Inhibition of thrombin and/or factor Xa is the mechanism of action of several antithrombotic agents. For example, standard heparin catalyzes the rate of inactivation of thrombin and factor Xa by antithrombin. Antithrombin undergoes a conformational change when it binds to a specific sequence in heparin that facilitates its interaction with factor Xa. In contrast, thrombin binds indiscriminately along the linear heparin polysaccharide molecule, which facilitates its diffusion to bind to and be inhibited by antithrombin. Low-molecular-weight heparins and fondaparinux, which retain the antithrombin binding site but are not long enough to facilitate the diffusion of thrombin, selectively enhance inhibition of factor Xa by antithrombin. Additionally, thrombin can be selectively targeted by specific inhibitors, including bivalirudin and argatroban. There is a fine line between the degree of anticoagulation necessary for inhibition of coagulation for antithrombotic purposes and that which pathologically disrupts hemostatic function. Thus, the option to reverse the anticoagulant activity of an antithrombotic agent is desirable, as in the case with reversal of heparin activity by protamine.
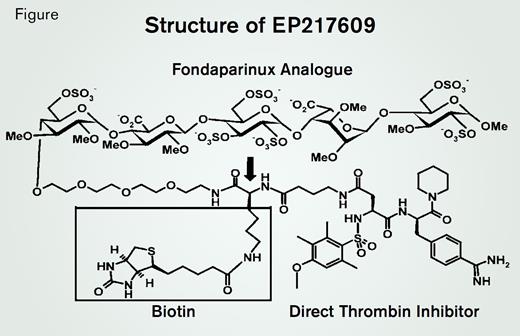
In a phase 2 clinical trial, EP217609 is being tested as a reversible antithrombotic in patients undergoing cardiopulmonary bypass surgery. The drug contains three functional sites: a fondaparinux analog for factor Xa inhibition, an active-site directed thrombin inhibitor, and a biotin tag that provides a mechanism for reversal of anticoagulant activity through binding to avidin (Figure). The dual inhibition of thrombin and factor Xa, together with the antithrombin dependence of the factor Xa component, present a challenging task to investigators attempting to establish the basis of the antithrombotic properties of EP217609. Steven Olson and colleagues at the University of Illinois at Chicago have developed elegant biophysical methods to characterize the inhibition of coagulation enzymes by antithrombin and the mechanism of action of heparin,1-4 and now, in a Plenary Paper in Blood, this group reports delineation of the biochemical basis of the potency and selectivity of EP217609. Based on the intrinsic fluorescence of antithrombin that occurs when the molecule interacts with EP217609, they found that antithrombin binds EP217609 with high affinity (30 nM). Additionally, kinetic modeling revealed that the rate of inhibition of factor Xa by the EP217609-antithrombin complex was more than 20-fold higher than that of other proteases. EP217609 competitively inhibited small substrate hydrolysis by thrombin, a property that allowed the authors to determine that EP217609 binds thrombin rapidly (kon > 107 M-1s-1) and with very high affinity (30-40 pM). In contrast, affinity of EP217609 for other coagulation enzymes was over two orders of magnitude lower than for thrombin.
The binding of EP217609 to antithrombin could affect its interaction with thrombin and vice versa. The inhibition by EP217609 of thrombin-catalyzed small substrate hydrolysis revealed a modest decrease in affinity of the EP217609-antithrombin complex for thrombin compared with uncomplexed EP217609. The reciprocal experiment in which the kinetics of inhibition of factor Xa by the EP217609-antithrombin complex were analyzed in the presence and absence of thrombin revealed a similar modest decrease in inhibition when thrombin was present in the reaction mixture. Together, these results indicate that the two inhibitor moieties of EP217609 (Figure) act nearly independently.
Although the biotin tag was designed to provide a mechanism for rapid in vivo clearance of EP217609 through binding to avidin, the authors found that avidin weakened the direct inhibitory effect of EP217609 on thrombin by over two orders of magnitude. Avidin also reduced the rate constant for EP217609-antithrombin inhibition five-fold and decreased the affinity of EP217609 for antithrombin 20- to 30-fold.
In Brief
The study by Olson et al. provides a rigorous thermodynamic and kinetic analysis of the mechanism of action of EP217609, a complex antithrombotic agent that is undergoing clinical evaluation. It builds on a broad foundation of work involving the study of coagulation enzymes, antithrombin, and heparin by many groups over many years and provides a rational underpinning for the development of novel antithrombotic agents.
References
Competing Interests
Dr. Lollar indicated no relevant conflicts of interest.