On page 139 of the Hematology 2014 American Society of Hematology Education Program, a sentence is omitted from the last footnote of Table 1. The footnote should have read, “†Enrollment as of June 2014. Of enrolled patients, 173 have early-stage disease.” Also on page 139, there is an error in Figure 3E. An incorrect version of the panel was included. The corrected Table 1 and Figure 3 are shown below.
Prospective noncontrolled response-adapted studies in adult early-stage (I-II) HL

SCT, stem cell transplantation; and NA, not available.
* Biopsy done if PET-4 is positive; patients receive same therapy as PET-4− for negative biopsy and salvage therapy for positive PET-4 biopsy.
†Enrollment as of June 2014. Of enrolled patients, 173 have early-stage disease.
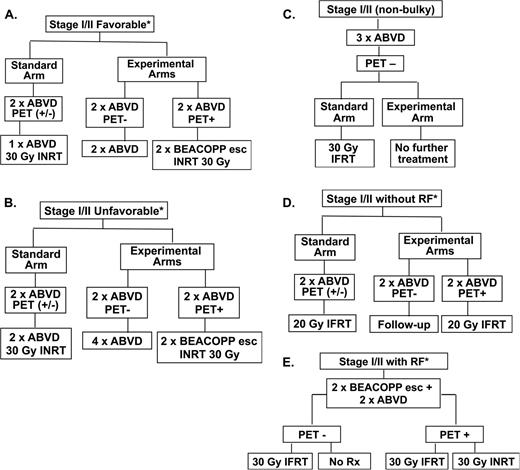
Clinical trial designs of recently completed and ongoing phase 3 randomized studies of response-adapted therapy for adult early-stage HL. (A) EORTC/LYSA/FIL H10F study. *None of the following present: large mediastinal mass, age >50 years, high ESR, or 4 or more areas. (B) EORTC/LYSA/FIL H10U study. *Any of the following present: large mediastinal mass, age >50 years, high ESR, and/or 4 or more areas. (C) UK-led RAPID study; all PET 3+ patients received a 4th cycle of ABVD followed by 30 Gy of IFRT. (D) GHSG HD16 favorable trial. *None of the following present: large mediastinal mass, extranodal disease, high ESR, or 3 or more areas. (E) GHSG HD17 unfavorable trial. *Any of the following present: large mediastinal mass, extranodal disease, high ESR, and/or 3 or more areas. High ESR for all of above defined as: >50 mm without B symptoms or ESR <30 mm with B symptoms. esc indicates escalated; ESR, erythrocyte sedimentation rate; LYSA, Lymphoma Group and the Lymphoma Study Association; FIL, Fondazione Italiana Linfomi; and pts, patients.
Clinical trial designs of recently completed and ongoing phase 3 randomized studies of response-adapted therapy for adult early-stage HL. (A) EORTC/LYSA/FIL H10F study. *None of the following present: large mediastinal mass, age >50 years, high ESR, or 4 or more areas. (B) EORTC/LYSA/FIL H10U study. *Any of the following present: large mediastinal mass, age >50 years, high ESR, and/or 4 or more areas. (C) UK-led RAPID study; all PET 3+ patients received a 4th cycle of ABVD followed by 30 Gy of IFRT. (D) GHSG HD16 favorable trial. *None of the following present: large mediastinal mass, extranodal disease, high ESR, or 3 or more areas. (E) GHSG HD17 unfavorable trial. *Any of the following present: large mediastinal mass, extranodal disease, high ESR, and/or 3 or more areas. High ESR for all of above defined as: >50 mm without B symptoms or ESR <30 mm with B symptoms. esc indicates escalated; ESR, erythrocyte sedimentation rate; LYSA, Lymphoma Group and the Lymphoma Study Association; FIL, Fondazione Italiana Linfomi; and pts, patients.