and proteolytic enzymes, influence stem cell homing and repopulation in opposite directions. Increased homing of transplanted progenitors to the BM, similar levels of stem cell retention but significantly decreased donor-derived repopulation levels (which require transplantation of very high cell doses) are documented in nonirradiated mice compared with TBI preconditioned recipients. The latter require much lower cell doses for donor cell repopulation (discussed in Zhong et al,37 Quesenberry et al,38 and Collis et al39 ). These results suggest that stem cell homing to other organs and mobilization induced by DNA damage reduce direct homing to and/or early retention of progenitors within the BM of irradiated recipients 1 to 2 days after TBI. However, durable repopulation by transplanted stem cells is enhanced in recipients preconditioned with TBI, which eliminates host stem cells and hematopoiesis. In addition, stress signals transmitted following conditioning with DNA damage as part of the repair process can also influence definitive stem cells in a time-dependent manner by enhancing their migration, proliferation, and differentiation, and the time interval of transplantation after TBI has major impacts on repopulation and survival.19,20
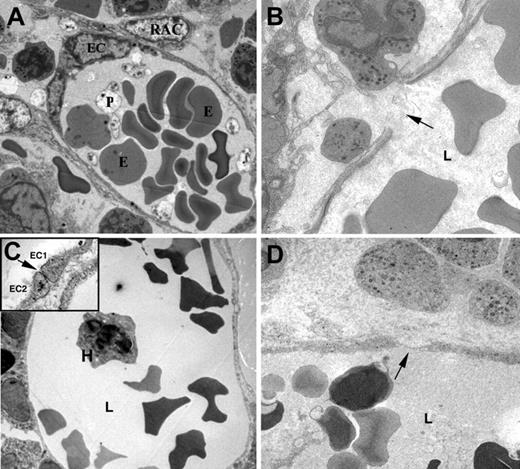
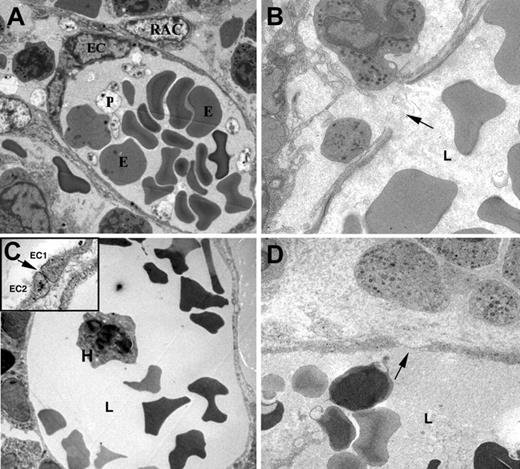
Disruption of human and murine BM sinusoids after irradiation and chemotherapy. Transmission electron microscope images of murine (A,B) and human (C,D) BM sinusoids. Elongated endothelial cells (EC) form the luminal layer of the sinusoid, surrounded by discontinuous cytoplasmic protrusions of reticular adventitial cells (RAC). Erythrocytes (E), platelets (P), and nucleated hematopoietic cell (H) are seen within the sinusoid lumen (L). The continuous murine endothelial cell layer is damaged after irradiation, resulting in gap formation due to rupture of the EC cytoplasm (B, arrow). Tight junctions between adjacent EC (C inset, arrow) are frequently seen in normal human sinusoids (C). Chemotherapy and irradiation cause significant narrowing of EC cytoplasm (D, arrow) together with rupture of the cell membrane and EC necrotic appearance (D). Images were acquired with a Techai-12 microscope (Philips, Eindhoven, The Netherlands) and Megaview III (Soft Imaging System, Munster, Germany). Samples were counterstained in uranyl acetate and lead citrate. Original magnification, × 3700 (panels A, C, D), × 5900 (panel B), and × 37 000 (panel C, inset).
Disruption of human and murine BM sinusoids after irradiation and chemotherapy. Transmission electron microscope images of murine (A,B) and human (C,D) BM sinusoids. Elongated endothelial cells (EC) form the luminal layer of the sinusoid, surrounded by discontinuous cytoplasmic protrusions of reticular adventitial cells (RAC). Erythrocytes (E), platelets (P), and nucleated hematopoietic cell (H) are seen within the sinusoid lumen (L). The continuous murine endothelial cell layer is damaged after irradiation, resulting in gap formation due to rupture of the EC cytoplasm (B, arrow). Tight junctions between adjacent EC (C inset, arrow) are frequently seen in normal human sinusoids (C). Chemotherapy and irradiation cause significant narrowing of EC cytoplasm (D, arrow) together with rupture of the cell membrane and EC necrotic appearance (D). Images were acquired with a Techai-12 microscope (Philips, Eindhoven, The Netherlands) and Megaview III (Soft Imaging System, Munster, Germany). Samples were counterstained in uranyl acetate and lead citrate. Original magnification, × 3700 (panels A, C, D), × 5900 (panel B), and × 37 000 (panel C, inset).
Human and mouse BM also have species-specific structures that need to be considered when alternative clinical procedures (intravenous vs intraosseous transplantation) are applied. Intraosseous transplantation of human CD34+- and CD34--enriched CB progenitors in immune-deficient mice resulted in much higher seeding efficiencies of both short- and long-term repopulating cells40-42 compared with intravenous transplantation. In contrast, no significant differences were observed in hematopoietic recovery of human individuals undergoing BM transplantation with both procedures.44,45 This may be attributed to structural and size-related differences between murine bones (smaller with compact bone marrow) as opposed to human bones (less compact BM containing many fat cells, bigger blood vessels, and higher blood volume; Figure 2), which may require implantation of stem cells directly to their niche, or with niche-supporting osteoblasts and endothelial cells together with an anchoring matrix needed to retain the progenitors in the injected bone.
Homing of human stem cells in preclinical animal models
The biology of human progenitor cell homing has been studied in 2 major preclinical animal models. The preimmune sheep model, developed by Zanjani et al,46 uses preimmune sheep fetuses that are implanted with immature human CD34+ and CD34- cells without preconditioning, allowing short- and long-term stem cell engraftment in sheep recipients of both primary and serially transplanted secondary transplants. In this model, immature human BM CD34+-enriched cells transplanted to sheep fetuses can be detected 24 and 48 hours later in the recipient BM.64 The second model is based on immune-deficient mice preconditioned with sublethal TBI. SCID mice given transplants of normal and leukemic human progenitor cells developed by Dick's group, facilitate high levels of multilineage (both myeloid and lymphoid) human hematopoiesis in the murine BM,47 and identified leukemic CD34+/CD38- stem cells (SCID leukemia initiating cell [SLIC]) obtained from patients with acute myeloid leukemia (AML).48 Later, NOD/SCID mice with additional reduced immunity were created by Shultz et al.128 These mice were used to identify primitive, normal human CD34+/CD38- SCID repopulating cells (SRCs),49 CD34-/CD38- SRCs,50 and SLIC from different AML French-American-British (FAB) subtypes which can initiate the disease in mice that receive serial transplants.51 B2mnull NOD/SCID mice have further reduced innate immunity due to the lack of natural killer (NK) activity. Definitive SRC function of high-level multilineage engraftment in primary NOD/SCID mice and B2mnull NOD/SCID mice that received serial transplants is dependent on human CXCR4 signaling by murine SDF-1, which is cross-reactive4 ; SLIC function is also CXCR4 dependent.8,9 CD34+/CD38+ short-term repopulating cells (STRCs), which are enriched in mobilized peripheral blood (MPB), were identified by Eaves's group using B2mnull NOD/SCID mice52 or by inactivation of NK cell activity in NOD/SCID mice by other groups.53,54 Mechanisms of human stem cell homing were mainly characterized in immune-deficient NOD/SCID and B2mnull NOD/SCID mice55 ; however, the xenogeneic barrier–dependent limitations of these models may explain the low frequency of homing progenitors. Nevertheless, a positive correlation between engraftment in patients and NOD/SCID and B2mnull NOD/SCID mice was demonstrated.56
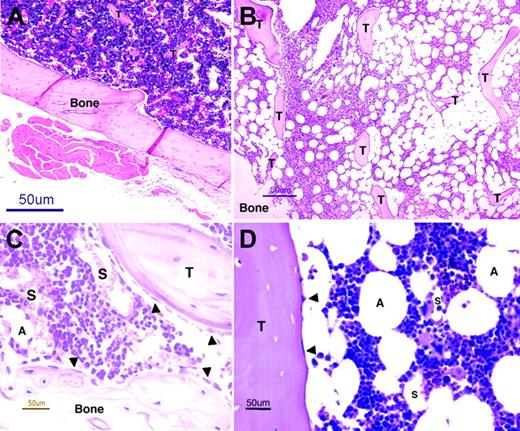
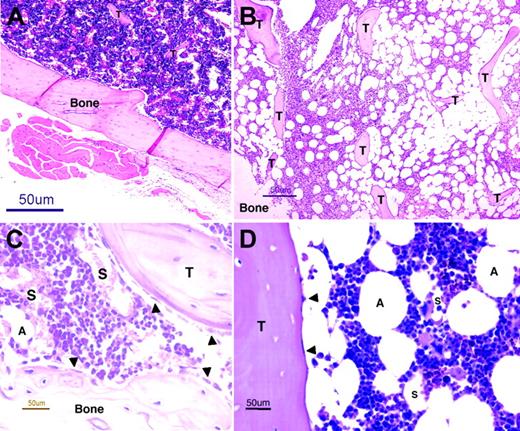
BM architecture. Light microscopy images of murine (panels A,C) and human (panels B,D) BM sections stained with hematoxylin and eosin. Extensive invasions of spongy bones (trabeculae, T) and many fat cells (adipocytes, A), residing between the hematopoietic niches, characterize human BM (B) and distinguish it from the murine BM (A). Murine (C) and human (D) BM contains many small blood vessels (sinusoids, S), which are the site where progenitors home to the BM and where maturing cells and erythrocytes egress to the circulation (B). A monolayer of immature osteoblasts, which lines the bone and the trabeculae, defines the endosteum region (arrowheads, panels C,D), wherein hematopoietic stem cells reside. Images were acquired using an Eclipse E800 microscope (Nikon, Tokyo, Japan) and a Nikon DXM1200 using Nikon ACT-1 software. For panels A and B, objective × 4/15.7 NA was used (original magnification, × 40); for panels C and D, objective × 40/0.14 NA was used (original magnification, × 400).
BM architecture. Light microscopy images of murine (panels A,C) and human (panels B,D) BM sections stained with hematoxylin and eosin. Extensive invasions of spongy bones (trabeculae, T) and many fat cells (adipocytes, A), residing between the hematopoietic niches, characterize human BM (B) and distinguish it from the murine BM (A). Murine (C) and human (D) BM contains many small blood vessels (sinusoids, S), which are the site where progenitors home to the BM and where maturing cells and erythrocytes egress to the circulation (B). A monolayer of immature osteoblasts, which lines the bone and the trabeculae, defines the endosteum region (arrowheads, panels C,D), wherein hematopoietic stem cells reside. Images were acquired using an Eclipse E800 microscope (Nikon, Tokyo, Japan) and a Nikon DXM1200 using Nikon ACT-1 software. For panels A and B, objective × 4/15.7 NA was used (original magnification, × 40); for panels C and D, objective × 40/0.14 NA was used (original magnification, × 400).
Homing is a rapid process
Murine progenitors57 and human CD34+ cells58 transplanted intravenously are rapidly cleared from the recipient circulation, within minutes. Human CD34+ progenitors home to different organs including spleen, liver, and lungs,13,59 and may home later from these organs to the BM as previously shown for murine cells.12 Of interest, radiolabeled BM CD34+ cells transplanted into patients with acute myocardial infarction rapidly homed to the liver and spleen (but were not detected in the BM) within a few hours.59 In B2mnull NOD/SCID mice that received transplants, we could detect homing of human CB CD34+ cells to the BM and spleen just 2 hours after infusion, which is twice as fast as homing of the same cell population in NOD/SCID mice.60
Origin-dependent properties of CD34+ cells
Neonate, umbilical CB–derived CD34+ cells differ from adult BM or MPB CD34+-derived cells in their BM homing (which is reduced) and SRC repopulation properties (which are increased) in NOD/SCID mice that received transplants. CB CD34+ cells demonstrate reduced rolling on BM endothelium, which is a prerequisite step for transendothelial extravasation and successful homing compared with adult CD34+ cells. Partial expression of a nonfunctional form of the P-selectin ligand, P-selectin glycoprotein ligand 1 (PSGL-1), on the surface of some CB CD34+ progenitors enriched with the primitive CD34+/CD38- subset, mediates this reduced function.58 Of interest, children who received transplants of a cell dose of BM 13 times higher than CB cells gained faster neutrophil and platelet recovery; however, the levels of progenitors in the BM of children 1 year after transplantation with CB were significantly higher.61 These data imply a different repopulation potential, which may result from different homing, proliferation, and differentiation properties of transplanted stem cells and suggest that CB progenitors which have successfully homed to and are retained in the host BM do not fully respond to differentiating signals and are biased to maintaining their immature state.
Progenitor subsets
Different cell subpopulations may vary in their homing properties. Total nucleated cells from human CB, BM, PB, and CB CD34+-enriched cells transplanted into lethally irradiated NOD/SCID mice demonstrate similar seeding efficiencies in the murine BM and spleen within 24 hours.62 However, despite similar homing efficiencies 3 hours after transplantation of enriched CD34+ cells (cotransplanted with irradiated mature cells) derived from the above-mentioned sources, they display different repopulation rates.54 We have demonstrated that enriched CB CD34+/CD38- cells, home successfully to the murine BM and spleen while sorted CD34+/CD38+ maturing progenitor cells, which lack long-term repopulation capacity, have poor homing capabilities unless prestimulated with cytokines such as SCF in vitro or G-CSF introduced for mobilization of adult progenitors in vivo63 or cotransplanted with mature mononuclear cells.54,55 In contrast to CD34+ progenitors, sorted Lin-/CD34-/CD38- cells have very low engraftment levels in vivo and rare colony formation in vitro.50 These cells express low levels of membrane CXCR463 and have negative regulators of CXCR4 signaling.64 While these cells display enhanced random migration, they poorly migrate toward a gradient of SDF-1 in vitro, explaining their extremely reduced homing and repopulation.50,63 Bypassing the mechanical blood/BM barrier by direct injection into the bone enabled the development of CD34+ cells in the murine bone injected with human CB CD34- cells, and also their migration in vivo to bones that were not injected.41 This transplantation approach revealed that CD34- primitive SRCs/stem cells have higher migration, proliferation, and secondary transplantation potential.65 Interestingly, when Lin-/CD34- cells were precultured with CD34+ cells, their repopulation potential as well as their development into CD34+ cells are increased.66 Alternatively, in vitro incubation of CD34- progenitors with stromal cells and their stimulation with cytokines induce CD34+ expression, colony formation, and definitive SRC repopulation.67
The accessory effect
Nonrepopulating maturing and mature cells facilitate the repopulation potential of cotransplanted CD34+ progenitors.55,68 Cotransplantation of CD34+/CD38+ maturing progenitors with CD3-depleted CB cells facilitated their homing and transient engraftment.54 We found that prestimulation of CB CD34+ cells with SCF induces surface CXCR4 expression4 and the appearance of cycling G1 CD34+/CD38+ cells with increased production of the proteolytic enzyme matrix metalloproteinase 9 (MMP-9), SDF-1–directed migration and homing to the BM and spleen of NOD/SCID mice that received transplants. More importantly, these G1 CD34+ cells actively support and enhance SDF-1–induced migration, homing, and BM repopulation capacity of cotransplanted, quiescent G0 CD34+ cells, which include all the primitive CD38-/low cells.69 Possible mechanisms to explain the accessory effect exerted by maturing cells are cytokine secretion70 or production of proteolytic enzymes such as MMP-9,69 which enhance CXCR4 expression, cell motility, and SDF-1–directed migration of progenitor cells.13 Other cell types, such as CD8+ T cells,71 were also shown to augment the homing of CD34+ cells in cotransplantation studies. A prolonged accessory effect is apparently exerted by cotransplanted mesenchymal cells, which home to the murine lungs, and augment engraftment but not homing of human CB CD34+ cells.72
SDF-1/CXCR4 interactions
SDF-1/CXCR4 interactions and signaling have been implicated as a principal axis regulating retention, migration, and mobilization of hematopoietic stem cells during steady-state homeostasis and injury (reviewed in Cottler-Fox et al5 ). We have shown that enriched human CD34+ SRCs/stem cells homing to the BM require functional interactions between human CXCR4 and murine SDF-1, which is presented on the vasculature endothelium and various BM stromal cells. Repopulation by SRCs4,20,63 and SLIC function (both homing and development) are also CXCR4 dependent.8,9
Intracellular and surface expression
CXCR4 expression is very dynamic. Cytokine stimulation with SCF + interleukin-6 (IL-6) or hepatocyte growth factor (HGF) up-regulates CXCR4 expression by human CD34+ cells within 24 to 48 hours, leading to their increased SDF-1–mediated migration, in vivo homing, and repopulation.4,13,63 Similarly, SCF + IL-6 stimulation increases CXCR4 expression by Sca-1+/Lin- murine progenitors73 and membrane-bound SCF is essential for murine stem cell homing to the endosteum region.74 Overexpression of this receptor by human CB and MPB CD34+ cells by lentiviral infection led to increased cell motility and SDF-1–induced migration, higher proliferation rates in vitro, and, moreover, increased SRC repopulation.75,76 Interestingly, CXCR4-overexpressing CD34+ cells demonstrate increased homing to the spleen but not to the BM of NOD/SCID recipients,75 suggesting that human cells home first to the more permissive tissue, the spleen, and only later to the murine BM.59,60 High doses of SDF-1 induce desensitization, receptor internalization, cell survival, and quiescence, while low doses promote cell motility, proliferation, and migration (reviewed in Lapidot and Kollet55 ). Low-dose stimulation with SDF-1 or anti-CXCR4 antibody (Ab) both increase migration to this ligand and repopulation by MPB CD34+ cells, but not BM CD34+ cells.3 Eaves et al showed that in vivo treatment with a high dose of SDF-1,77 or in vitro pretreatment with a lower SDF-1 concentration,78 increased engraftment by human repopulating cells.
A single injection of human SDF-1 to nonirradiated B2mnull NOD/SCID mice followed by CD34+ cell transplantation increased homing of MPB and CB CD34+ cells to the murine BM and spleen. The injected ligand can cross the BM endothelium in a CXCR4-dependent manner to the endosteum region, conveying messages from peripheral organs and increasing homing of human progenitors including primitive CD34+/CD38-/low cells.33 Sorted CB CD34+ cells, which do not express CXCR4, are less motile and home at a much slower rate compared with their CXCR4+ counterparts. These CD34+/CXCR4- cells harbor low levels of intracellular CXCR4. Nevertheless, membrane expression of this receptor is induced in vivo upon transplantation, mediating their delayed homing and reduced repopulation. In vitro prestimulation (24-48 hours) with cytokines can also convert these cells into definitive SRCs with high surface and intracellular CXCR4 expression and SDF-1–induced migration.79 In order to block new surface CXCR4 expression from the large intracellular pool of CD34+ cells, cell transplantation with excess amounts of unbound anti-CXCR4 Ab is required.9,63,79 The low levels of engraftment obtained with high doses of human CD34+/CXCR4+ and CXCR4- sorted cells, both treated with neutralizing Ab which were washed before transplantation,80 most probably reflect the dynamic expression of this receptor. CXCR4 can also be functionally transferred from platelet microparticles to BM CD34+ cells and augment their SDF-1–mediated migration, adhesion, homing, and engraftment potential.81
CXCR4 colocalization in lipid rafts
Gene knockout studies in mice demonstrate the essential role of Rac1 in BM homing of murine stem/progenitor cells.82 Interestingly, Rac1 colocalization with CXCR4 in lipid rafts is suggested to potentiate human BM CD34+ cell migration and repopulation83 as previously shown for human T cells.84 However, SDF-1–mediated chemotaxis and in vivo homing can be augmented by other mechanisms, as shown by stimulation with the complement component activation protein, C3a85 , or other leukopheresis products from mobilized patients83 that increased incorporation of CXCR4 into membrane lipid rafts of CD34+ cells and their homing.
Homing and repopulation by CD34+ cells can also be manipulated by enhancing the production of BM SDF-1 via application of DNA-damage–inducing agents such as TBI or chemotherapy,20 or hypoxia that has been shown to regulate SDF-127 and CXCR4 expression86 by HIF-1α. Incubation of CD34+/CD38- BM cells in hypoxic conditions led to HIF-1α stabilization, their CXCR4 expression, cell survival, in vivo homing, and repopulation potential.87 In addition, CD8+ T cells, which improved the migration, BM homing, and engraftment of CD34+ cells, also reduced focal adhesion kinase (FAK) phosphorylation in CD34+ cells cultured with them in response to SDF-1 stimulation.71
CXCR4 signaling and regulation of homing
An important component of the CXCR4 signaling pathway is atypical protein kinase C ζ (PKCζ), which translocates to the cell membrane upon SDF-1 stimulation. We revealed that SDF-1–induced chemotaxis, polarization, MMP-9 secretion, and adhesion of human CD34+ cells are PKCζ dependent. Interestingly, while a PKCζ inhibitor prevented BM repopulation and in vitro migration to SDF-1 by human CD34+ cells, their homing was not affected, suggesting a role for PKCζ in human BM seeding of the endosteal region and retention of SRCs.88 The broad-range PKC inhibitor, chelerythrine chloride, totally abrogates homing of both human CD34+ cells and murine progenitors to the murine BM.63,89 In addition, PKCζ mediates functional elevation of CXCR4 expressed on immature human CD34+ cells and MMP2/9 secretion, in response to cyclic adenosine 3′,5′-monophosphate (cAMP) stimulation. cAMP-treated CD34+ cells gain increased SDF-1–induced transendothelial migration, adhesion to stromal cells, and enhanced homing to the BM, which was dependent on Rac-1 and PKCζ signaling (P. Goichberg, A. Kalinkovich, N. Bordovsky, et al., manuscript submitted, 2005). Pertussis toxin (PTX), known to inhibit signaling of Gαi protein–coupled receptors such as CXCR4, blocks in vitro migration of human CD34+ cells toward a gradient of SDF-1; however, their in vitro SDF-1–mediated adhesion and in vivo homing were contrary enhanced when a 1-hour incubation with PTX was applied.63 Similarly, PTX treatment of murine progenitors reduced their colonization of the murine spleen but not the BM.90 Interestingly, costimulation of murine fetal liver and BM cells with PTX and SCF for 24 hours significantly reduced their homing.91 This study suggests that longer PTX incubations than previously performed90 are needed to block homing of murine stem cells. However, in our original experiments with human CD34+ cells, when incubations with PTX were prolonged for 24 hours, no inhibition of homing was documented. Alternatively, short PTX treatment for 1 hour, which was preceded by 24 hours of stimulation with SCF, significantly reduced homing levels by human CB CD34+ cells (O.K., T.L., unpublished data, 2000), suggesting that costimulation with cytokines such as SCF and longer SCF stimulation with short PTX treatment block homing of human CB CD34+ progenitors since their motility is influenced by cytokine signaling, which induces cell polarization.92 Of interest, dissemination of leukemic murine cells to the liver and spleen but not the BM were blocked by PTX, while inhibition of Gq11 signaling prevented both liver and spleen dissemination and BM colonization.93 Interactions mediated by the Rac, Rho, and Cdc42 pathway are also implicated in stem cell migration. The lysophospholipid sphingosine 1–phosphate (S1P) and its specific receptors S1PRs, a novel class of G-protein–coupled receptors, interact via the Rho family proteins, and are implicated in cell migration. In the mouse system, S1P synergizes with SDF-1 to augment its chemotactic effect on primitive hematopoietic cells.94 Activation of S1PRs expressed by human MPB CD34+/CD38- cells increases their CXCR4 signaling, transendothelial migration and in vivo homing without affecting CXCR4 expression; however, increased engraftment was not statistically significant.95 Knocking out Rac-1 significantly reduced SDF-1 migration and abolished homing of murine stem cells to the endosteum, which is essential for long-term, stem cell–mediated repopulation.96 Cells deleted of both Rac-1 and Rac-2 alleles highly express CXCR4, fail to migrate to SDF-1, and are massively mobilized to the circulation.82
The homing machinery: cell adhesion molecules
Adhesion molecules (selectins and integrins) mediate rolling and adhesion of homing cells to the blood vessel wall prior to their extravasation and seeding of the BM. A subset of CB CD34 cells, which are enriched in primitive CD38- cells, demonstrate reduced rolling and repopulation due to partial expression of a nonfunctional form of the P-selectin ligand PSGL-1.58 Neutralizing the integrin VLA-4 blocks homing of human CD34+ cells to the BM of fetal sheep.46 While VLA-4, VLA-5, and LFA-1 participate in the homing of CD34+ cells to the BM and spleen of NOD/SCID mice,63 VLA-4 and VLA-5,97 but to a much lesser extent LFA-1,98 are essential for BM repopulation by these cells. We found that SDF-1 is involved in the regulation of CD34+ cell adhesion during their homing to the BM. Stimulation by SDF-1 induces transendothelial migration,98 activates cell-surface integrins, and mediates firm arrest of CD34+ progenitors under shear flow.99 SDF-1 synergizes with other molecules to potentiate CD34+ cell adhesion and motility. Thus, we have shown that the adhesion molecule CD44 expressed by human CD34+ cells and its ligand hyaluronic acid (HA) expressed in BM sinusoid endothelium and the endosteum region, are essential for homing and repopulation, which are both SDF-1 dependent.100 Of interest, inhibition of CD44 vetoed CD34+ cell activation and protrusion formation by SDF-1, suggesting that combined CD44 ligands, HA and osteopontin (binding different sites of CD44) expressed in the stem cell niche, prevent SDF-1–induced migration and secretion of MMP-2/9 by CXCR4+ progenitor cells. Interestingly, human CD34+ cells express HA (preferentially the CD38- subset101 ), and a unique isoform of CD44, which is not expressed by mature cells, shown to mediate rolling on endothelial E/L-selectin ligand under flow.102 Preliminary results demonstrate an essential role for the CD45 phosphatase, which negatively regulates adhesion interactions in homing and repopulation by human CD34+ cells. Neutralization of this receptor impaired migration to SDF-1, BM homing, and repopulation of NOD/SCID mice.104
Degrading enzymes
Proteolytic enzymes are widely expressed by various cell types during steady-state and injury conditions, regulating hematopoietic cell migration and tissue localization. SDF-1 and other factors induce the secretion of matrix metalloproteinases MMP-2 and MMP-9. These enzymes are secreted by circulating but not BM CD34+ cells, facilitating their in vitro migration toward SDF-1.104 Freshly isolated CB CD34+ cells do not express MMP-2/969,105 ; however, upon incubation with MMP-2/9–rich conditioned media, these cells gain increased CXCR4 expression and SDF-1–directed migration.13 In accordance, MMP-2/9 inhibition reduces homing of human CB CD34+ cells. Similarly, incubation with SCF induces expression of MMP-2/9 and moreover, increased homing and repopulation of NOD/SCID mice.69,105 Preliminary results document that the cell-surface proteolytic enzyme membrane type 1 matrix metalloproteinase (MT1-MMP) is expressed by human CD34+ progenitors, is activated by SDF-1 and G-CSF, and is needed for SDF-1–induced migration in vitro and for in vivo homing and mobilization.106,107 Interestingly, proteolytic enzymes also negatively regulate cell migration. N-terminal cleavage of SDF-1 associated with its chemotactic inactivation was demonstrated by several degrading enzymes including neutrophil elastase, cathepsin G,108-110 cathepsin K,111 and MMP-2/9,112 and C-terminal cleavage was attributed to carboxypeptidase N.113 Neutrophil elastase and cathepsin G also cleave CXCR4 on human T cells and CD34+ cells, which can also affect their motility.110,114 These studies demonstrate that proteolytic enzymes are involved in both turning on and off the locomotion machinery of human CD34+ cells, thus mediating stem cell recruitment and organ localization. CD26, a cell-surface peptidase, is expressed on the surface of different hematopoietic subsets, including normal BM CD34+ cells115 and a subfraction of CB CD34+ cells that are mainly CXCR4-expressing cells.116 CD26 inhibition enhanced the migratory response of CB CD34+-treated cells to SDF-1,117 supporting a previous finding that CD26 cleaves and inactivates many ligands, including SDF-1.118 Murine Sca-1+/Lin- stem cells obtained either from CD26 KO mice or wild-type cells treated with a CD26 inhibitor demonstrate increased (∼ 30%) homing to the BM of wild-type mice preconditioned with TBI.73 Higher levels of functional SDF-1 documented in the circulation of CD26 KO mice suggest that CD26 is mostly responsible for inactivation of this ligand in the circulation.119
A shortcut to the BM: intraosseous stem cell transplantation
In an attempt to improve the efficiencies of BM homing, seeding, and repopulation, transplanted cells were injected directly into the BM cavity of immune-deficient NOD/SCID and normal mice (intraosseous [IO], intra–bone marrow [IBM], or intrafemur [IF] transplantation). Studies with enriched human progenitors demonstrate superior seeding efficiencies when the endothelial and ECM barriers are bypassed. IBM injections yielded a 15-fold higher frequency of SRCs in sorted CB CD34+/CD38- cells in comparison with conventional intravenous injection. Increased engraftment and improved abilities of BM cells obtained from primary recipients to repopulate recipients of secondary transplants were also documented. Simultaneous blocking of the integrins VLA-4/5, which have been implicated in the homing and repopulation processes, profoundly inhibit engraftment of human progenitor cells injected intraosseously. Interestingly, CXCR4 neutralization similarly abolishes engraftment by human CD34+-enriched cells administered in both protocols, demonstrating an essential role for this receptor not only in SRC homing to the BM but also in BM seeding and colonization.40 Thus, this method enables researchers to dissect different steps of BM repopulation. Dao et al120 revealed that CD34 expression by human progenitors is dynamic, where positive cells can lose and regain CD34 expression in vivo. CB-derived Lin-CD34- primitive cells display rare and low repopulation potential when transplanted via the tail vein,50,66 as opposed to high SRC activity when transplanted intraosseously,41 despite low expression of CXCR4 and reduced migration to SDF-1.41,63,79 These progenitors acquire in vivo expression of the CD34+ marker, demonstrating slower differentiation and reconstitution kinetics but higher absolute numbers of CD45+ and immature human CD34+ cells in comparison with human cells initially expressing CD34.41,65 Applying intrafemoral injection of CB CD34+CD38-/lowCD36- purified cells, Dick's group could identify a new short-term SRC subset that can migrate to other bones, colonizing them with myeloid and erythroid cells.43 Taken together, this method enables the revealing of repopulating human stem cell subsets that can hardly be detected when injected intravenously.121 Of interest, when total CB mononuclear cells (MNCs) were transplanted, increased engraftment, including a dramatic increase in immature human CD34+ cells, were documented transiently only 1 month after intraosseous transplantation compared with intravenous transplantation, but not 2 and 3 months after transplantation.42
Studies with primitive murine BM cells demonstrate similar results. IBM injection of BM MNCs led to a 1.4-fold increase in donor-type cells in the recipient BM, while their levels in the peripheral blood were lower (1.3-fold) in comparison with intravenous injection, 3 months after transplantation.42 Intrafemoral injection of murine Lin- cells supported increased homing after 20 hours both to the noninjected femurs and spleens of nonablated mice, in comparison with intravenous transplantation. However, effective bypass of the endothelial and ECM barriers is not sufficient for improved long-term BM reconstitution. TBI preconditioning, which is widely used for clinical transplantation, led to egress of some Lin- cells transplanted immediately after TBI, from the injected femur to the circulation of ablated mice only (within 20 hours), suggesting that implanted progenitors are mobilized or not retained well due to the disruptive BM endothelium. Despite reduced IF homing in irradiated recipients, donor cell proliferation and repopulation rates are increased compared with nonirradiated mice. However, retention of undifferentiated donor stem cells in the BM of nonconditioned recipients is evident by transplantation into lethally irradiated secondary recipients,37 implying that TBI is further needed to reduce competition by host stem cells, and to provide signals which enhance BM reconstitution in a time-dependent manner.19 Intra–bone marrow transplantation (IBMT) of marked cells without preconditioning in primates also led to low levels of engraftment in the circulation (< 0.1%), despite much higher levels in the implanted bone (up to 30%).122
Finally, to prevent stress and severe damage to the BM structure associated with intrafemoral injection, and to ensure cell introduction to the femur blood vessels rather than to the BM cavity, Scadden's group has examined homing properties of murine BM Lin- cells injected to the femoral artery. While homing to the BM and spleen was significantly increased by femoral artery versus tail-vein injection in mice, no differences were found in long-term donor cell repopulation,123 suggesting that BM repopulation is not enhanced by this procedure and that stress and damage due to the intraosseous implantation procedure also contribute to the repopulation levels. These results, together with the need for anchoring intraosseously transplanted human progenitors with a matrix to prevent their release to the circulation, may explain the lack of improvement in human intraosseous transplantation compared with conventional intravenous transplants and suggest modified intraosseous protocols.
Clinical relevance and concluding remarks
Key experiments by Voermans et al, showing a correlation between spontaneous and SDF-1–mediated migration in vitro of CD34+ cells and hematopoietic neutrophil and platelet recovery in patients who received transplants of autologous CD34+-enriched MPB cells, emphasize the importance of definitive human stem cell motility and repopulation in clinical protocols.2 Similar results revealed a correlation between CXCR4 expression and repopulation with allogeneic CB transplantation and the rate of platelet recovery in allogeneic-matched transplantations with CD34-enriched cells.124,125 The levels of SDF-1–induced migration of human CD34+ cells also correlate with repopulation in NOD/SCID mice3,4 and, moreover, human SRC function (including platelet and colony-forming unit–megakaryocyte [CFU-Mk]) in transplanted B2mnull NOD/SCID mice correlates with platelet recovery in patients who received autologous transplants.56 Mobilizing agents not only affect the levels of cell egress but also have an impact on CXCR4 function and the potential of mobilized cells to home back to the recipient BM.83 Human CD34+ cells from donors mobilized with both G-CSF and the CXCR4 inhibitor AMD3100 express higher levels of VLA-4, migrate significantly better to SDF-1 (7-fold), and engraft at much higher levels (8-fold) in NOD/SCID recipients who received transplants, compared with cells obtained from donors treated with G-CSF alone.126 Similarly, Rhesus CD34+-enriched cells mobilized with AMD3100 have higher VLA-4 and CXCR4 expression, SDF-1–induced migration in vitro, and in vivo BM repopulation in autologous primate transplantation compared with G-CSF–mobilized cells.127
Stem cell homing to the endosteum. Homing of human CD34+/CXCR4+ stem cells from the blood circulation via the murine BM sinusoids to the endosteum. Key molecules involved in this process, reported in recent studies (in addition to P/E-selectins and SDF-1–activated LFA-1, VLA-4, VLA-5, and their ligands [Peled et al,98,99 Kollet et al,63 and Manfredini et al64 ] which were omitted for simplicity) are illustrated.
Stem cell homing to the endosteum. Homing of human CD34+/CXCR4+ stem cells from the blood circulation via the murine BM sinusoids to the endosteum. Key molecules involved in this process, reported in recent studies (in addition to P/E-selectins and SDF-1–activated LFA-1, VLA-4, VLA-5, and their ligands [Peled et al,98,99 Kollet et al,63 and Manfredini et al64 ] which were omitted for simplicity) are illustrated.
This review has surveyed similarities between human progenitor cell motility, homing to the BM (Figure 3), and repopulation. Stem cell migration and repopulation are sequential events that are largely overlapped in their regulation. However, differences between homing and repopulation and the critical role of DNA damage and stress in successful repopulation but not BM homing (which is reduced) are also described.
Future directions should be aimed at a better understanding of the factors involved in homing and repopulation. Specific modifications of host factors such as SDF-1, SCF, and MMP-2/9 levels in the recipient BM, as well as further characterization of the human stem cell niche, including manipulation of these niches (eg, intraosseous transplantation with an anchoring matrix directly to the niche or intraosseous cotransplantation together with niche-supporting osteoblasts and endothelial cells) may lead to improved in vivo navigation and repopulation potential of definitive stem cells. In parallel, donor factors such as increased CXCR4 and c-kit expression, Rac-1 activation, and signaling by a wide variety of agonists will add to host manipulations in order to develop improved transplantation protocols.
Prepublished online as Blood First Edition Paper, May 12, 2005; DOI 10.1182/blood-2005-04-1417.
Supported in part by Ares-Serono, Geneva, Switzerland.
The authors have a few pending patents related to topics discussed in this review.
We thank Dr Vera Shinder for processing and analysis of electron microscope specimens and Dr Mariusz Ratajczak for reviewing the manuscript.

![Figure 3. Stem cell homing to the endosteum. Homing of human CD34+/CXCR4+ stem cells from the blood circulation via the murine BM sinusoids to the endosteum. Key molecules involved in this process, reported in recent studies (in addition to P/E-selectins and SDF-1–activated LFA-1, VLA-4, VLA-5, and their ligands [Peled et al,98,99 Kollet et al,63 and Manfredini et al64] which were omitted for simplicity) are illustrated.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/106/6/10.1182_blood-2005-04-1417/6/m_zh80180584140003.jpeg?Expires=1769109810&Signature=GxDN4-5WdufmkzDoEP36NTdbU2Q7U7Ot4DWA-Eps4XN6ucUnomx0XIV38Lqmuwj3Y4l3Uy4KYRPq9j6XBbmmDt4IjhADj4DqTSPLC5zng9rafqCb5NqneqE3jWUI~Se~9oF4CyMifHLaBN0Jx9n~7~QuZ6CO9b~ltYk0fekTtmORWHTYGbBwLXU3Uc-YPot638POx0fsM2wsaZ6Vk9zMrh~DlW3wrvW-EfIet~m81kkZC7pPcxxiSTJfCOFzMBNIT-kG2WTabW~gQyAGt3umDbVl~UeBC0~bJDze7uFig1nZr9O3QoAzIKSVxWL2SP6iJaLndNq9~M55K3jQyYB8sw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Figure 3. Stem cell homing to the endosteum. Homing of human CD34+/CXCR4+ stem cells from the blood circulation via the murine BM sinusoids to the endosteum. Key molecules involved in this process, reported in recent studies (in addition to P/E-selectins and SDF-1–activated LFA-1, VLA-4, VLA-5, and their ligands [Peled et al,98,99 Kollet et al,63 and Manfredini et al64] which were omitted for simplicity) are illustrated.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/106/6/10.1182_blood-2005-04-1417/6/m_zh80180584140003.jpeg?Expires=1769327614&Signature=btFSJdsaYLV46i7bezl7z6L5~J7rnPYoUlC7cEgIUdx-Mpk60p5gpiDD1lTdOgwgsdrO8bKlukbP7oUb6TQPUPO9P30uD1Lk4JguXxSeSOzvf4UzFQqX18283lDy0oNJTDpfSVQ74d7dgVz4NCfj3~~2plVBh~1ECOfoXB6yaKj1LaZgI2BenpMC52XcrfYM~X9kh~zYTbe2ucbhAwP35dbW~zjE9h618dgM~Udwl6RFeypQPVe44trPnfFLl92WuwvuKhJ8VRq6U6kWk9GSVbq39kyodb9lhsH2DN5lNMmRG2kf5BgcVhSpmgsn2vL2-28NToLjE8CC2pncXymYgg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)