Key Points
Cardiovascular toxicities are higher in older patients, but nonrelapse mortality at 100 days and 1 year is comparable to younger patients.
Although mitigation of BEAM toxicities is needed, withholding this potentially curative therapy, based on age only, is not recommended.
High-dose chemotherapy and autologous hematopoietic cell transplantation is an effective consolidation therapy in lymphoma; however, its use in elderly patients has been limited because of concerns for greater toxicity in this group. We investigated the toxicities of carmustine, etoposide, cytarabine, and melphalan (BEAM) and autologous hematopoietic cell transplantation (AHCT) in 346 patients in 2 age groups: 279 patients aged 60 to 69 years and 67 patients aged ≥70 years. The majority developed severe toxicities; the most common were febrile neutropenia, gastrointestinal, infections, and cardiovascular. Older patients were at higher risk for grade ≥3 cardiovascular toxicities (hazard ratio [HR], 3.36; 95% confidence interval [CI], 2.25-5.00; P < .001) and skin toxicities (HR, 2.45; 95% CI, 1.08-5.54, P = .032). In the older group, nonrelapse mortality at 100 days and at 2 years was 2.99% (95% CI, 0.55-9.32) and 6.2% (95% CI, 1.97-13.95), respectively, vs 1.79% (95% CI, 0.68-3.92) and 2.91% (95% CI, 1.37-5.42), respectively, in the younger group. When adjusting for the number of grade ≥3 toxicities within the first 100 days, older patients had a 1.71-fold (95% CI, 1.08-2.71) increased risk for progression or death relative to younger patients. Although BEAM followed by AHCT is effective, it is associated with significant organ toxicities, especially in patients aged ≥70 years. Interventions to mitigate toxicities while maintaining efficacy are much needed.
Introduction
High-dose chemotherapy followed by autologous hematopoietic cell transplantation (HDT-AHCT) is an established therapeutic approach in lymphoma treatment, either as upfront therapy or, most commonly, in the relapsed or refractory setting.1-5 Although effective, some studies in patients ≥60 years old have reported higher rates of nonrelapse mortality (NRM) compared with the younger population.6-12 Furthermore, in patients who are ≥70 years of age, evidence to support HDT-AHCT is limited13-16 because of concerns about the greater toxicity and mortality in this age group. Therefore, it is essential to understand the risks and toxicities of HDT-AHCT in older patients before withholding this potentially curative therapy from this age group who account for a large fraction of lymphoma cases.
BEAM (carmustine, etoposide, cytarabine, and melphalan) has been the most frequently used preparatory regimen for autologous hematopoietic cell transplantation (AHCT) in non-Hodgkin lymphoma (NHL). Although organ toxicities are well-known adverse events associated with BEAM followed by AHCT (BEAM-AHCT) in younger adults, toxicities in older patients, especially those aged ≥70 years are not well characterized. Previously, we reported the outcomes of HDT-AHCT in older patients.15 In this study, we evaluated the incidence and nature of individual BEAM toxicities in a large group of patients (age ≥ 60 years) and compared them in 2 age groups: older (age ≥70 years) and younger (ages 60-69 years). The rationale for selecting these 2 age groups is that most patients with NHL are older than 60 years, the arbitrary cutoff for BEAM-AHCT is age 70 years, and exclusion of patients younger than 60 years minimizes the confounding factors associated with age-related comorbidities.
Patients and methods
This retrospective study was performed in patients ≥60 years of age with NHL who underwent AHCT with BEAM conditioning (carmustine, 300 mg/m2 on day −7; etoposide, 100 mg/m2 every 12 hours on days −6 through −3; cytarabine, 200 mg/m2 every 12 hours on days −6 through −3; and melphalan, 140 mg/m2 on day −2) at Memorial Sloan Kettering Cancer Center (MSKCC) between January of 2000 and December of 2018. The Institutional Review Board at MSKCC approved data collection. Data were primarily extracted from comprehensive chart review and institutional databases. The study was conducted in accordance with the Declaration of Helsinki. Date of diagnosis, disease histology at the time of transplant, number of prior lines of therapies, and disease status at transplantation were extracted for each patient. Patients who underwent BEAM-AHCT for transformation of a prior indolent lymphoma to diffuse large B-cell lymphoma (DLBCL) are listed as having DLBCL. Complete remission (CR) was defined as the absence of radiological evidence of lymphoma on positron emission tomography, computed tomography, and/or magnetic resonance imaging. Partial remission (PR) was defined as ≥50% reduction in lymphoma mass, as assessed by imaging. Pretransplant studies included physical examination, complete blood count, metabolic panel, chest radiography, computed tomography of the chest, abdomen, and pelvis, 18F-deoxyglucose positron emission tomography, bone marrow aspiration and biopsy, echocardiography, and pulmonary function testing. The hematopoietic cell transplantation comorbidity index (HCT-CI)17 was calculated retrospectively through chart review of data immediately pre–BEAM-AHCT. HCT-CI was categorized as low risk (0 points), intermediate risk (1-2 points), or high risk (≥3 points). Peripheral blood hematopoietic cells were collected by leukapheresis following mobilizing cytokines, with or without chemotherapy priming.18-21 The minimal acceptable progenitor cell dose was 2 × 106 CD34+ cells per kilogram. The day of hematopoietic cell infusion was considered day 0. Patients received filgrastim, 5 μg/kg per day, from day +5 until neutrophil recovery (before 2006) or pegfilgrastim, 6 mg on day +1 (since 2006). All patients were hospitalized for HDT-AHCT until engraftment that was not supported by transfusions and adequate gastrointestinal (GI) recovery. Neutrophil engraftment was defined as the first day of 3 consecutive days of an absolute neutrophil count > 500 × 106/L. Disease response was determined by the criteria in use at the time of treatment and abstracted from the medical chart. Data cutoff for follow-up was April of 2020. Patients were kept in single rooms and were managed clinically according to MSKCC standard guidelines, including infection prophylaxis. Using the Common Terminology Criteria for Adverse Events version 5, grade 3-5 nonhematologic toxicities were retrospectively collected from admission through day +100 in all patients. For validation, another expert separately graded a randomly selected sample (20%) of patients in the toxicity review process. Individual toxicities were organized into 91 toxicity categories and further into 17 organ-based groups, as previously described (supplemental Table 1).22 Transplant-related toxicities and outcomes were analyzed and compared in 2 cohorts by age: ≥70 years and 60 to 69 years.
Statistical analysis
Descriptive statistics were used to summarize patient characteristics. Overall survival (OS) was calculated from the date of transplantation to death or censored at the time of last clinical evaluation. Progression-free survival (PFS) was calculated from the date of transplantation to disease progression or death from any cause. Kaplan-Meier method was used to estimate the median PFS and OS along with survival probabilities at 1, 2, and 5 years. The hazard ratios (HRs) for older patients relative to younger patients were also estimated for OS and PFS using Cox regression. Cumulative incidence functions were used to estimate the cumulative incidence of progression of disease and NRM, with NRM and progression as competing events. NRM probabilities were calculated at 100 days and at 1 and 2 years after AHCT. Univariate Cox regression analyses of the baseline factors (age, Karnofsky performance status [KPS], HCT-CI score, disease histology, number of prior regimens, and disease status at transplantation) and of the 17 toxicity groups were conducted to examine the relationships between each factor and the 3 outcomes: NRM, PFS, and OS. Each of the toxicities was treated as a time-dependent covariate. The incidences of the most common grade ≥ 3 toxicities were estimated using cumulative incidence functions. The risk of each toxicity was assessed using univariable Cox regression for select baseline factors. To further evaluate the relationship among age, survival, and the number of toxicities, we conducted a landmark multivariate analysis for each of the 3 outcomes (NRM, PFS, and OS) with the 2 covariates (age group and whether patients had developed more grade ≥3 toxicities than the median number for all patients). Analyses were performed using R statistical software version 4.0.1 (R Foundation for Statistical Computing, Vienna, Austria).23
Results
Patient characteristics
A total of 346 patients aged ≥60 years who had received BEAM-AHCT for NHL between 2000 and 2018 were identified. The median age at the time of AHCT was 65.8 years (range, 60-77). The majority (66%) were male. Histologies included DLBCL in 136 (39.3%), mantle cell lymphoma (MCL) in 119 (34.4%), T-cell NHL in 61 (17.6%), and follicular lymphoma (FL) in 21 (6.1%). Nine patients (2.6%) had other lymphoma subtypes. Upfront consolidative AHCT was performed in 179 (51.7%) patients. Two or more lines of treatment prior to transplant were received by 189 (54.6%) patients. Thirty-seven patients (10.47%) had received prior radiation therapy. The most common induction regimen was cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) or a CHOP-like regimen (n = 302), and the most common salvage therapy was ifosfamide, carboplatin, and etoposide (ICE) (n = 109). Thirty patients received oxaliplatin prior to BEAM-AHCT as part of a gemcitabine/oxaliplatin or a dexamethasone/cytarabine/oxaliplatin regimen. Eighty percent of patients had received rituximab with their conditioning regimen. All patients had chemosensitive disease (295 in CR; 51 in PR). High-risk HCT-CI (≥3) was noted in 196 patients (57%). KPS ≥90 was noted in 212 (62%) patients (Table 1). Sixty-seven patients (19.4%) aged ≥70 years were compared with 279 patients (80.6%) aged 60 to 69 years (Table 1). The median age was 71.8 years (range, 70-77) in the older group and 64.8 years (range, 60-69.9) in the younger group. The 2 age groups were balanced with respect to histology distribution, number of prior lines of therapy, remission status, HCT-CI, KPS, albumin, and stem cell dose. The median cell dose in the older vs the younger group was 4.8 × 106 CD34+ cells per kilogram (range, 1.7-9.8) vs 5.2 × 106 CD34+ cells per kilogram (range 1.9-14.7), respectively. The median follow-up time calculated using the reverse Kaplan-Meier method was 6.55 years (95% CI, 5.73-7.37).
Patient baseline characteristics
| Patient characteristics . | Total . | Age 60 to 69 y . | Age ≥70 y . | p . |
|---|---|---|---|---|
| Sample size, n (%) | 346 (100) | 279 (80.6) | 67 (19.4) | |
| Females, n (%) | 118 (34) | 97 (34) | 21 (31) | .6 |
| Age at HCT, median (range), y | 65.8 (60-77) | 64.8 (60-69.9) | 71.8 (70-77) | <.001 |
| Histology and status at HCT, n (%) | .7* | |||
| DLBCL | 136 (39) | 110 (39) | 26 (38) | |
| CR1 | 5 (4) | 5 (4.5) | 0 | |
| ≥CR2 | 102 (75) | 82 (74.5) | 20 (77) | |
| PR | 29 (21) | 23 (21) | 6 (23) | |
| MCL | 119 (34) | 99 (35) | 20 (30) | |
| CR1 | 95 (80) | 80 (81) | 15 (75) | |
| ≥CR2 | 13 (11) | 10 (10) | 3 (15) | |
| PR | 11 (9) | 9 (9) | 2 (10) | |
| FL | 21 (6.1) | 17 (6.1) | 4 (6.0) | |
| CR1 | 0 | 0 | 0 | |
| ≥CR2 | 16 (76) | 12 (71) | 4 (100) | |
| PR | 5 (24) | 5 (30) | 0 | |
| TCL | 61 (18) | 47 (17) | 14 (21) | |
| CR1 | 52 (85) | 40 (85) | 12 (86) | |
| CR2 | 3 (5) | 3 (7) | 0 | |
| PR | 6 (10) | 4 (8) | 2 (14) | |
| Other NHL | 9 (2.6) | 6 (2.2) | 3 (4.5) | |
| CR1 | 3 (33) | 2 (33) | 1 (33) | |
| ≥CR2 | 6 (67) | 4 (67) | 2 (66) | |
| Transplant in first remission | 179 (52) | 145 (52) | 34 (51) | .9 |
| Lines of therapy | .2 | |||
| 1 | 157 (45) | 127 (46) | 30 (45) | |
| 2 | 148 (43) | 115 (41) | 33 (49) | |
| ≥3 | 41 (12) | 37 (13) | 4 (6.0) | |
| Prior rituximab | 278 (80) | 225 (81) | 53 (79) | .8 |
| History of radiation | 37 (11) | 33 (12) | 4 (6.0) | .2 |
| HCT-CI, median (range) | 3 (0-10) | 3 (0-10) | 3 (0-10) | .4† |
| Low (0) | 61 (17) | 48 (17) | 13 (19) | |
| Intermediate (1-2) | 89 (26) | 75 (27) | 14 (21) | |
| High (≥3) | 196 (57) | 156 (56) | 40 (60) | |
| KPS | >.9 | |||
| ≥90 | 212 (62) | 171 (62) | 41 (62) | |
| <90 | 130 (38) | 105 (38) | 25 (38) | |
| Missing | 4 (1.1) | 3 (1.07) | 1 (1.5) | |
| Albumin (g/dL) at HCT, median (range) | 4.1 (2.8-5) | 4.1 (2.8-5) | 4.1 (3.7-4.7) | .6 |
| Absolute lymphocyte count (103/μL), median (range) | 0.5 (0.1-2.8) | 0.5 (0.1-2.8) | 0.4 (0.1-1.6) | .6 |
| Stem cell dose (CD34/kg (×106), median (range) | 5.05 (1.70-14.70) | 5.16 (1.90-14.70) | 4.80 (1.70-9.80) | .3 |
| Patient characteristics . | Total . | Age 60 to 69 y . | Age ≥70 y . | p . |
|---|---|---|---|---|
| Sample size, n (%) | 346 (100) | 279 (80.6) | 67 (19.4) | |
| Females, n (%) | 118 (34) | 97 (34) | 21 (31) | .6 |
| Age at HCT, median (range), y | 65.8 (60-77) | 64.8 (60-69.9) | 71.8 (70-77) | <.001 |
| Histology and status at HCT, n (%) | .7* | |||
| DLBCL | 136 (39) | 110 (39) | 26 (38) | |
| CR1 | 5 (4) | 5 (4.5) | 0 | |
| ≥CR2 | 102 (75) | 82 (74.5) | 20 (77) | |
| PR | 29 (21) | 23 (21) | 6 (23) | |
| MCL | 119 (34) | 99 (35) | 20 (30) | |
| CR1 | 95 (80) | 80 (81) | 15 (75) | |
| ≥CR2 | 13 (11) | 10 (10) | 3 (15) | |
| PR | 11 (9) | 9 (9) | 2 (10) | |
| FL | 21 (6.1) | 17 (6.1) | 4 (6.0) | |
| CR1 | 0 | 0 | 0 | |
| ≥CR2 | 16 (76) | 12 (71) | 4 (100) | |
| PR | 5 (24) | 5 (30) | 0 | |
| TCL | 61 (18) | 47 (17) | 14 (21) | |
| CR1 | 52 (85) | 40 (85) | 12 (86) | |
| CR2 | 3 (5) | 3 (7) | 0 | |
| PR | 6 (10) | 4 (8) | 2 (14) | |
| Other NHL | 9 (2.6) | 6 (2.2) | 3 (4.5) | |
| CR1 | 3 (33) | 2 (33) | 1 (33) | |
| ≥CR2 | 6 (67) | 4 (67) | 2 (66) | |
| Transplant in first remission | 179 (52) | 145 (52) | 34 (51) | .9 |
| Lines of therapy | .2 | |||
| 1 | 157 (45) | 127 (46) | 30 (45) | |
| 2 | 148 (43) | 115 (41) | 33 (49) | |
| ≥3 | 41 (12) | 37 (13) | 4 (6.0) | |
| Prior rituximab | 278 (80) | 225 (81) | 53 (79) | .8 |
| History of radiation | 37 (11) | 33 (12) | 4 (6.0) | .2 |
| HCT-CI, median (range) | 3 (0-10) | 3 (0-10) | 3 (0-10) | .4† |
| Low (0) | 61 (17) | 48 (17) | 13 (19) | |
| Intermediate (1-2) | 89 (26) | 75 (27) | 14 (21) | |
| High (≥3) | 196 (57) | 156 (56) | 40 (60) | |
| KPS | >.9 | |||
| ≥90 | 212 (62) | 171 (62) | 41 (62) | |
| <90 | 130 (38) | 105 (38) | 25 (38) | |
| Missing | 4 (1.1) | 3 (1.07) | 1 (1.5) | |
| Albumin (g/dL) at HCT, median (range) | 4.1 (2.8-5) | 4.1 (2.8-5) | 4.1 (3.7-4.7) | .6 |
| Absolute lymphocyte count (103/μL), median (range) | 0.5 (0.1-2.8) | 0.5 (0.1-2.8) | 0.4 (0.1-1.6) | .6 |
| Stem cell dose (CD34/kg (×106), median (range) | 5.05 (1.70-14.70) | 5.16 (1.90-14.70) | 4.80 (1.70-9.80) | .3 |
Unless otherwise noted, data are n (%).
Corresponds to histology.
Corresponds to the continuous version for HCT-CI.
Transplant outcomes
Engraftment.
The median days to neutrophil and platelet engraftment was 10 and 21 days, respectively, in both age groups. One patient in each age group was not evaluable for neutrophil or platelet engraftment. One died on day +4 from infection, and the other died on day +10 from multiorgan failure before engraftment. The median duration of hospital stay was 22 days in both age groups.
Toxicities.
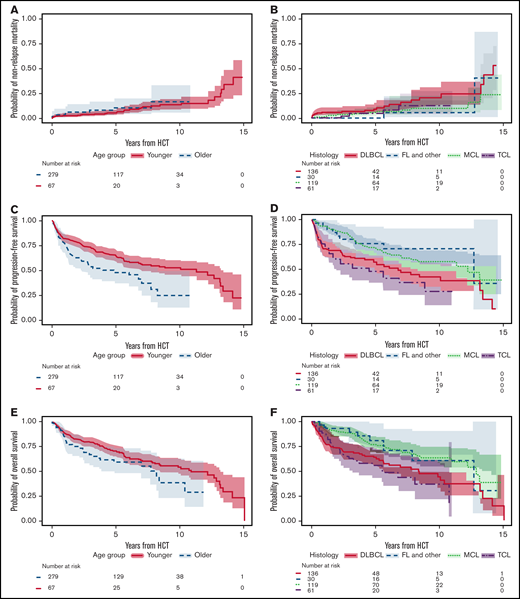
Table 2 shows the number of individual grade ≥3 toxicities related to BEAM-AHCT from the start of conditioning (day −7) through day 100 post-AHCT (day +100); Table 3 shows the number of patients with these toxicities. All patients in the older group had ≥1 grade ≥3 toxicity, with a median of 3 (range, 1-13) toxicities per patient. In the younger cohort, the median toxicity per patient was 2 (range 0-10); 23 patients (8%) did not experience any grade ≥3 toxicities. In 346 patients, a total of 874 grade ≥3 toxicities were observed: 250 toxicities in 67 older patients vs 624 toxicities in 256 younger patients. Of the 874 grade ≥3 toxicities, 780 (89%) occurred within day −7 to day 30 post-AHCT. The most prevalent toxicities were febrile neutropenia in 218 (63%) patients, oral/GI in 178 (51%) patients, infection in 108 (31%) patients, and cardiovascular in 99 (29%) patients. Mucositis and enterocolitis/diarrhea were the most common oral/GI toxicities. No patient developed sinusoidal obstruction syndrome. Bloodstream bacterial infections and pneumonia were the most common infections noted. The most common cardiovascular toxicities included syncope, arrhythmia, and hypertension. Figure 1 summarizes the details of toxicities in the entire cohort with the comparison of the 4 most common toxicities in the 2 age groups. Figure 2 shows the cumulative incidence estimates for the 4 most common grade ≥3 toxicities at 100 days posttransplant, separated by age group. Although the rate of all toxicities was higher in the older cohort, the risk of cardiovascular toxicity (HR, 3.36; 95% CI, 2.25-5.00; P < .001) and skin toxicity (HR, 2.45; 95% CI, 1.08-5.54; P = .032) was markedly higher in older patients, as indicated by univariable regression analysis. Even after adjusting for the presence of baseline cardiac risk factors (arrhythmia, coronary artery disease, congestive heart failure, low ejection fraction, and heart valve disease), the age group was still statistically significant, with an HR for cardiovascular toxicity of 3.39 (95% CI, 2.26-5.10) for older vs younger patients. Furthermore, a higher risk for cardiovascular toxicity was observed for patients with lower than the median absolute lymphocyte count (ALC) (0.5 K/μL; HR, 1.63; 95% CI, 1.11-2.39) and lower than the median albumin value pre-AHCT (4.10 g/dL; HR, 1.80; 95% CI, 1.23-2.63). The ALC was not associated with the number of lines of prior therapy. There was no association between the lines of prior therapy and risk for cardiovascular toxicity in univariable analysis. A multivariable analysis of cardiovascular toxicity on ALC group while adjusting for the number of lines of prior therapy showed that the ALC group remained statistically significant, whereas the lines of prior therapy remained statistically insignificant. A higher risk for oral/GI toxicity (HR, 1.75; 95% CI, 1.30-2.35) was observed in patients with a cell count below the median (5.05 × 106 CD34 cells per kilogram) compared with patients with a cell count above the median. Time to engraftment was similar in the 2 cell dose groups. Male sex and those with KPS ≥90 were at lower risk for oral/GI events. Figure 3 shows the cumulative incidence of the 4 most common toxicities with respect to histology. Figure 4 shows the cumulative incidence of the 4 most common toxicities with respect to the number of lines of prior treatment.
Toxicity distribution. Distributions of individual toxicities in the entire cohort (A), oral/GI toxicities (B), infections (C), and cardiovascular toxicities (D).
Toxicity distribution. Distributions of individual toxicities in the entire cohort (A), oral/GI toxicities (B), infections (C), and cardiovascular toxicities (D).
Cumulative incidence function for the 4 most common grade ≥3 toxicities stratified by age group.
Cumulative incidence function for the 4 most common grade ≥3 toxicities stratified by age group.
Incidence of toxocities with respect to histology. Cumulative incidence of febrile neutropenia (A), oral/GI (B), infections (C), and cardiovascular toxicities (D) with respect to histology.
Incidence of toxocities with respect to histology. Cumulative incidence of febrile neutropenia (A), oral/GI (B), infections (C), and cardiovascular toxicities (D) with respect to histology.
Incidence of toxicities with respect to number of lines of therapy. Cumulative incidence of febrile neutropenia (A), oral/GI (B), infections (C), and cardiovascular toxicities (D) with respect to the number of lines of therapy.
Incidence of toxicities with respect to number of lines of therapy. Cumulative incidence of febrile neutropenia (A), oral/GI (B), infections (C), and cardiovascular toxicities (D) with respect to the number of lines of therapy.
Number of individual grade ≥3 toxicities (day −7 to day +100 post-AHCT)
| . | Total . | Age 60-69 y . | Age ≥70 y . |
|---|---|---|---|
| Sample size | 346 (100) | 279 (80.6) | 67 (19.4) |
| Number of individual grade ≥ 3 toxicities day −7 to day +100 post-HCT | 874 (100) | 624 (100) | 250 (100) |
| Febrile neutropenia (no source) | 220 (25) | 172 (28) | 48 (19) |
| Infection | 157 (18) | 123 (20) | 34 (14) |
| Oral/GI | 193 (22) | 147 (24) | 46 (18) |
| Cardiovascular | 136 (16) | 78 (12) | 58 (23) |
| Pulmonary | 44 (5.0) | 32 (5.1) | 12 (4.8) |
| Metabolic | 37 (4.2) | 24 (3.8) | 13 (5.2) |
| Skin | 24 (2.7) | 15 (2.4) | 9 (3.6) |
| Neurologic | 18 (2.1) | 11 (1.8) | 7 (2.8) |
| Renal | 11 (1.3) | 8 (1.3) | 3 (1.2) |
| Hematologic | 12 (1.4) | 5 (0.8) | 7 (2.8) |
| Hepatic | 5 (0.6) | 4 (0.6) | 1 (0.4) |
| Immune | 3 (0.3) | 1 (0.2) | 2 (0.8) |
| Musculoskeletal and connective tissue | 5 (0.6) | 3 (0.5) | 2 (0.8) |
| Other | 9 (0.9) | 1 (0.2) | 8 (3.2) |
| . | Total . | Age 60-69 y . | Age ≥70 y . |
|---|---|---|---|
| Sample size | 346 (100) | 279 (80.6) | 67 (19.4) |
| Number of individual grade ≥ 3 toxicities day −7 to day +100 post-HCT | 874 (100) | 624 (100) | 250 (100) |
| Febrile neutropenia (no source) | 220 (25) | 172 (28) | 48 (19) |
| Infection | 157 (18) | 123 (20) | 34 (14) |
| Oral/GI | 193 (22) | 147 (24) | 46 (18) |
| Cardiovascular | 136 (16) | 78 (12) | 58 (23) |
| Pulmonary | 44 (5.0) | 32 (5.1) | 12 (4.8) |
| Metabolic | 37 (4.2) | 24 (3.8) | 13 (5.2) |
| Skin | 24 (2.7) | 15 (2.4) | 9 (3.6) |
| Neurologic | 18 (2.1) | 11 (1.8) | 7 (2.8) |
| Renal | 11 (1.3) | 8 (1.3) | 3 (1.2) |
| Hematologic | 12 (1.4) | 5 (0.8) | 7 (2.8) |
| Hepatic | 5 (0.6) | 4 (0.6) | 1 (0.4) |
| Immune | 3 (0.3) | 1 (0.2) | 2 (0.8) |
| Musculoskeletal and connective tissue | 5 (0.6) | 3 (0.5) | 2 (0.8) |
| Other | 9 (0.9) | 1 (0.2) | 8 (3.2) |
All data are n (%).
Number of patients with grade ≥3 toxicities (day −7 to day +100)
| . | Total . | Age 60-69 (%) . | Age ≥70 (%) . |
|---|---|---|---|
| Sample size | 346 (100) | 279 (80.6) | 67 (19.4) |
| Number of patients with grade ≥ 3 toxicities day −7 to day 100 post-HCT | 323 (93) | 256 (92) | 67 (100) |
| Febrile neutropenia (no source) | 218 (63) | 171 (61) | 47 (70) |
| Infection | 108 (31) | 85 (30) | 23 (34) |
| Oral/GI | 178 (51) | 139 (50) | 39 (58) |
| Cardiovascular | 99 (29) | 61 (22) | 38 (57) |
| Pulmonary | 39 (11) | 29 (10) | 10 (15) |
| Metabolic | 34 (9.8) | 22 (7.9) | 12 (17.9) |
| Skin | 24 (6.9) | 15 (5.4) | 9 (13) |
| Neurologic | 15 (4.3) | 10 (3.6) | 5 (7.5) |
| Renal | 11 (3.2) | 8 (2.9) | 3 (4.5) |
| Hematologic | 12 (3.5) | 5 (1.8) | 7 (10.4) |
| Hepatic | 5 (1.4) | 4 (1.4) | 1 (1.5) |
| Immune | 3 (0.9) | 1 (0.4) | 2 (3.0) |
| Musculoskeletal and connective tissue | 5 (1.4) | 3 (1.1) | 2 (3.0) |
| Other | 6 (1.7) | 0 | 6 (9.0) |
| No. of individual grade ≥ 3 toxicities per patient, median (range) | 2 (0-13) | 2 (0-10) | 3 (1-13) |
| . | Total . | Age 60-69 (%) . | Age ≥70 (%) . |
|---|---|---|---|
| Sample size | 346 (100) | 279 (80.6) | 67 (19.4) |
| Number of patients with grade ≥ 3 toxicities day −7 to day 100 post-HCT | 323 (93) | 256 (92) | 67 (100) |
| Febrile neutropenia (no source) | 218 (63) | 171 (61) | 47 (70) |
| Infection | 108 (31) | 85 (30) | 23 (34) |
| Oral/GI | 178 (51) | 139 (50) | 39 (58) |
| Cardiovascular | 99 (29) | 61 (22) | 38 (57) |
| Pulmonary | 39 (11) | 29 (10) | 10 (15) |
| Metabolic | 34 (9.8) | 22 (7.9) | 12 (17.9) |
| Skin | 24 (6.9) | 15 (5.4) | 9 (13) |
| Neurologic | 15 (4.3) | 10 (3.6) | 5 (7.5) |
| Renal | 11 (3.2) | 8 (2.9) | 3 (4.5) |
| Hematologic | 12 (3.5) | 5 (1.8) | 7 (10.4) |
| Hepatic | 5 (1.4) | 4 (1.4) | 1 (1.5) |
| Immune | 3 (0.9) | 1 (0.4) | 2 (3.0) |
| Musculoskeletal and connective tissue | 5 (1.4) | 3 (1.1) | 2 (3.0) |
| Other | 6 (1.7) | 0 | 6 (9.0) |
| No. of individual grade ≥ 3 toxicities per patient, median (range) | 2 (0-13) | 2 (0-10) | 3 (1-13) |
Unless otherwise noted, all data are n (%).
Survival.
The median follow-up time calculated using the reverse Kaplan-Meier method was 6.55 years (95% CI, 5.73-7.27); 137 of the 346 patients died. Causes of death were relapse or progression of disease (n = 96), second primary malignancy (n = 17) (solid tumors, n = 12; myelodysplastic syndrome and acute myeloid leukemia (n = 4), and cardiac amyloid, n = 1), infection (n = 8), organ failure (n = 2), complications of a subsequent allogeneic HCT (n = 5), neurologic event (n = 1), cardiac event (n = 1), and unknown (n = 7). A total of 3 patients in the older group and 5 patients in the younger group died from infection. The cause of death between 1 and 2 years posttransplant was relapse in all younger patient vs relapse (60%), infection (20%), and secondary malignancy (20%) in older patients. The median PFS and OS for the entire cohort were 8.32 years (95% CI, 6.10-13.22) and 10.45 years (95% CI, 8.24-13.22), respectively. Figure 5 shows NRM, PFS, and OS by age group, as well as by histology subtypes. In the older group, NRM at 100 days and at 1 and 2 years was 2.99% (95% CI, 0.55-9.32), 2.99% (95% CI, 0.55-9.32), and 6.2% (95% CI, 1.97-13.95), respectively, vs 1.79% (95% CI, 0.68-3.92), 2.51% (95% CI, 1.12-4.88), and 2.91% (95% CI, 1.37-5.42), respectively, in the younger group. From the univariate regression analysis, the risk of NRM in MCL patients was lower relative to patients with DLBCL (HR, 0.40; 95% CI, 0.19-0.84; P = .015). Using a univariate Cox regression model, patients with MCL were at lower risk for progression or death (HR, 0.55; 95% CI, 0.38-0.81; P = .002) relative to patients with DLBCL, as were patients with indolent histology (FL and others) (HR, 0.46; 95% CI, 0.23-0.92; P = .028). Furthermore, the risk of death was lower in MCL patients relative to patients with DLBCL (HR, 0.52; 95% CI, 0.35-0.79; P = .002). MCL patients had a lower risk for relapse (HR, 0.62; 95% CI, 0.40-0.98; P = .04). Univariable regression analysis of the older cohort showed that patients in PR did not have a statistically significant higher risk for death (HR, 2.26; 95% CI, 0.96-5.32), relapse (HR, 2.21; 95% CI, 0.94-5.22), or death or progression (HR, 1.71; 95% CI, 0.74-3.94) compared with those in CR.
Non-relapse mortaity and Kaplan-Meier (KM) survival curves by age and histology. Cumulative incidence of NRM stratified by age group (A) and histology (B). Kaplan-Meier curves for PFS stratified by age group (C) and histology (D). Kaplan-Meier curves for OS stratified by age group (E) and histology (F).
Non-relapse mortaity and Kaplan-Meier (KM) survival curves by age and histology. Cumulative incidence of NRM stratified by age group (A) and histology (B). Kaplan-Meier curves for PFS stratified by age group (C) and histology (D). Kaplan-Meier curves for OS stratified by age group (E) and histology (F).
Discussion
Studies have demonstrated the overall safety of HDT-AHCT in select older patients with NHL.15,16,24 However, there remains a paucity of data on the individual risks and toxicities of HDT-AHCT in older patients.16 To our knowledge, this is the most comprehensive assessment of BEAM-AHCT toxicities in a large group of NHL patients older than 60 years. We categorized the patients into 2 groups (60-69 years and ≥70 years), in which histology, prior lines of therapy, HCT-CI, KPS, stem cell dose, albumin, and remission status had similar distributions. Many of our patients (196/346, 57%) had high-risk HCT-CI, with a median HCT-CI score of 3. Other studies have reported much lower comorbidity scores in elderly patients undergoing AHCT. In the study by Hermet et al, 73% of transplanted elderly patients had an HCT-CI score of 0,14 and in Hosing et al’s study, only 36% had an HCT-CI > 2.25 This shows that age and comorbidities have continued to be the main factors in selecting patients for AHCT. In our study, like in some previously published reports, HCT-CI score did not have a prognostic impact on survival or toxicity outcome.14,16,25 However, the prognostic utility has been shown in larger cohorts from the Center for International Blood and Marrow Transplant Research study.26
Almost all patients (93%) developed ≥1 transplant-related toxicity that was grade ≥3. The most common grade ≥3 toxicities were febrile neutropenia, infection, and oral/GI, cardiovascular, and pulmonary toxicities. A higher percentage of older patients developed grade ≥3 toxicities within 100 days posttransplant compared with younger patients. All older patients had ≥1 toxicity that was grade ≥3 vs 92% of younger patients. Older patients were at significantly higher risk for cardiovascular and skin toxicities than were their younger counterparts. Older age (≥70 years) was associated with a higher probability of toxicities and disease progression or relapse. When adjusting for the number of toxicities, older patients had a 2.04-fold (95% CI, 1.29-3.23) higher risk for relapse. We also analyzed the association between the number of lines of prior treatment and each of the most common toxicities. Patients who were heavily pretreated with ≥2 lines of treatment were at a significantly higher risk for oral/GI toxicity relative to patients with only 1 line of treatment (HR, 1.35; 95% CI, 1.01-1.81). Most MCL patients underwent AHCT in first CR and had a lower probability of progression in the first 5 years of the follow-up period. When the 2 largest NHL subtypes were compared, patients with MCL had a statistically significant lower risk for death, progression or death, and NRM relative to patients with DLBCL. Previously published studies of HDT-AHCT in older lymphoma patients reported 100-day NRM ranging from 4% to 18%.11,24,27,28 The 100-day cumulative incidence of NRM for our patient population was 2.02% (95% CI, 0.90-3.95). When separated by age group, it was 2.99% (95% CI, 0.55-9.32) in the older cohort vs 1.79% (95% CI, 0.68-3.92) in the younger cohort. In conclusion, within this cohort of elderly patients with NHL undergoing BEAM-AHCT with extended follow-up, we demonstrate feasibility and efficacy in patients as old as 77 years of age, but with a high toxicity burden. Although these results support the use of HDT-AHCT in the older population overall, interventions to minimize toxicities while maintaining efficacy, especially in patients older than 70 years, are crucial. A strategy that is currently under investigation (ClinicalTrials.gov #NCT03352765) is using an alternative conditioning regimen in older patients with comorbidities. Furthermore, incorporation of additional screening with geriatric assessments or newer tools may help to risk stratify older patients. Tucci et al showed that integrating comprehensive geriatric assessment into the initial evaluation of elderly patients with aggressive NHL was efficient in identifying patients who can benefit from a curative approach.29 National Comprehensive Cancer Network guidance has also suggested the integration of comprehensive geriatric assessment in transplant assessment, acknowledging the need for prospective studies.30
Limitations of this study are primarily related to its retrospective nature. Inclusion of individual toxicities could only occur if there were appropriate documentation in the medical record. Although it is likely that most severe toxicities are captured, concurrent toxicities in prolonged complicated hospital stays may have been over- or underestimated when collected retrospectively. As a quality control for our collection, we conducted cross-reviews between abstractors and corrected discrepancies as they arose. In addition, we incorporated all objective data, such as laboratory values and culture results, to confirm the inclusion of relevant toxicities. We also acknowledge the sample size as a limitation, although this is 1 of the largest series of older patients treated with BEAM-AHCT. The small number of patients included in this study overall, and specifically in the group that was ≥70 years old, could have contributed to the lack of demonstrable prognostic impact of baseline factors, such as KPS, HCT-CI score, disease histology, number of prior regimens, and disease status at transplantation, on BEAM-AHCT toxicities and outcomes. Furthermore, our study incorporated only those patients who underwent AHCT; therefore, it does not describe an intent-to-treat analysis of older NHL patients who did not undergo AHCT because of age, comorbidity, toxicity, or disease progression. No patient in the study was older than 77 years; thus, we cannot draw conclusions regarding the safety of BEAM-AHCT in patients beyond this age. This study, while reaffirming the survival benefit associated with BEAM-AHCT in older NHL patients, establishes a benchmark to focus efforts on reducing toxicity burden and improving outcomes in older patients.
Acknowledgments
This work was supported in part by an Internal Diversity Enhancement Award and by National Institutes of Health National Cancer Institute grants P01 CA23766 and P30 CA008748.
Authorship
Contribution: P.B.D. and S.A.G. conceived and designed the study; J.R., M.M., C.R.-C., E. Petrlik, and P.B.D. collected and assembled data; J.L., S.M.D., and P.B.D. analyzed and interpreted data; and all authors provided study material or patients, wrote and approved the final version of the manuscript, and are accountable for all aspects of the work.
Conflict-of-interest disclosure: P.B.D. has served on an advisory board for Kite (Gilead). M.-A.P. has received honoraria from AbbVie, Bellicum, Bristol Myers Squibb, Incyte, Merck, Novartis, Nektar Therapeutics, and Takeda; serves on Data Safety Monitoring Boards for Servier and Medigene and on scientific advisory boards for MolMed and NexImmune; and has received research support for clinical trials from Incyte, Kite (Gilead), and Miltenyi Biotec. C.S.S. has served as a paid consultant on advisory boards for Juno Therapeutics, Sanofi-Genzyme, Spectrum Pharmaceuticals, Novartis, Genmab, Precision Biosciences, Kite (Gilead), Celgene, Gamida Cell, and GSK and has received research funds for clinical trials from Juno Therapeutics, Celgene, Bristol Myers Squibb, Precision Biosciences, and Sanofi-Genzyme. S.A.G. has served as a consultant for Amgen, Actinium, Celgene, Johnson & Johnson, Jazz Pharmaceuticals, Takeda, Novartis, Kite (Gilead), and Spectrum Pharma and has received research funding from Amgen, Actinium, Celgene, Johnson & Johnson, Miltenyi Biotec, Takeda, Quintiles, Pfizer, CLS Behring, Sanofi, and Adienne. M.S. has served as a consultant for McKinsey & Company, Angiocrine Bioscience, Inc., and Omeros Corporation; has received research funding from Angiocrine Bioscience, Inc.; and has served on an ad hoc advisory board for Kite (Gilead). The remaining authors declare no competing financial interests.
Correspondence: Parastoo B. Dahi, MSK Koch, 530 E. 74th St, New York, NY 10021; e-mail: dahip@mskcc.org.
References
Author notes
Data sharing requests should be sent to Parastoo B. Dahi (dahip@mskcc.org).
The full-text version of this article contains a data supplement.