Abstract
With the advent of effective antiretroviral therapy, the treatment of patients with HIV-related malignancies, especially lymphoma, has greatly improved, yielding results comparable to those seen in patients with lymphoma unrelated to HIV. The platform of transplantation of hematopoietic stem cells has facilitated studies of genetically modified stem cells engineered to express antiretroviral genes to resist infection by the HIV virus, testing the concept that engraftment of these cells will lead to HIV resistance and elimination of the reservoir of virus in the body. Results in patients with HIV and lymphoma have now led to studies that will test these principles in HIV patients without concomitant malignancy. In addition, in a patient with HIV and acute myeloid leukemia, the success of an allogeneic transplantation from an unrelated donor carrying a mutation in the CCR5 genes has demonstrated that, in principle, such an approach could also lead to cure of patients with HIV. Case studies in HIV patients with leukemia undergoing allogeneic transplantation also suggest that there may be a therapeutic effect on the HIV reservoir that could alter the natural history of HIV in the allogeneic setting.
Introduction
It has been more than 20 years since the concept was first proposed of using a preparative transplantation regimen to destroy the lymphohematopoietic reservoir of HIV.1 Such an approach posed the dilemma of how to protect donor cells from reinfection by the virus, a question that must be addressed for successful hematopoietic stem cell transplantation (HSCT). Currently, the predominant role of HSCT in the setting of HIV infection is in the treatment of AIDS patients who have concomitant hematologic malignancy. It has been in this setting that parallel questions of control of malignancy and HIV have been posed, seeking to accomplish the dual purpose of cure of the malignancy and control, if not cure, of the HIV infection.
Autologous transplantation for AIDS-related lymphoma
Hematopoietic cell therapy in HIV/AIDS has been most applicable in the setting of AIDS-related lymphoma. Since the advent of combination antiretroviral therapy (cART), the survival of patients with HIV infection has improved dramatically, but as life-threatening opportunistic infections have become less common, AIDS-related cancers have become a more predominant source of morbidity and mortality.2 The majority of associated cancers are non-AIDS defining, such as squamous cell cancer, lung cancer, and melanoma, which have increased as the HIV-infected population has aged. AIDS-defining cancers such as B-cell lymphoma are of significant concern3 and are predominantly of the B-cell subtypes having aggressive histology, such as diffuse large B-cell lymphoma and Burkitt's lymphoma, but also plasmablastic and primary effusion lymphoma. In addition, there has been a rise in HIV-related Hodgkin lymphoma, presumably reflecting immune stimulation driven by the higher CD4 counts.4 In general, AIDS-related lymphomas are typified by advanced age at presentation and a more aggressive clinical course.
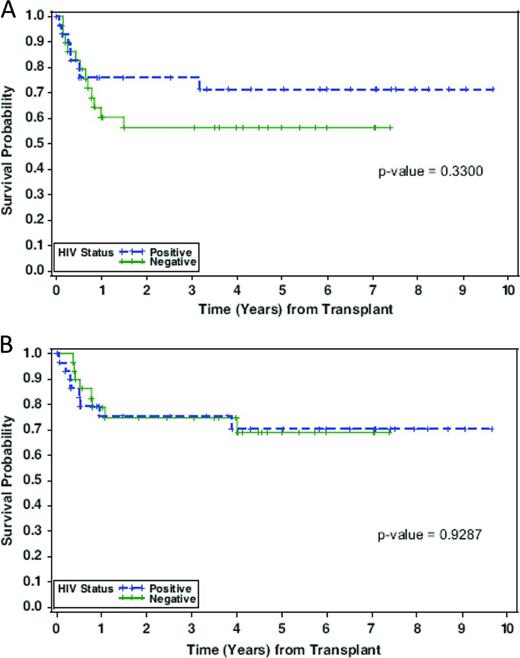
Autologous stem cell transplantation (ASCT) in HIV-positive patients was initially attempted before the era of cART, and whereas the posttransplantation course was complicated by multiple opportunistic infections, it demonstrated the feasibility of stem cell mobilization and successful engraftment in the BM milieu of active viral replication. The first real step forward using ASCT in the setting of HIV infection came in the post-cART era in patients with lymphoma but undetectable HIV viral load at the time of treatment.5 The City of Hope experience with advanced lymphoma and HIV focused on patients who were responsive to chemotherapy as treatment for relapse.6 In an initial series of 20 patients with either Hodgkin or non-Hodgkin lymphoma, all patients were on cART and had viral loads < 10 000 gc/mL by RT-PCR. With the exception of one patient who had delayed engraftment until day 23, engraftment of HSCs was similar to that seen in HIV-negative patients. At the time of the initial report, progression-free survival was 85% and overall survival 81%, with a low transplantation-related mortality and no patient deaths from opportunistic infection. Although an attempt was made to continue cART therapy throughout the transplantation period, only half of the patients were able to tolerate the medications due to nausea and mucositis; however, control of virus after transplantation was readily achieved. This experience was evaluated as a case-control analysis of patients with HIV-related lymphoma compared with HIV-negative patients with the same lymphoma histologies.7 The nonrelapse mortality was similar between the groups and the 2-year disease-free survival and overall survival were similar at 75% (Figure 1). These results confirmed another retrospective case control study comparing 53 HIV-infected lymphoma patients with negative controls adjusted for histology, International Prognostic Index score, and disease status, demonstrating overall survivals of 61% versus 70%, respectively.8,9 A European Group for Blood and Marrow Transplantation (EBMT) retrospective multicenter experience with 68 high risk patients with HIV and lymphoma in first complete remission at 20 institutions reported a progression-free survival of 56% after ASCT.10 Table 1 shows the outcomes of ASCT for high-risk AIDS lymphoma patients from 6 different studies.8-13
Probability of disease-free survival (A) and overall survival (B) by HIV-1 status. Green indicates HIV-1−; blue, HIV-1+.7
Probability of disease-free survival (A) and overall survival (B) by HIV-1 status. Green indicates HIV-1−; blue, HIV-1+.7
Outcome of ASCT in high-risk AIDS lymphoma
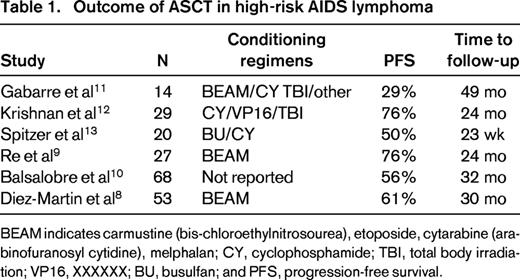
BEAM indicates carmustine (bis-chloroethylnitrosourea-BCNU), etoposide, cytarabine (arabinofuranosyl cytidine), melphalan; CY, cyclophosphamide; TBI, total body irradiation; VP16, etoposide; BU, busulfan; and PFS, progression-free survival.
The favorable outcome of salvage therapy in AIDS lymphoma prompted investigators to determine the potential effect of myeloablative conditioning on the HIV reservoir. To address whether the transplantation procedure itself affected HIV viral infection, Cillo et al performed a retrospective analysis of frozen specimens from patients on cART who had no detectible HIV RNA in plasma.14 Specimens before and after ASCT were studied with single-copy–sensitive assays for HIV RNA and DNA as a surrogate measure of the HIV reservoir. Despite the absence of detectable HIV RNA in the plasma using conventional methods, 9 of the 10 patients were found to have detectable HIV DNA. The transplantation recipients were likely “reinfected” with endogenous virus under cover of cART, and the conclusion was that the myeloablative chemotherapy followed by cART did not have a substantive effect on the HIV reservoir, suggesting that dose-intense cytoreductive therapy by itself is not sufficient to eliminate HIV infection. This finding has prompted efforts to modify the infused T cells so that they are resistant to HIV.
Genetic modification of HSPC for HIV/AIDS
The goal of an HSCT-based strategy for HIV/AIDS is to generate a new immune system to control HIV infection while at the same time destroying the endogenous reservoir, thereby curing the infection. Success would be largely predicated upon the creation of a durable (HIV-resistant) immune system through transplantation of innately resistant or genetically altered hematopoietic stem/progenitor cells (HSPCs); xenogeneic, allogeneic, and autologous stem cell sources have all been tested. The feasibility of transferring HIV resistance via HSPCs was demonstrated when an AIDS patient in Berlin with acute myeloid leukemia received a transplantation with HLA-matched, unrelated donor HSPCs containing a homozygous 32-bp deletion in the chemokine receptor 5 gene (CCR5Δ32/Δ32).15 Individuals who are homozygous at this locus do not express CCR5 on the surface of CD4+ T cells, and therefore cannot be infected with CCR5 (R5)-tropic HIV. The recipient attained complete hematopoietic reconstitution with the donor graft, suspended cART early after transplantation, and has remained with undetectable HIV RNA in the blood and HIV DNA in the tissues using single-copy–sensitive PCR methods for at least 4 years after transplantation. Based on this case, genetic modification and engraftment of HSCs to confer HIV resistance might be a promising alternative to homozygous natural deletions in potential donors, addressing the larger question of cure of HIV in nonmalignancy HIV patients. Retroviral or lentiviral transfer of HIV resistance genes into HSCs would presumably generate progeny that are resistant to reinfection by any endogenous virus and, in the absence of a suitable reservoir, the original virus would be eliminated.
A phase 2 trial of gene-modified autologous cell therapy with a trial design using cART interruption but with no pretransplantation conditioning led to only transient effects on HIV levels.16 Given these observations and the feasibility and advisability of ASCT for HIV patients who have concomitant lymphoma, studies have been conducted in patients with high-risk AIDS-related lymphoma, for whom myeloablative ASCT is necessary. Recent studies show that patients undergoing high-dose chemotherapy and ASCT infused with a combination of gene-modified and unmodified stem cells could be successfully engrafted without affecting normal hematopoiesis.17 Another promising single-treatment therapeutic option involves autologous transplantation of CD34+ cells transduced with a short hairpin RNA against CCR5; this strategy has shown successful low-toxicity long-term downregulation of CCR5 in macaques.18 In a macaque simian-human immunodeficiency virus (SHIV) model, autologous transplantation of CD34+ cells transduced with the mC46 membrane-bound viral fusion inhibitor and a chemotherapy-resistance marker has demonstrated stable protection from viral infection and has been suggested as a potential alternative to antiretroviral therapy.19
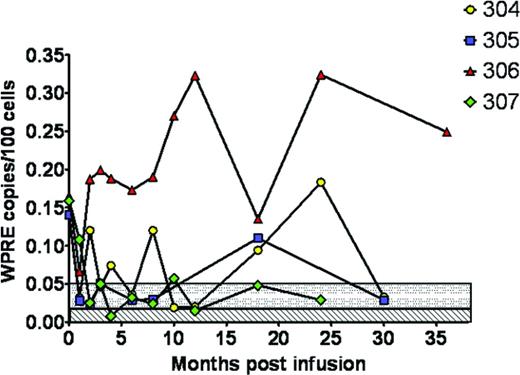
At the City of Hope, 4 patients with HIV undergoing transplantation for recurrent lymphoma were treated with HSPCs that had been modified by a lentivirus containing 3 different RNA-based antiretroviral genes without serious adverse events attributable to the research HSPC product (Figure 2).17 Gene marking was present in all patients but at a level < 0.35% of PBMCs. This study demonstrated the safety and feasibility of the approach after myeloablative conditioning, but also showed the limitation in the ability of the autologous approach to engraft adequate numbers of gene-modified cells. Given that clinical trials on the correction of human genetic diseases using gene-modified HSPCs have shown success using busulfan-based regimens, these approaches are now being evaluated in the setting of HIV/AIDS.20 In addition to the introduction of HIV resistance genes into hematopoietic cells, other approaches include zinc finger nucleases targeted to CCR5, which has been shown to protect from HIV infection in animals.21,22 Zinc finger–based strategies have the advantage of a transient cell treatment ex vivo that produces a permanent genetic mutation; preclinical development of this approach suggests that it should be feasible in human clinical trials.
Allogeneic transplantation in HIV/AIDS
The advent of effective cART has affected not only the ability to treat patients with lymphoma and Hodgkin lymphoma who need ASCT, but also has facilitated allogeneic transplantation in those patients for whom an ASCT would not be suitable. Solid organ transplantation has actually paved the way for stem cell transplantation because this procedure laid to rest concerns about graft rejection and demonstrated the feasibility of maintaining HIV-infected patients on chronic immunosuppression without worsening the underlying HIV infections as long as cART therapy is continued. The “Berlin patient” index case, showing that transplantation of hematopoietic cells from a donor with homozygous constitutional deletion mutations of the CCR5 receptor could be cured of his underlying AIDS infection, combined with the observation of the apparent modification of the HIV reservoir after non-CCR5–deleted matched unrelated donor transplantations, has raised the possibility of a graft versus HIV effect that could be used therapeutically.
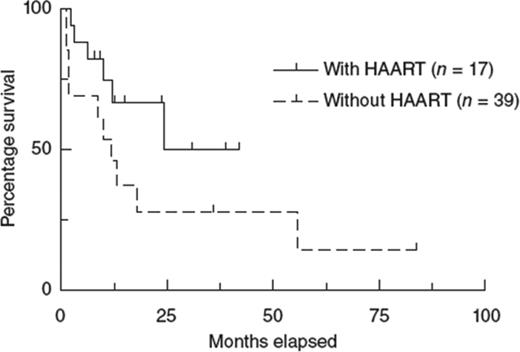
In the pre-cART era, allogeneic transplantation was completely unsuccessful in the setting of HIV infection, in large part due to high infection-related mortality.23-25 A retrospective study from the EBMT confirmed the negative outcome in the pre-cART era, but showed that the introduction of cART has improved the overall survival of patients undergoing allogeneic transplantation (Figure 3).24
Kaplan-Meier estimation of the survival of HIV-1–infected patients after allogeneic HSCT during the period 1983-2010. HAART here is equivalent to cART referred to in the text. Used with permission from Hutter and Zaia.24
Kaplan-Meier estimation of the survival of HIV-1–infected patients after allogeneic HSCT during the period 1983-2010. HAART here is equivalent to cART referred to in the text. Used with permission from Hutter and Zaia.24
The pattern of immune recovery after allogeneic transplantation in HIV patients shows that the CD4 compartment does have capacity to recover after stem cell transplantation.26 The question is whether, similar to the GVL effect that eradicates malignant cells, a graft-versus-HIV effect may attack the cellular compartment containing the HIV reservoir. The outcome in 2 HIV-infected recipients of reduced intensity conditioning for allogeneic transplantation demonstrated that reconstitution of the T-cell CD4 subsets was similar to that seen in other nonmyeloablative transplantations in HIV negative recipients and that the new HIV-negative donor cells could target HIV epitope specificities that were different from the pretransplantation patterns of the recipient.26 In the patient who achieved full donor chimerism, there was also loss of detectable HIV proviral DNA. This may suggest that the pool of latently infected lymphocytes died out after transplantation, a phenomenon not yet seen in the ASCT setting. Adding to this suggestive but anecdotal data is a series of patients demonstrating that all HIV-infected recipients of reduced intensity transplantations on cART survived, in remission and off immunosuppression at 1 year of follow-up, with 2 patients remaining HIV RNA negative (Figure 2)16 by clinical PCR assay.27
Two patients heterozygous for the CCR5Δ32 mutation who received reduced intensity conditioning from donors with wild-type CCR5 have been followed for changes in HIV peripheral blood reservoirs.28 In-depth analysis of the HIV reservoir in peripheral blood, coreceptor use, and specific antibody responses were performed on samples up to 3.5 years after transplantation. Although HIV DNA was readily detected in peripheral blood mononuclear cells before and 2 to 3 months after transplantation, HIV DNA and RNA were undetectable in the peripheral blood mononuclear cells, CD4, and plasma at 21 and 42 months after transplantation. This loss of detectable HIV correlated with full donor chimerism, development of GVHD, and decreases in HIV-specific antibody levels. These patients demonstrate the ability of donor cells to engraft without evidence of ongoing HIV infection, suggesting that HIV replication may be fully suppressed during cART and that it does not contribute to the maintenance of viral reservoirs in the peripheral blood in patients undergoing transplantation. Therefore, engraftment, even with wild-type CCR5 donor cells, can lead to a sustained reduction in the size of the peripheral reservoir of HIV. Currently, a formal study of allogeneic transplantation for treatment of hematologic malignancies in HIV patients by the Blood and Marrow Clinical Trials Network (www.clinicaltrials.gov identifier NCT01410344) will help to resolve the question of whether an allogeneic attack on the HIV reservoir could be a curative strategy. Such a strategy is not without its concerns, because patients are often unable to continue cART therapy during the recovery phase posttransplantation, leading to a rapid increase in HIV viral load. However, this viral proliferation is typically reversed once gastrointestinal function improves after transplantation and cART therapy is resumed. Nevertheless, the data in the allogeneic setting suggests that an allogeneic approach to patients with HIV infection may be feasible, but is limited by the risks of GVHD. Therefore, this approach is not yet ready to be used in the patients with HIV who do not have a standard indication for allogeneic transplantation for treatment of underlying malignancy.
Another potential approach to engrafting patients with natural HIV resistance uses umbilical cord blood cells. Studies conducted at the City of Hope by Zaia and Rossi demonstrated that cord blood units could be identified that are either heterozygous or homozygous for the CCR5 deletion and proposed the concept that cord blood transplantation could be used to engraft HIV-resistant cells.29,30 To date, no successful cord blood transplantation has shown that this would lead to cure of HIV, similar to the “Berlin patient.” A pediatric AIDS patient with relapsed acute lymphoblastic leukemia recently received transplantation at the University of Minnesota with a cord blood unit identified to be CCR5 double mutant; however, due to complications, the patient did not survive long enough to determine the impact of the donor graft on the HIV infection.
Summary
The emerging data and clinical trials in patients with HIV infection suggest that transplantation of autologous gene-modified cells has the potential to alter the relationship between the virus and the human host, modifying the natural history of the disease and possibly reducing or eliminating the need for antiretroviral drugs to protect the immune system from the ravages of the virus. Such an approach requires that a resistant population of immune cells lead to immunocompetence and possibly mediate antiretroviral immune responses as well. A complete cure for the disease such that patients are clear of virus, immune competent, and noninfectious would require elimination of both the infected T cells and the reservoir of virus in the body. Emerging studies in allogeneic recipients will help to determine whether a naturally occurring or engineered protected donor-derived immune system has the capacity to eliminate all HIV infected host cells, resulting in a functional cure of the disease.
Disclosures
Conflict-of-interest disclosure: S.J.F. declares no competing financial interests. J.A.Z. is on a data monitoring committee for Bluebird Bio and Calimmune and holds patents with or receives royalties from Vical. Off-label drug use: None disclosed.
Correspondence
Stephen J. Forman, MD, Chair, Department of Hematology and Hematopoietic Cell Transplantation, City of Hope Comprehensive Cancer Center, 1500 E Duarte Road, Duarte, CA 91010; Phone: 626-256-4673, ext. 62404; Fax: 626-301-8256; e-mail: sforman@coh.org.