TO THE EDITOR:
Philadelphia-like (Ph-like) or BCR-ABL1–like B-cell precursor acute lymphoblastic leukemia (B-ALL) is genetically heterogenous,1,2 with a gene expression profile similar to that of BCR-ABL1+ ALL.3,4 It comprises 15% to 30% of B‐ALLs, with a peak of incidence in young adults.5,6 The most commonly mutated pathways are the ABL and JAK-STAT pathways with multiple rearrangements and lesions, which are druggable with a variety of tyrosine kinase inhibitors (TKIs).7,8 Rarer and more heterogeneous alterations, although often targetable with TKIs (eg, ETV6-NTRK3),9 may be missed by targeted genomic diagnostic approaches. We describe a case of Ph-like ALL and a novel gene fusion identified by RNA sequencing (RNA-seq) that juxtaposed exon 7 of G3BP2 with exon 11 of KIT and encoded a fusion protein retaining the nuclear transport factor 2 domain of G3BP2 and the entire kinase domain of KIT. In hematological malignancies, activating KIT mutations play a crucial role in systemic mastocytosis and acute myeloid leukemia10 ; however, fusions have not been described in leukemia or in other tumors.
We report a 52-year-old White man with second relapsed B-ALL after chemotherapy and blinatumomab (registered at www.clinicaltrials.gov as #NCT02013167).11 A bone marrow aspirate specimen obtained at study entry in the INOVATE trial (#NCT01564784)12 was analyzed by RNA-seq, given the patient’s therapeutic resistance to date. At relapse, the patient had 51% blast cells in bone marrow by flow cytometric analysis. Of note, the patient was not treated with TKIs, and primary leukemia cells were not available for functional studies.
The fusion gene G3BP2-KIT was synthesized by the BioXp 3200 system (Codex DNA, Inc.) and cloned into a Gateway®-compatible cl20–murine stem cell virus–internal ribosome entry site–green fluorescent protein (cl20-MSCV-G3BP2-KIT-IRES-GFP) lentiviral vector. The truncated (TR) transcript portion of KIT fused to G3BP2 and coding for amino acids 560 to 976 (TR KIT 560-976AA) was amplified and cloned (cl20–MSCV–TR KIT 560-976AA–IRES–GFP). These constructs and the empty vector (MIG) were used for virus production and expression in interleukin-3 (IL-3)–dependent mouse hematopoietic Ba/F3 cells and IL-7–dependent ARF-null pre-B cells.13 Cell proliferation was examined in the absence and presence of cytokines. Sublethally irradiated (550 rad) 8-week-old wild-type C57BL/6 female mice underwent transplantation by tail vein injection of ARF-null pre-B cells expressing G3BP2-KIT or empty vector. Leukemia development was monitored by retroorbital bleeding; moribund mice were euthanized, and blood, bone marrow, and spleen samples were analyzed for evidence of leukemia by flow cytometry, histopathology, and genomic sequencing. Full methods are provided in the data supplement.
This B-ALL sample was predicted to be Ph-like B-ALL using prediction analysis of microarrays (Ph score, 1) and clustered with Ph-like B-ALL on t-distributed stochastic neighbor embedding analysis in our B-ALL subtyping pipeline.14 Moreover, RNA-seq detected a chimeric fusion between exon 7 of G3BP2 (chromosome 4:76580296, hg19) and exon 11 of KIT (chromosome 4:55593611, hg19), juxtaposing the amino acid residues 2227 and 560 of G3BP2 and KIT, respectively, and retaining the nuclear transport factor 2 domain of G3BP2 and part of the catalytic domain and the entire kinase domain of KIT (Figure 1A; supplemental Table 1). This fusion was not previously detected in larger cohorts of B-ALL cases.2,5,14 Although G3BP2 is ubiquitously expressed in the different hematopoietic lineages (supplemental Figure 1A), KIT is not normally expressed in B-lymphoid cells14 (supplemental Figure 1B). Thus, the fusion drives ectopic expression of the KIT transcript region fused to G3BP2 in B cells, as demonstrated by RNA-seq (Figure 1B). Analysis of sequence mutations detected few additional alterations, but not in recurrently mutated genes in ALL, except for ETV6, RPL22, and NT5C215 (supplemental Table 2), suggesting that the chimeric fusion transcript may have been a sufficient leukemogenic driver lesion in this patient. The fusion protein lacks the transmembrane domain of KIT, and immunofluorescence analysis of transiently transfected HEK293T cells showed a cytoplasmic localization (supplemental Figure 2A-B).
G3BP2-KIT fusion. (A) Schematic representation of wild-type G3BP2 and KIT and the chimeric fusion between exon (Ex) 7 of G3BP2 and exon 11 of KIT, juxtaposing the first 227 amino acids (AAs) of G3BP2 and the C-terminus portion of KIT including AAs 560 to 976. The joined Exs from both G3BP2 and KIT are in red and bold. The dotted red lines indicate breakpoint positions within Ex 7 and Ex 11 of G3BP2 and KIT, respectively. (B) Gene expression data for G3BP2 and KIT from RNA-seq visualized by the Integrative Genomics Viewer showing overexpression of the KIT portion fused to G3BP2 (hg19). Electropherogram shows the fusion junction between G3BP2 and KIT. (C) Schematic representation of the viral vector used to express fusion protein G3BP2-KIT and TR KIT (TR KIT 560-976AA) in Ba/F3 cell lines and ARF-null pre-B cells. (D) Cytokine-independent assays in Ba/F3 cells and ARF-null pre-B cells expressing the chimeric transcript G3BP2-KIT, the only portion of KIT fused to G3BP2 (encoding for AAs 560 to 976; TR KIT 560-976AA), or empty vector (MIG) cultured without recombinant mouse IL-3 or IL-7. (E) Whole-cell lysates from GFP+ sorted Ba/F3 and ARF-null pre-B cells were subjected to protein capillary electrophoresis with the Jess instrument (Protein Simple) with the following antibodies from Cell Signaling: KIT (#3074S), phosphorylated KIT (pKIT) Tyr719 (#3391S), pSTAT5 Tyr694 (#9359S), pSTAT3 Tyr705 (#9145S), pPLCγ1 Tyr783 (#14008S), p44/42 MAPK (Erk1/2; #4695S), p4E-BP1 Thr37/46 (#2855S), and β-actin (#4970S). Borders are used for specifying different antibodies.
G3BP2-KIT fusion. (A) Schematic representation of wild-type G3BP2 and KIT and the chimeric fusion between exon (Ex) 7 of G3BP2 and exon 11 of KIT, juxtaposing the first 227 amino acids (AAs) of G3BP2 and the C-terminus portion of KIT including AAs 560 to 976. The joined Exs from both G3BP2 and KIT are in red and bold. The dotted red lines indicate breakpoint positions within Ex 7 and Ex 11 of G3BP2 and KIT, respectively. (B) Gene expression data for G3BP2 and KIT from RNA-seq visualized by the Integrative Genomics Viewer showing overexpression of the KIT portion fused to G3BP2 (hg19). Electropherogram shows the fusion junction between G3BP2 and KIT. (C) Schematic representation of the viral vector used to express fusion protein G3BP2-KIT and TR KIT (TR KIT 560-976AA) in Ba/F3 cell lines and ARF-null pre-B cells. (D) Cytokine-independent assays in Ba/F3 cells and ARF-null pre-B cells expressing the chimeric transcript G3BP2-KIT, the only portion of KIT fused to G3BP2 (encoding for AAs 560 to 976; TR KIT 560-976AA), or empty vector (MIG) cultured without recombinant mouse IL-3 or IL-7. (E) Whole-cell lysates from GFP+ sorted Ba/F3 and ARF-null pre-B cells were subjected to protein capillary electrophoresis with the Jess instrument (Protein Simple) with the following antibodies from Cell Signaling: KIT (#3074S), phosphorylated KIT (pKIT) Tyr719 (#3391S), pSTAT5 Tyr694 (#9359S), pSTAT3 Tyr705 (#9145S), pPLCγ1 Tyr783 (#14008S), p44/42 MAPK (Erk1/2; #4695S), p4E-BP1 Thr37/46 (#2855S), and β-actin (#4970S). Borders are used for specifying different antibodies.
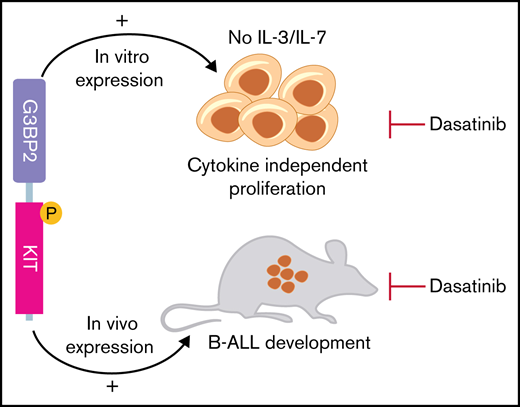
To investigate its oncogenic role, we cloned and expressed G3BP2-KIT fusion transcript, TR KIT (TR KIT 560-976AA), or empty viral vector in IL-3–dependent Ba/F3 (Figure 1C) and IL-7–dependent ARF-null pre-B cells (Figure 1D). Only the cells expressing G3BP2-KIT sustained cytokine-independent proliferation, suggesting that 5′ G3BP1 is necessary to confer oncogenic properties to TR KIT, likely by promoting KIT dimerization. Both Ba/F3 and ARF-null pre-B cells expressing G3BP2-KIT, but not those transduced with empty vector, showed phosphorylation of KIT, activation of JAK-STAT signaling with phosphorylated STAT3 and STAT5, MAPK pathway activation with phosphorylation of p44/p42 MAPK, and phosphorylation of phospholipase C (Figure 1E; supplemental Figure 3). Notably, transplantation of ARF-null pre-B cells expressing G3BP2-KIT in C57BL6 sublethally irradiated mice resulted in the development of serially transplantable B-ALL (Figure 2A), positive for B-cell lymphoid markers B220, CD19, and PAX5 (supplemental Figure 4A-B). Tumors were characterized by splenomegaly (Figure 2B) and multiorgan infiltration (supplemental Figure 4C). Whole-exome sequencing did not detect pathogenetic cooperating driver sequence mutations or copy-number changes (supplemental Table 3), confirming that G3BP2-KIT is a necessary and sufficient oncogenic driver alteration. Mouse leukemic cells showed enrichment of genes involved in JAK/STAT signaling (eg, CSF3R, IL2RA, CXCL3, IL10RA, and PIK3R5; supplemental Figure 4D-E), concordant with phosphorylation of STAT5 and STAT3 in Ba/F3 and ARF-null pre-B cells expressing G3BP2-KIT (Figure 1E).
Genetically engineered mouse models of G3BP2-KIT leukemia. (A) Kaplan-Meier survival curves of C57BL6 mice receiving ARF-null pre-B cell transplants expressing G3BP2-KIT (after 14 days in culture without IL-7 [n = 4] or within 48 hours after transduction [n = 4]) or empty vector (MIG; n = 5) and of secondary recipients undergoing transplantation with cells from primary mouse tumors. Survival comparison was analyzed by log-rank test. (B) Spleen weight from primary and secondary recipient mice with G3BP2-KIT and from control mice (MIG). The mean weight is shown by the horizontal line in the scatter dot plot, and the error bars represent the standard deviations (SDs). (C) Dose-response curve of BaF3 cells expressing empty vector (MIG) or G3BP2-KIT fusion after 72 hours treatment with dasatinib, ponatinib, imatinib, nilotinib, sunitinib, or avapritinib. The cells transduced with the empty vector were grown in media with recombinant mouse IL-3. Values are normalized to dimethyl sulfoxide controls and represent means ± SDs from 3 independent experiments performed in triplicate. (D) Left panel shows levels (means + SDs) of GFP+ cells in the peripheral blood of C57BL6 mice receiving ARF-null pre-B leukemia cell transplants expressing G3BP2-KIT from 2 primary mouse tumor cohorts (a and b groups). In group a (n = 5), dasatinib (20 mg/kg daily) or vehicle (n = 5) was started 1 week after transplantation; in group b, treatment started 2 weeks after transplantation. Right panel shows Kaplan-Meier survival curves of mice in study treatment. IC50, 50% inhibitory concentration; ns, not significant.
Genetically engineered mouse models of G3BP2-KIT leukemia. (A) Kaplan-Meier survival curves of C57BL6 mice receiving ARF-null pre-B cell transplants expressing G3BP2-KIT (after 14 days in culture without IL-7 [n = 4] or within 48 hours after transduction [n = 4]) or empty vector (MIG; n = 5) and of secondary recipients undergoing transplantation with cells from primary mouse tumors. Survival comparison was analyzed by log-rank test. (B) Spleen weight from primary and secondary recipient mice with G3BP2-KIT and from control mice (MIG). The mean weight is shown by the horizontal line in the scatter dot plot, and the error bars represent the standard deviations (SDs). (C) Dose-response curve of BaF3 cells expressing empty vector (MIG) or G3BP2-KIT fusion after 72 hours treatment with dasatinib, ponatinib, imatinib, nilotinib, sunitinib, or avapritinib. The cells transduced with the empty vector were grown in media with recombinant mouse IL-3. Values are normalized to dimethyl sulfoxide controls and represent means ± SDs from 3 independent experiments performed in triplicate. (D) Left panel shows levels (means + SDs) of GFP+ cells in the peripheral blood of C57BL6 mice receiving ARF-null pre-B leukemia cell transplants expressing G3BP2-KIT from 2 primary mouse tumor cohorts (a and b groups). In group a (n = 5), dasatinib (20 mg/kg daily) or vehicle (n = 5) was started 1 week after transplantation; in group b, treatment started 2 weeks after transplantation. Right panel shows Kaplan-Meier survival curves of mice in study treatment. IC50, 50% inhibitory concentration; ns, not significant.
Activating mutations in KIT have been described in acute myeloid leukemia, gastrointestinal stromal tumor, most cases of systemic mastocytosis, and other tumors.10 They result in activation of the receptor in the absence of ligand, playing a critical role in cell proliferation and differentiation. In these tumors where KIT deregulation is the driver oncogenic event, treatment with TKIs has been a successful approach.16 Therefore, we next examined the effect of kinase inhibition in Ba/F3 and ARF-null cell lines expressing G3BP2-KIT. These cells showed sensitivity to a wide range of TKIs with different specificities against KIT and the highest sensitivity to the dual SRC/ABL inhibitor dasatinib (Figure 2C; supplemental Figure 5A). In vitro treatment resulted in cell death and loss of KIT phosphorylation, indicating that constitutive activation of KIT is an important oncogenic signaling pathway for this fusion (supplemental Figure 5B). To confirm the efficacy of tyrosine kinase inhibition on leukemia cells in vivo, the established genetically engineered models of G3BP2-KIT–driven leukemia were randomized to receive dasatinib (20 mg/kg per day by oral gavage; n = 10) or vehicle (n = 10). Dasatinib prolonged survival in the engineered fusion mouse cell models, but it was not curative (Figure 2D).
G3BP2 is a member of the family of Ras-GTPase–activating SH3 domain–binding proteins, which includes 2 additional family members, G3BP1 and G3BP3. These are RNA-binding proteins that are ubiquitously expressed and, under extracellular stresses, bind to untranslated regions of target transcripts and downregulate translation by either degrading the transcript or blocking ribosomal recruitment.17 Recently, a chimeric G3BP1-PDGFRB fusion sensitive to ABL1/PDGFRB kinase inhibition18 was described in a patient with a myeloid neoplasm with eosinophilia.
In conclusion, these results identify KIT rearrangement as a driver oncogenic alteration in leukemia and provide the rationale for therapeutic targeting with TKIs in KIT-rearranged Ph-like B-ALL.
Acknowledgments: The authors thank the Animal Resources Center and the Small Animal Imaging Center, the Compound Management Center, the Department of Chemical Biology & Therapeutics, the Department of Structural Biology for the use of BioXp 3200 system, the Genome Sequencing Facility of the Hartwell Center for Bioinformatics and Biotechnology, and the Flow Cytometry and Cell Sorting core facility of St. Jude Children’s Research Hospital.
This work was supported in part by the American Lebanese Syrian Associated Charities of St. Jude Children’s Research Hospital, the Leukemia and Lymphoma Society Translational Research Program (C.G.M.), a St. Baldrick’s Foundation Robert J. Arceci Innovation Award (C.G.M.), the Henry Schueler 41 & 9 Foundation (C.G.M.), National Cancer Institute, National Institute of Health, Outstanding Investigator Award R35 CA197695 (C.G.M.), and a Research Agreement with Pfizer, Inc. (C.G.M).
Contribution: I.I. designed, directed, and performed research, analyzed data, and wrote the manuscript; R.F., J.D.F., P.B., and E.B. performed research and analyzed data; C.Q. performed genomic analyses; L.J.J. performed histopathology analyses; Y.Z. performed sample processing; P.C. contributed to phosphoproteomic analysis; A.S. contributed to drug screening analysis; A.D.L. and A.S.A contributed to conception and design of clinical sample analyses; C.G.M. edited the manuscript and gave final approval; and all authors critically reviewed the manuscript before submission.
Conflict-of-interest disclosure: I.I. reports honoraria from Amgen and Mission Bio. A.D.L. reports employment and stock and/or other ownership interests in Pfizer. A.S.A. reports consulting for Jazz Pharmaceuticals, Taiho Pharmaceutical, Team Pharmaceuticals, Amgen, Pfizer, Nkarta, Kite Pharmaceuticals, and Glycomimetics and research funding from Servier, Incyte, Amgen, Pfizer, Glycomimetics, Kite Pharmaceuticals, Seattle Genetics, Immunogen, and Macrogenics. C.G.M. reports research funding from AbbVie, Loxo Oncology, and Pfizer; speaking and travel fees from Illumina and Amgen; and stock ownership in Amgen. The remaining authors declare no competing financial interests.
Correspondence: Ilaria Iacobucci, St. Jude Children’s Research Hospital, 262 Danny Thomas Place, Mail Stop 343, Memphis TN 38105; e-mail: ilaria.iacobucci@stjude.org; and Charles G. Mullighan, St. Jude Children’s Research Hospital, 262 Danny Thomas Place, Mail Stop 342, Memphis TN 38105; e-mail: charles.mullighan@stjude.org.
References
Author notes
I.I. and R.F. contributed equally to this study.
I.I. and R.F. are joint first authors.
RNA- and whole-exome sequencing data from mouse models have been deposited in the Sequence Read Archive under BioProject ID PRJNA717917. RNA-sequencing data from the patient have been deposited in the European Genome-phenome Archive under EGAS00001005181.
The full-text version of this article contains a data supplement.


![Genetically engineered mouse models of G3BP2-KIT leukemia. (A) Kaplan-Meier survival curves of C57BL6 mice receiving ARF-null pre-B cell transplants expressing G3BP2-KIT (after 14 days in culture without IL-7 [n = 4] or within 48 hours after transduction [n = 4]) or empty vector (MIG; n = 5) and of secondary recipients undergoing transplantation with cells from primary mouse tumors. Survival comparison was analyzed by log-rank test. (B) Spleen weight from primary and secondary recipient mice with G3BP2-KIT and from control mice (MIG). The mean weight is shown by the horizontal line in the scatter dot plot, and the error bars represent the standard deviations (SDs). (C) Dose-response curve of BaF3 cells expressing empty vector (MIG) or G3BP2-KIT fusion after 72 hours treatment with dasatinib, ponatinib, imatinib, nilotinib, sunitinib, or avapritinib. The cells transduced with the empty vector were grown in media with recombinant mouse IL-3. Values are normalized to dimethyl sulfoxide controls and represent means ± SDs from 3 independent experiments performed in triplicate. (D) Left panel shows levels (means + SDs) of GFP+ cells in the peripheral blood of C57BL6 mice receiving ARF-null pre-B leukemia cell transplants expressing G3BP2-KIT from 2 primary mouse tumor cohorts (a and b groups). In group a (n = 5), dasatinib (20 mg/kg daily) or vehicle (n = 5) was started 1 week after transplantation; in group b, treatment started 2 weeks after transplantation. Right panel shows Kaplan-Meier survival curves of mice in study treatment. IC50, 50% inhibitory concentration; ns, not significant.](https://ash.silverchair-cdn.com/ash/content_public/journal/bloodadvances/6/11/10.1182_bloodadvances.2021004854/2/m_advancesadv2021004854f2.png?Expires=1765886153&Signature=sOkHI~qydhpoth0WXaP2p~sC6KsKEozAszeCe310F344~ywmeXcFMrjvvCwziDyphiMr9RX9xnQzB~MWVKp8EzuoRmvdOH-8xsmUWFQJTcgqxBIGvNNBGijA5NIhMnfPJZcdmkjDEnll4gxMd6FFwCaeOzUka5DaBElwER3etpPQhQMHhLB14eCNnvjGeTbP3OpFOAOhlv-WooZyGjYDk~TkcSN2Ftp01u5tMCgMo1608UnBguHkUcUolLMpbMP0GGj-vhw2hRtq6zHayRriiOaCf~wXWOBs7Dxlsq1y-6K0m6bMwBJX3IsaHw-F1u~lew2vXpS4hQyPU0VV7prhyw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)