Amyloidosis is characterized by the conversion of soluble proteins into highly ordered cross-β-fibrils. The process of amyloid formation, starting from the misfolded protein to prefibrillar aggregates and finally fibrils, results in cellular stress and death and subversion of the normal tissue architecture, leading to organ dysfunction, and eventually death, if the disease is untimely recognized or treated ineffectively.1 The process of amyloid formation is accelerated by the presence of preformed fibrils, which can capture and catalyze the conversion of monomeric precursors, even at very low concentrations, into misfolded, toxic, and aggregation-competent structures.1,2 The minimum concentration of the amyloid precursor necessary to fuel the amyloid process varies from protein to protein, and it is practically impossible to determine the amyloidogenic potential of each protein in the clinical setting. In light chain (AL) amyloidosis, increased concentrations of the amyloid precursor protein consistently precede the clinical manifestations of disease with organ dysfunction.3 Thus, in AL amyloidosis, it is essential to reduce the concentration of the amyloid precursor protein as quickly, deeply, and durably as possible. A large international effort has defined and validated criteria for response to therapy.4 Because the disease is caused by the amyloidogenic light chain, the criteria for hematologic response are based on the degree of reduction of this parameter. Profound reductions of the difference between involved and uninvolved free light chains (dFLC) concentration to ≤40 mg/L define a very good partial response (VGPR) and are associated with significant survival extension; thus, achieving at least a VGPR is the aim of therapy.4 However, the maximum benefit in terms of likelihood of organ, particularly cardiac, response, and extended survival, is achieved with complete hematologic response (ie, the disappearance of the amyloid light chain from serum and urine using standard methods such as serum and urine immunofixation and measurement of the serum free light chains [FLC] concentration).4 Nonetheless, the production of amyloid light chains at very low concentrations, undetectable by standard methods, can still hamper organ recovery and contribute to further organ deterioration. Recently, the Mayo Clinic investigators reported that the presence of any residual monotypic plasma cells by routine flow cytometry is associated with inferior progression-free survival in patients who achieve a complete response (CR) by standard criteria. They reported that the likelihood of achieving organ response with time is lower in patients with residual monotypic plasma cells, who have a minimal but continuous production of the amyloid light chain, than in those without any residual monotypic cells.5 Improvement in cardiac response was observed in 33% of patients in hematologic CR and without monotypic plasma cells compared with 15% of patients with residual monotypic plasma cells (P = .003). Similarly, renal response was achieved in 19% of patients without residual monotypic plasma cells vs 3% in those with a persistent monoclonal plasma cell population (P = .02). Using next-generation flow cytometry (Euroflow protocol) to determine the minimal residual disease (MRD), we found that 68% of AL patients who have a hematologic CR are MRD negative. Cardiac or renal responses were more likely in patients who were MRD negative.6 These data further support that even trace production of amyloid protein impairs improvement of the targeted organs and may further worsen end organ dysfunction. In this context, attempts to further improve depth of response should be considered. There is agreement that upfront chemotherapy can be prolonged for 2 cycles after best hematological response has been achieved.7 Consolidation after high-dose chemotherapy and autologous stem cell transplantation improves the rate of CR.8
The definitions of progression of cardiac and renal involvement in AL amyloidosis have also been established and validated in several large collaborative studies.4,9,10 These studies consistently showed that cardiac progression predicts shorter survival, and renal progression predicts earlier need of dialysis.4,9,10 Although criteria for clonal disease progression were established by consensus of the experts of the International Society of Amyloidosis in 2005,11 at present, there is no consensus on the best time to resume therapy in relapsing patients, and few studies address this important and difficult issue.12-14
The Boston University Group reported the outcome of 82 patients who had recurrent detectable clonal disease after having achieved CR with autologous stem cell transplant. In 13 of these patients (16%), the reappearance of the monoclonal component was not accompanied by organ progression and did not require additional treatment. Unfortunately, in this study only a minority of patients had measurements of FLC, and the extent of clonal relapse could not be quantified.15
The Mayo Clinic investigators analyzed the patterns of relapse/progression and the timing of second-line therapy in 235 patients initially treated with autologous stem cell transplant.14 They reported that patients who reached VGPR may be able to tolerate a gradual rise in dFLC, assuming they did not have a dFLC <50 mg/L at diagnosis. Thus, waiting for a dFLC of 50 mg/L (as long as it remains under 30% of the diagnostic level) appears safe in patients who had previously achieved VGPR or better. On the other side, nearly a quarter of patients who had organ relapse or progression had a baseline dFLC <50 mg/L at diagnosis, and nearly two-thirds of these same patients had a dFLC <50 mg/L at a time when they had documented organ relapse or progression. These findings confirm that even low concentrations of serum amyloid light chain are sufficient to cause further deterioration of organ function. Importantly, patients with organ progression at the time of second-line therapy had significantly inferior survival compared with patients whose organ function did not deteriorate (P = .02), suggesting intervention before organ progression.
We analyzed 259 patients who responded to upfront nontransplant therapy in order to assess criteria for starting second-line therapy.16 Almost two-thirds of patients who started rescue therapy had “high-risk dFLC progression” defined as at least a 50% increase in dFLC from the value reached after upfront therapy to an absolute value of ≥20 mg/L that corresponded to at least 20% of the baseline value observed at diagnosis. “High-risk dFLC progression” preceded cardiac progression by a median of 6 months in 85% of cases. Importantly, initiation of rescue therapy after cardiac progression was associated with a median survival of only 17 months, which was significantly shorter than that of patients treated despite that they did not have cardiac progression. In light of the importance of even small increases in dFLC after response, maintenance therapy may have a role in AL amyloidosis, similarly to multiple myeloma. However, no studies addressed maintenance therapy in AL amyloidosis, and prospective, controlled studies are warranted before it can be considered in this disease. Overall, there is agreement of independent studies showing that organ progression should not be awaited to resume anticlone therapy in relapsing patients with AL amyloidosis. Unfortunately, the present criteria for cardiac, renal, and liver progression are not adequate for deciding to resume therapy. Because it is impossible to predict when hematologic relapse or progression will translate into organ progression, resuming treatment should be considered when the amyloid FLC reappears or progresses. Novel criteria for grading the organ response have been recently proposed17 ; similar studies for grading the organ relapse may allow the definition of thresholds for triggering treatment when the minimal deterioration of organ function still does not impact on survival. This may spare unnecessary treatment, particularly in fragile patients. Novel biomarkers for early cardiac and renal progression are needed. Recently, it has been reported that growth differentiation factor-15 (GDF-15) is a powerful predictor of survival and renal outcomes in AL amyloidosis.18 GDF-15 that remained or increased to ≥4000 pg/mL after intervention was associated with end-stage renal disease and dialysis. This deserves investigation in future studies as a potential trigger for retreatment in patients with renal involvement.
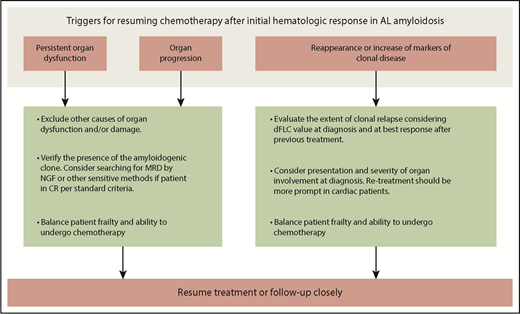
What triggers should be used to reinstitute plasma cell clone-directed therapy among patients with AL amyloidosis? Probably several factors need to be taken into account in addition to the patient performance status, such as the pattern of organ involvement and the dFLC level at diagnosis, the depth of response after upfront therapy, and the increase in dFLC at relapse. A few patients can have organ progression even in the absence of dFLC increase. Additional chemotherapy should be considered if persistence or reappearance of clonal disease (even MRD using flow cytometry or the detection of clonal FLC in serum or urine by sensitive techniques, such as mass spectrometry19 ) is documented and when alternative causes underlying organ progression are excluded. Some other patients may have stable organ dysfunction despite the reappearance or increase of the serum concentration of the amyloid light chain. Unfortunately, in most instances, hematologic relapse foretells an organ progression that is bound to occur in a few months and therefore represents a useful warning that demands careful consideration. The risk of the forthcoming recurrence of a life-threatening and disabling disease should not be underestimated. In these cases, early re-treatment is our first option (Figure 1); however, in fragile patients particularly sensitive to chemotherapy toxicity, attentive monitoring could be considered as an alternative, until novel, robust, and validated criteria will allow us to reliably identify patients in real need of further treatment.
Clinical reasoning for starting second-line therapy after initial response to frontline therapy in AL amyloidosis. Persistent or worsening organ involvement and/or relapse of clonal disease should be the triggers for resuming therapy. Non–amyloid-related causes of organ damage and/or dysfunction should be excluded, in order to avoid unnecessary exposure to chemotherapy. The presence of clonal disease should also be verified before resuming chemotherapy. In some cases, persistence of MRD is sufficient to maintain organ disease or cause organ progression. In the absence of clinically significant organ involvement, clonal relapse should be interpreted based on disease severity and amyloid free light chain level at diagnosis. For instance, clones that were associated with cardiac involvement and those that were able to cause organ involvement at low amyloid free light chain concentration should be treated earlier. Patient’s frailty remains a key issue also at relapse in AL amyloidosis, and the decision to resume chemotherapy should be carefully balanced in light of patient performance status and expected toxicity. NGF, next-generation flow cytometry.
Clinical reasoning for starting second-line therapy after initial response to frontline therapy in AL amyloidosis. Persistent or worsening organ involvement and/or relapse of clonal disease should be the triggers for resuming therapy. Non–amyloid-related causes of organ damage and/or dysfunction should be excluded, in order to avoid unnecessary exposure to chemotherapy. The presence of clonal disease should also be verified before resuming chemotherapy. In some cases, persistence of MRD is sufficient to maintain organ disease or cause organ progression. In the absence of clinically significant organ involvement, clonal relapse should be interpreted based on disease severity and amyloid free light chain level at diagnosis. For instance, clones that were associated with cardiac involvement and those that were able to cause organ involvement at low amyloid free light chain concentration should be treated earlier. Patient’s frailty remains a key issue also at relapse in AL amyloidosis, and the decision to resume chemotherapy should be carefully balanced in light of patient performance status and expected toxicity. NGF, next-generation flow cytometry.
Acknowledgments
This work was supported in part by “Associazione Italiana per la Ricerca sul Cancro–Special Program Molecular Clinical Oncology 5 per mille” grant 9965, CARIPLO “Structure-function relation of amyloid: understanding the molecular bases of protein misfolding diseases to design new treatments” grant 2013-0964, and the Italian Ministry of Health target project grant RF-2013-02355259.
Authorship
Contribution: G.P. and G.M. wrote the paper and revised the final version.
Conflict-of-interest disclosure: G.P. has received honoraria from Jannsen; honoraria and travel support from Prothena; and travel support from Celgene. G.M. has been a consultant for Millennium Pharmaceuticals, Inc, Pfizer, Janssen, Prothena, and IONIS.
Correspondence: Giampaolo Merlini, Foundation IRCCS Policlinico San Matteo, Viale Golgi, 19, 27100 Pavia, Italy; e-mail: gmerlini@unipv.it.