Abstract
Erythrocytosis results when there is an increased red cell mass and thus an increased hemoglobin. The causes can be divided into primary intrinsic defects of the erythroid progenitor cell and secondary defects, where factors external to the erythroid compartment are responsible. Both can then be further divided into congenital and acquired categories. Congenital causes include mutations of the erythropoietin receptor and defects of the oxygen-sensing pathway including VHL, PHD2 and HIF2A mutations. When fully investigated there remain a number of patients in whom no cause can be elucidated who are currently described as having idiopathic erythrocytosis. Investigation should start with a full history and examination. Having eliminated the common entity polycythemia vera, further direction for investigation is guided by the erythropoietin level. Clinical consequences of the various erythrocytoses are not clear, but in some groups thromboembolic events have been described in young patients. Evidence is lacking to define best management, but aspirin and venesection to a target hematocrit should be considered.
The average adult produces 2.3 × 106 red cells every second.1 These red cells contain hemoglobin (Hb), which is the means whereby oxygen is supplied to the tissues. There are precise homeostatic mechanisms to ensure sufficient, but not excess, red cell production. Any imbalance in the homeostatic mechanisms can lead to excess red cell production known as erythrocytosis.
The exact extent of an erythrocytosis is ascertained by measuring the red cell mass. The red cell mass is defined as increased if it is greater than 125% above that expected for sex and body mass.2 If this occurs, then the subject has an absolute erythrocytosis. The presence of an absolute erythrocytosis is reflected in the fact that the Hb and hematocrit (Hct) are also increased. The various parameters do not completely reflect each other as other factors may affect the measurements; however, an Hct of 0.60 or greater is always associated with an increased red cell mass.3 An Hb above 18.5 g/dL in a male or 16.5 g/dL in a female or an Hct above 0.52 in a male or 0.48 in a female suggests that there is an erythrocytosis. It may be necessary to carry out a red cell mass to establish unequivocally that an absolute erythrocytosis is present.
Classification of Erythrocytoses
An erythrocytosis can be classified depending on the identified cause. The main division is on the basis of primary causes, where an intrinsic defect in the erythroid progenitor cell is associated with an enhanced response to cytokines; or secondary, where the increased red cell production is driven by factors external to the erythroid compartment, such as increased erythropoietin (EPO) production for any reason. Primary and secondary causes can be classified further as either congenital or acquired (Table 1 ).
Primary Erythrocytosis
Congenital Erythropoietin Receptor Mutations
The cytokine EPO binds to the EPO receptor on the surface of the erythroid cell. When this occurs a phosphorylation cascade is initiated. Janus Kinase2 (JAK2) is auto-phosphorylated and then phosphorylates tyrosines in the cytoplasmic region of the EPO receptor. These tyrosines then act as a docking site for the signal transducer and activator of transcription factor 5 (STAT5) protein, which homodimerizes and translocates to the nucleus, where it initiates gene transcription, proliferation of erythroid precursors and ultimately the production of red cells. This process is regulated by a mechanism to turn off red cell production. About 30 minutes after EPO binds, the phosphatase SHP1 is recruited to the receptor and dephosphorylates the receptor and JAK2. The receptor then goes on to be ubiquitinated and degraded in the proteasome.4
A number of mutations have been described in the EPO receptor that results in a truncation of the protein so that the JAK2-binding site is preserved but the SHP1-binding site is lost. These mutations result in a receptor that is “switched on” to stimulate red cell production but has no “switch off” mechanism. Thus they result in continued red cell production and erythrocytosis in the presence of low EPO levels, as the receptor only needs an initial signal to start red cell production. The known mutations result in the loss of between 57 and 127 amino acids from the receptor. At least 14 different mutations have been described in single families, although several, including the original one described,5 have arisen independently in different families (reviewed in Percy6).
Acquired Polycythemia Vera JAK2 mutations
The classical acquired primary erythrocytosis is polycythemia vera. Patients with polycythemia vera have an acquired abnormal clone, an erythrocytosis and frequently an increased white cell and platelet count. There are now well-accepted diagnostic criteria7,8 for polycythemia vera. The majority of patients have a clone of cells with mutated JAK2 where there is a gain-of-function mutation in exon 14, Val617Phe, which leads to a constitutively activated JAK2.9 A small minority of patients, who are negative for the JAK2V617F mutation but have a polycythemia vera–like phenotype, have been found to have mutations in exon 12 of JAK2.10 Individuals with these mutations tend to have a predominately erythroid phenotype. Many were described as idiopathic erythrocytosis before the discovery of the clone.11
Secondary Erythrocytoses
The Oxygen-sensing Pathway
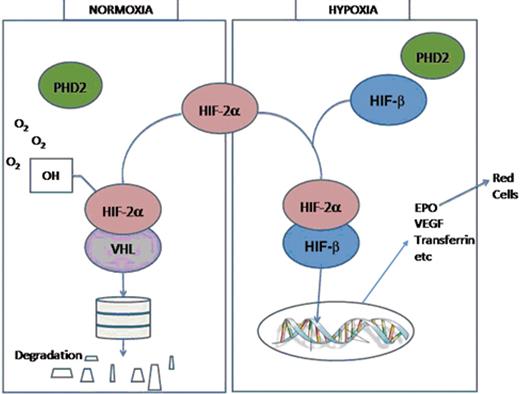
The human has sensitive mechanisms for the maintenance of erythropoiesis in response to hypoxia. This system involves a number of proteins: the prolyl hydroxylases (PHDs), which exist in the isoforms PHD1, PHD2 and PHD3; hypoxia-inducible factor (HIF), which is composed of an unstable alpha subunit and a stable beta subunit; and the von-Hippel-Lindau tumor suppressor protein (VHL). HIF-α has three isoforms: HIF-1α, HIF-2α and HIF-3α.
In normoxic conditions the PHDs hydroxylate the α-subunits of HIF. This hydroxylation allows binding by VHL, which is the substrate recognition unit of an E3 ubiquitin ligase complex. This then allows ubiquitination and degradation of HIF in the proteasome and maintenance of low HIF levels. In hypoxic conditions, PHD-catalyzed hydroxylation is diminished, allowing HIF to escape VHL-mediated proteasome degradation. HIF alpha levels then rise, associate with the beta subunit, and bind to the transcriptional enhancer element located 3′ of the EPO gene. HIF then regulates EPO transcription and production. It also regulates the production of a number of other proteins including those involved in glucose uptake, glycolysis, pH regulation and angiogenesis, such as VEGF (Figure 1 ).
Congenital Defects of the Oxygen-sensing Pathway
VHL-Chuvash Polycythemia
The first defect discovered in the oxygen-sensing pathway was in the VHL gene. A large number of people in the Chuvash area in the upper Volga region of Russia were found to have erythrocytosis. Those with erythrocytosis were discovered to be homozygous for a single mutation C598T in the gene leading to a change in amino acid Arg200Trp.12 Four families of Pakistani and Bangladeshi origin were identified with the mutation,13 and a further study suggested that all these cases might have arisen from a single founder 14,000 to 62,000 years ago.14 Another cohort with this mutation has been identified in the Italian island of Ischia.15 A few individuals have been identified who are compound heterozygotes for the C598T mutation and another VHL mutation. There are also some individuals who are heterozygotes only for C598T or another VHL mutation with erythrocytosis in whom a defect on the other allele has not yet been identified (reviewed in Percy6). Homozygotes have been tested and shown to have abnormal cardiopulmonary physiology characteristic of acclimatization to the hypoxia of high altitude.16 They have also been shown to have elevated homocysteine, glutathione and cysteinylglycine levels.17
PHD2
A family was identified with a mutation in the PHD2 gene, a heterozygous C950G resulting in a proline-to-arginine change at codon 317.18 Further PHD2 mutations have been identified: G1112A resulting in an arginine-to-histidine change at codon 371,19 and A1121G resulting in a histidine-to-arginine change at position 374.20 All these mutations have been shown to have abnormal activity in vitro including decreased HIF binding, defective HIF hydroxylase activity and decreased HIF inhibitory activity. Three further mutations in PHD2 have been described, two frameshift mutations and a premature stop codon, all leading to truncation of the protein.21 Further elucidation of the role of the PHDs in erythrocytosis comes from mouse knock-outs. PHD1/3 double deficiency causes erythrocytosis partly by activating the hepatic HIF-2α pathway. In contrast PHD2 deficiency leads to erythrocytosis by activation of the renal EPO pathway.22
HIF2A
The first described mutation in the HIF genes was a gain-of-function mutation in the HIF2A gene present in three generations of a family with erythrocytosis. The affected individuals were heterozygous for G1609T, resulting in a change of glycine to tryptophan at position 537. This results in an altered protein which binds more weakly than control to PHD2, is less hydroxylated, and binds VHL less efficiently than wild-type protein. Induction of downstream genes is significantly increased by the mutation.23 A different amino acid change at 537, Gly537Arg,24 and two mutations at 535, Met535Val25 and Met535Ile,26 and a Pro534Leu mutation27 have been identified. The different mutations exhibit distinct effects on PHD2 binding and VHL interaction.27
Other Congenital Secondary Erythrocytoses
A high-affinity hemoglobin shifts the oxygen dissociation curve to the left. Oxygen is released less readily to the tissues, resulting in tissue hypoxia and a secondary erythrocytosis. Over 90 hemoglobin variants with altered oxygen affinity have been identified, with defects of both the α and β globin genes described. A high-affinity hemoglobin should be ruled out in cases of erythrocytosis.28
The conversion of 1,3 bisphosphoglycerate (BPG) to 2,3 BPG is catalyzed by the enzyme bisphosphoglycerate mutase. Hemoglobin is converted to a low oxygen affinity state by binding to 2,3 BPG and presence of 2,3 BPG shifts the oxygen affinity curve to the right. Deficiency of 2,3 BPG would therefore shift the oxygen affinity curve to the left, and lead to tissue hypoxia and a compensatory erythrocytosis. Deficiency of bisphophoglycerate mutase can produce a deficiency of 2,3 BPG, and such rare cases have been described causing erythrocytosis.29
Acquired Secondary Erythrocytosis
Many clinical conditions can result in an acquired secondary erythrocytosis. Tissue hypoxia is detected and leads to EPO production. This can be a central process, where there is hypoxia due to pulmonary or cardiac disease, or a process leading to decreased oxygen supply such as high altitude or obstructive sleep apnea.30 EPO is produced by the kidney, and any process that leads to hypoxia in the kidney, for example, renal artery stenosis, can result in increased EPO production and thus erythrocytosis.
EPO can also be produced pathologically. A number of EPO-secreting tumors have been described including cerebellar hemangioblastomas, meningomas, parathyroid adenomas and carcinomas, hepatocellular carcinomas, renal cell carcinomas, pheochromocytomas and uterine leiomyomas.31 For example, in a case of a patient with a leiomyoma who had a raised serum EPO it was observed that EPO levels fell rapidly to normal when the tumor was removed. The tumor tissue had increased EPO mRNA expression and EPO protein.32
Finally, EPO can be administered deliberately to produce an erythrocytosis and enhance performance. Administration of exogenous androgens can have the same effect.33
Idiopathic Erythrocytosis
There remain a group of patients with erythrocytosis in whom no cause has been identified. This group is labelled idiopathic erythrocytosis. One third have EPO levels below the normal range. The other two thirds have normal or elevated EPO levels, and so have a secondary erythrocytosis of unknown cause. Defects have been identified in some patients in these groups; however, no mechanism for the erythrocytosis has been identified in the majority of patients.
Investigation of an Erythrocytosis
Once an erythrocytosis has been established identification of the cause is the next focus. This should start with a comprehensive history and examination with exploration for secondary causes such as chronic respiratory disease. Laboratory investigation can then start with simple tests such as a repeat full blood count. The extent and direction of further investigation should be driven by the clinical scenario and initial results. A possibility of polycythemia vera can be confirmed by JAK2 mutation testing.
In the patient who has an erythrocytosis and does not have polycythemia vera or other obvious cause, checking EPO levels is an initial way to guide further investigation. Those with an EPO level below the normal range are likely to have an abnormality of the EPO-signalling pathway, and this pathway should be investigated first. Those with an inappropriately normal (as if the Hb is elevated the normal physiological response is a decreased EPO level) or elevated EPO are likely to have an abnormality of the oxygen sensing pathway; therefore, it is logical to look for defects in these pathways.
There remain a group of patients in whom no cause has yet been identified for the erythrocytosis therefore with idiopathic erythrocytosis.
Clinical Consequences
A raised red cell count will increase the viscosity and thus may have clinical consequences. The clinical groups, however, are very disparate and therefore it is difficult to ascertain a consistent clinical picture. The largest single group of patients with erythrocytosis are those in Chuvashia with autosomal recessive disease homozygous for a single mutation C598T, the VHL mutation. The clinical characteristics of 99 members of this group have been studied retrospectively compared with a group of 64 spouses and 94 age- and sex-matched community controls. Homozygotes had a lower median survival than controls and had an increased mortality risk from cerebro-vascular events and mesenteric thrombosis. They also had a history of more frequent thromboses, lower systolic and diastolic blood pressures and more frequent venous varicosities. As anticipated, the Hb and EPO levels in this group were higher than in controls. Ophthalmologic and imaging studies were carried out in a subgroup of 33 homozygotes and compared with a control group. More old cerebral ischemic lesions and vertebral body hemangiomas were seen in those with Chuvash polycythemia. There were no reports of the tumors found in classical VHL syndrome and no increased mortality from cancer.34 In the other extensive group reported with the C598T mutation, over 50% were affected by hypotension and varicose veins. No cancer or other signs were reported.15
The other erythrocytoses associated with defects of the oxygen-sensing pathway involve reports of isolated families. The index case of a mutation in the PHD2 gene had superficial thrombophlebitis four years after presentation and an affected sibling had paresthesia.18 A further PHD2 patient at presentation had a sagittal sinus thrombosis.19 Other cases presented with inflammatory arthromyalgia, visual symptoms and hypertension (which improved on treatment) and tinnitus.21 One patient with a PHD2 mutation has been reported with a clearly documented recurrent paraganglioma presenting 13 years after the erythrocytosis was first documented. Loss of heterozygosity of the wild-type PHD2 was documented in the tumor, suggesting that PHD2 can act as a tumor-suppressor gene.20
The propositus in the initial family with a HIF2A gene mutation had a deep venous thrombosis at the age of 42 years, 19 years after initial presentation. His affected mother had a myocardial infarction at the age of 63 years and his maternal grandmother was reported alive and well at 89 years of age with the only a history of note being hypertension.23 In a further series of HIF2A mutations, a girl who presented at 16 years had a mesenteric infarct at the age of 21 years, and her father who was known to have erythrocytosis, died of a mesenteric thrombosis aged 41.25 In another case, the 56-year-old parent with erythrocytosis died of a pulmonary embolus.26 Two affected members of another kindred developed pulmonary hypertension in their sixth decade.35 Thus there is clinical evidence from these cases that HIF2A mutations may be associated with recurrent thromboembolic events.
There is little information about the clinical consequences of the other congenital and acquired erythrocytoses as they have mostly been reported as isolated cases. There is a need for an extensive audit of the clinical manifestations of erythrocytosis.
Management of an Erythrocytosis
Management of polycythemia vera has been discussed by many groups. There is little evidence to guide management of erythrocytosis due to congenital defects or in those with idiopathic erythrocytosis. Reducing the Hct by phlebotomy/venesection reduces the blood viscosity and may be of benefit. The facts that support this are scarce. In the Framingham study, higher antecedent Hbs within the normal range were associated with an increased incidence of cerebro-vascular events in both sexes. However, when this was correlated with other factors such as blood pressure and smoking, the Hb was no longer a statistically significant factor.36 This does not provide any guidance in the pathological situation of a markedly raised Hb. Some of the cohort of patients in Chuvashia were venesected, and those who averaged two or more venesections per year were analyzed in relation to the chance of thrombosis. The odds of thrombosis in those venesected was reduced 5.6-fold, but this was not statistically significant.34 This retrospective study does not inform us about the actual Hcts achieved or the relationship of events to actual Hct. A study in polycythemia vera, the European Collaboration on Low-dose Aspirin in Polycythemia Vera (ECLAP) study, showed that the Hct was not associated with occurrence of thrombotic events or mortality, but in this study Hcts were tightly controlled at a reduced level,37 again not really helpful in those with markedly raised Hcts due to erythrocytosis
The evidence for reduction of the Hct to a particular level comes from a small retrospective study in polycythemia vera where the incidence of vascular occlusive episodes is increased in men and women when the Hct is 0.45 or above. This study is the basis for the guidance in erythrocytosis to reduce the Hct to below 0.45 in both sexes.38
Due to the lack of evidence each patient with erythrocytosis should be individually assessed. If venesection is judged appropriate, the sparse evidence would suggest reduction of the Hct by venesection if the Hct is greater than 0.54. The target for the Hct with venesection from the retrospective evidence would be 0.45. This may be difficult to achieve and may not make the patient with a high Hb feel better. Thus a target of 0.50 may be a more practical. In those with an increased risk of thrombosis, previous thrombosis, peripheral vascular disease, diabetes or hypertension, venesection should be considered at Hcts less than 0.54.39 Cytoreductive therapy is not appropriate for the treatment of patients with erythrocytosis in whom there is no evidence of a malignant clone
Aspirin at low doses is widely used as prophylaxis against thrombotic events where this is considered a risk. Perhaps surprisingly, in the Chuvash cohort the use of aspirin was associated with a 2.4-fold increase of thrombosis, but this was not statistically significant. Information on dose of aspirin was not included.34 Therefore, this does not really provide any guidance on the benefit of aspirin therapy in erythrocytosis. In the ECLAP study, the benefit of low-dose aspirin in the prevention of thrombosis was clearly demonstrated in a prospective randomized trial.40 In those with no specific contraindication, low-dose aspirin is relatively safe and may be a benefit and therefore should be given to patients with an erythrocytosis.
Conclusions
Some patients with erythrocytosis should be considered for judicious venesection to a designated target Hct depending on the clinical scenario and events. Low-dose aspirin can be considered in any patient who does not have a specific contra-indication. There is a need for long-term follow-up and audit of treatment, events and outcomes in these rare patients.
Causes of an erythrocytosis.
| Primary Erythrocytosis |
| Congenital |
| Erythropoietin (EPO) receptor mutations |
| Acquired |
| Polycythemia vera (including JAK2 exon 12 mutations) |
| Secondary erythrocytosis |
| Congenital |
| Defects of the oxygen sensing pathway |
| VHL gene mutation (Chuvash erythrocytosis) |
| PHD2 mutations |
| HIF-2α mutations |
| Other congenital defects |
| High oxygen-affinity hemoglobin |
| Bisphosphoglycerate mutase deficiency |
| Acquired |
| EPO-mediated |
| Central hypoxia |
| Chronic lung disease |
| Right-to-left cardiopulmonary vascular shunts |
| Carbon monoxide poisoning |
| Smoker’s erythrocytosis |
| Hypoventilation syndromes including obstructive sleep apnea |
| High-altitude |
| Local hypoxia |
| Renal artery stenosis |
| End-stage renal disease |
| Hydronephrosis |
| Renal cysts (polycystic kidney disease) |
| Post-renal transplant erythrocytosis |
| Pathologic EPO production |
| Tumors |
| Cerebellar hemangioblastoma |
| Meningioma |
| Parathyroid carcinoma/adenomas |
| Hepatocellular carcinoma |
| Renal cell cancer |
| Pheochromocytoma |
| Uterine leiomyomas |
| Drug associated |
| Erythropoietin administration |
| Androgen administration |
| Idiopathic erythrocytosis |
| Primary Erythrocytosis |
| Congenital |
| Erythropoietin (EPO) receptor mutations |
| Acquired |
| Polycythemia vera (including JAK2 exon 12 mutations) |
| Secondary erythrocytosis |
| Congenital |
| Defects of the oxygen sensing pathway |
| VHL gene mutation (Chuvash erythrocytosis) |
| PHD2 mutations |
| HIF-2α mutations |
| Other congenital defects |
| High oxygen-affinity hemoglobin |
| Bisphosphoglycerate mutase deficiency |
| Acquired |
| EPO-mediated |
| Central hypoxia |
| Chronic lung disease |
| Right-to-left cardiopulmonary vascular shunts |
| Carbon monoxide poisoning |
| Smoker’s erythrocytosis |
| Hypoventilation syndromes including obstructive sleep apnea |
| High-altitude |
| Local hypoxia |
| Renal artery stenosis |
| End-stage renal disease |
| Hydronephrosis |
| Renal cysts (polycystic kidney disease) |
| Post-renal transplant erythrocytosis |
| Pathologic EPO production |
| Tumors |
| Cerebellar hemangioblastoma |
| Meningioma |
| Parathyroid carcinoma/adenomas |
| Hepatocellular carcinoma |
| Renal cell cancer |
| Pheochromocytoma |
| Uterine leiomyomas |
| Drug associated |
| Erythropoietin administration |
| Androgen administration |
| Idiopathic erythrocytosis |
Diagram of oxygen-sensing pathway. In normoxic conditions PHD2 hydroxylates HIF-2α which binds VHL. The proteins are then ubiquitinated and degraded in the proteasome. In hypoxic conditions HIF-2α associates with HIF-β, binds to the transcription enhancer element of the EPO gene, which leads to gene transcription and protein production including production of EPO, which drives red cell production.
Diagram of oxygen-sensing pathway. In normoxic conditions PHD2 hydroxylates HIF-2α which binds VHL. The proteins are then ubiquitinated and degraded in the proteasome. In hypoxic conditions HIF-2α associates with HIF-β, binds to the transcription enhancer element of the EPO gene, which leads to gene transcription and protein production including production of EPO, which drives red cell production.
Disclosures Conflict-of-interest disclosure: The author receives honoraria from Shire Pharmaceuticals, Bristol-Myers Squibb and Novartis. Off-label drug use: None disclosed.
References
Author notes
Centre for Cancer Research and Cell Biology, Queen’s University, Belfast, N. Ireland.